Scalable Production of o-Chloromethylphenylglycolic Acid Derivatives via Directed Ortho-Lithiation
Scalable Production of o-Chloromethylphenylglycolic Acid Derivatives via Directed Ortho-Lithiation
The development of efficient synthetic routes for high-value agrochemical intermediates remains a critical priority for the global fine chemical industry. Patent CN1073554C discloses a sophisticated preparation method for o-chloromethylphenylglycolic acid derivatives, which serve as pivotal building blocks for the synthesis of methoxyimino-phenyl glyoxylic ester series microbicides. These compounds are essential precursors in the manufacture of strobilurin fungicides, a class of agricultural chemicals renowned for their broad-spectrum activity and environmental safety profile. The disclosed technology leverages directed ortho-lithiation strategies to achieve precise functionalization on the aromatic ring, overcoming the regioselectivity challenges inherent in traditional electrophilic substitution methods. By establishing a robust pathway from simple benzylamine derivatives to complex glyoxylic acid esters, this innovation offers a compelling solution for manufacturers seeking to optimize their production workflows.
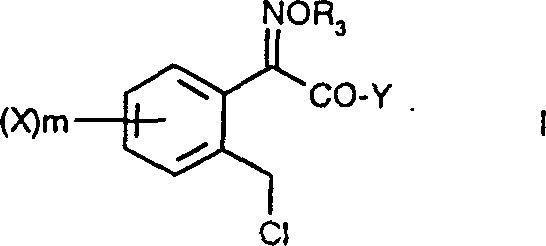
For procurement managers and supply chain directors, the implications of this patent extend beyond mere chemical curiosity; it represents a tangible opportunity for cost reduction in agrochemical intermediate manufacturing. The ability to synthesize these complex structures with high fidelity reduces the reliance on expensive chromatographic purification steps, directly impacting the bottom line. Furthermore, the versatility of the method allows for the generation of various ester and amide derivatives, providing flexibility in meeting diverse customer specifications. As a reliable agrochemical intermediate supplier, understanding the nuances of such proprietary technologies is essential for maintaining a competitive edge in the market. This report delves into the mechanistic details, process advantages, and commercial viability of the method described in CN1073554C, providing a comprehensive analysis for technical decision-makers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for preparing ortho-substituted phenylglyoxylic acid derivatives often rely on Friedel-Crafts acylation or direct halogenation of pre-functionalized aromatic rings. These classical approaches suffer from significant drawbacks, primarily concerning regioselectivity and functional group tolerance. Electrophilic aromatic substitution is inherently governed by the electronic nature of existing substituents, frequently leading to mixtures of ortho, meta, and para isomers that are difficult and costly to separate. In the context of synthesizing fungicide intermediates, where structural purity is paramount for biological efficacy, the presence of isomeric impurities can be detrimental. Moreover, harsh reaction conditions required for direct chlorination or acylation can degrade sensitive functional groups such as esters or oximes, necessitating protective group strategies that add unnecessary steps and reduce overall atom economy. The accumulation of waste streams from these inefficient processes also poses environmental compliance challenges for modern manufacturing facilities.
The Novel Approach
The methodology presented in CN1073554C introduces a paradigm shift by utilizing directed ortho-lithiation (DoL) to install the glyoxylic acid moiety with precision. This approach exploits the directing ability of the aminomethyl group to guide the organolithium reagent specifically to the ortho position of the benzene ring. By generating a reactive aryl lithium intermediate under controlled low-temperature conditions, the process ensures that subsequent reaction with oxalic acid derivatives occurs exclusively at the desired site. This high degree of regiocontrol eliminates the formation of unwanted isomers, thereby streamlining the purification process and significantly enhancing the yield of the target compound. Additionally, the mild conditions employed in the lithiation and acylation steps preserve the integrity of other functional groups, allowing for a more convergent synthesis strategy. The novelty lies not just in the reaction itself, but in the specific sequence of lithiation, acylation, and subsequent functional group transformation that enables the efficient production of high-purity intermediates.
Mechanistic Insights into Directed Ortho-Lithiation and Acylation
The core of this synthetic strategy involves the generation of a benzylic lithium species followed by nucleophilic attack on an electrophilic oxalate. Initially, an N-benzylamine derivative, such as N-benzyl-N,N-dimethylamine, is treated with a strong base like butyllithium in an aprotic solvent such as tetrahydrofuran or ether. The nitrogen atom coordinates with the lithium cation, directing the deprotonation to the adjacent ortho carbon on the aromatic ring. This forms a stable aryl lithium complex, which acts as a potent nucleophile. Upon addition of an oxalic acid derivative, such as dimethyl oxalate or ethyl oxalyl chloride, the aryl lithium attacks one of the carbonyl carbons. This step is critical as it installs the alpha-keto ester functionality directly onto the ring. The reaction temperature is carefully maintained between -50°C and 30°C to prevent side reactions such as over-lithiation or decomposition of the sensitive lithium intermediate.
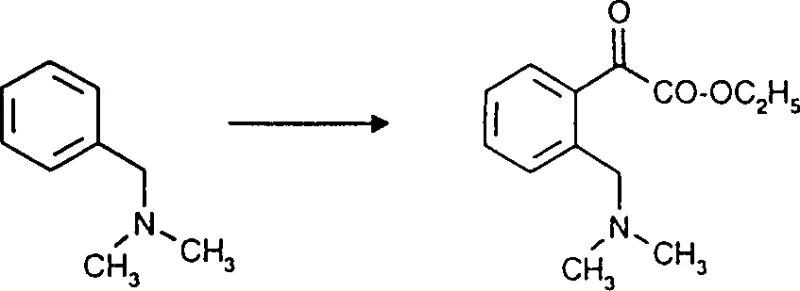
Following the acylation, the resulting intermediate contains an ortho-aminomethyl group which must be converted to the chloromethyl functionality found in the final fungicide precursor. This transformation is elegantly achieved using chloroformates, such as vinyl chloroformate or methyl chloroformate. The reaction proceeds through the formation of a carbamate intermediate, which subsequently undergoes elimination or substitution to yield the chloromethyl group. Alternatively, the keto-ester can be subjected to oximation using O-methylhydroxylamine to form the methoxyimino derivative directly. The choice between chlorination and oximation depends on the specific target molecule required for the downstream synthesis of the active pharmaceutical or agrochemical ingredient. The mechanistic clarity of these steps ensures that the process is reproducible and scalable, minimizing the risk of batch-to-batch variability.
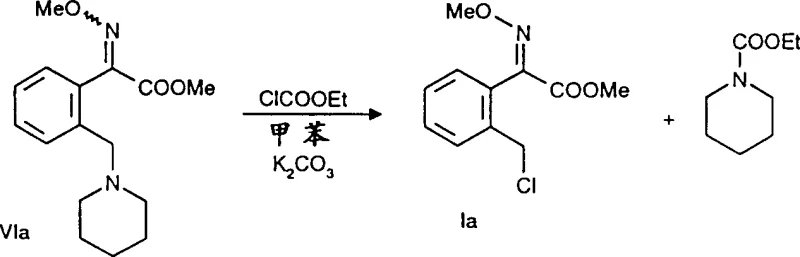
Impurity control is inherently built into this mechanism due to the specificity of the lithiation step. Unlike radical halogenation which can occur at multiple positions on the alkyl side chain or the ring, the DoL process is highly selective. Any unreacted starting material can typically be recovered and recycled, further improving the economic efficiency of the process. Furthermore, the use of stoichiometric amounts of organolithium reagents allows for precise control over the reaction extent, preventing the formation of poly-substituted by-products. The workup procedures described, involving aqueous acid washes and solvent extraction, are designed to remove lithium salts and excess reagents effectively, ensuring that the crude product meets stringent purity specifications before final crystallization. This level of control is essential for producing high-purity agrochemical intermediates that meet global regulatory standards.
How to Synthesize o-Chloromethylphenylglycolic Acid Derivatives Efficiently
The synthesis of these valuable intermediates requires careful attention to reaction parameters, particularly temperature and stoichiometry, to maximize yield and safety. The process begins with the preparation of the lithiated species under an inert atmosphere to prevent quenching by moisture or oxygen. Subsequent addition of the oxalate electrophile must be done slowly to manage the exotherm associated with the nucleophilic attack. Following the formation of the glyoxylic ester, the conversion to the chloromethyl derivative is performed using chloroformates in the presence of a base, often at elevated temperatures to drive the reaction to completion. The detailed standardized synthesis steps see the guide below.
- Perform directed ortho-lithiation on N-benzylamine derivatives using organolithium reagents like butyllithium in aprotic solvents.
- React the resulting lithium complex with oxalic acid diesters or acid chlorides to form the phenylglyoxylic acid ester intermediate.
- Convert the amino group to a chloromethyl group using chloroformates or perform oximation to finalize the fungicide intermediate structure.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting the synthetic route described in CN1073554C offers substantial strategic benefits for procurement and supply chain operations within the agrochemical sector. The primary advantage lies in the simplification of the manufacturing process, which translates directly into operational efficiency and cost savings. By eliminating the need for complex separation of isomeric by-products, manufacturers can reduce the consumption of solvents and energy associated with extensive purification protocols. This streamlined workflow not only lowers the variable cost per kilogram of the intermediate but also increases the throughput capacity of existing production facilities. For supply chain heads, the reliability of this method ensures a consistent supply of high-quality materials, mitigating the risk of production delays caused by quality failures or yield fluctuations common in less selective synthetic routes.
- Cost Reduction in Manufacturing: The use of commodity chemicals such as butyllithium, oxalic acid esters, and chloroformates ensures that raw material costs remain stable and predictable. Unlike processes requiring rare earth catalysts or specialized reagents, this method relies on widely available industrial inputs. The high regioselectivity of the lithiation step minimizes waste generation, reducing the costs associated with waste disposal and environmental compliance. Furthermore, the ability to recycle unreacted starting materials contributes to a higher overall material efficiency, driving down the effective cost of goods sold. These factors combine to create a economically robust manufacturing model that can withstand market volatility.
- Enhanced Supply Chain Reliability: The robustness of the chemistry described allows for flexible sourcing of raw materials, as the reagents used are standard items in the fine chemical supply chain. This reduces dependency on single-source suppliers for exotic catalysts or intermediates. The scalability of the process means that production volumes can be ramped up quickly to meet surges in demand for fungicides during peak agricultural seasons. Additionally, the stability of the intermediates formed allows for safer storage and transportation, reducing logistical risks. A reliable supply of these key building blocks is crucial for maintaining the continuity of downstream API and formulation manufacturing.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents like toluene and ether which are easily recovered and recycled in large-scale operations. The absence of heavy metal catalysts simplifies the regulatory approval process for new manufacturing sites and reduces the burden of heavy metal testing on final products. The cleaner reaction profile results in lower E-factors (mass of waste per mass of product), aligning with modern green chemistry principles and corporate sustainability goals. This environmental compatibility facilitates smoother permitting processes and enhances the company's reputation as a responsible manufacturer in the global agrochemical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. Understanding these aspects is vital for R&D teams evaluating the feasibility of adopting this route and for procurement professionals assessing supplier capabilities. The answers are derived directly from the technical disclosures and experimental data provided in the patent literature, ensuring accuracy and relevance.
Q: What is the primary advantage of the lithiation method described in CN1073554C?
A: The primary advantage is superior regioselectivity. Unlike traditional electrophilic substitution which can yield mixtures of isomers, directed ortho-lithiation ensures functionalization occurs specifically at the position adjacent to the aminomethyl group, drastically simplifying purification and improving overall yield.
Q: Can this process be scaled for commercial production of agrochemical intermediates?
A: Yes, the patent outlines conditions suitable for scale-up, utilizing common industrial solvents like toluene and ether. The use of standard reagents such as butyllithium and oxalic acid esters ensures that raw material sourcing is reliable for large-scale manufacturing.
Q: How does this method impact the purity profile of the final fungicide intermediate?
A: By avoiding harsh halogenation conditions that often degrade sensitive functional groups, this method preserves the integrity of the ester and oxime moieties. The result is a cleaner crude product with fewer by-products, reducing the burden on downstream crystallization and distillation units.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable o-Chloromethylphenylglycolic Acid Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of agrochemical formulations. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demands of both pilot projects and full-scale manufacturing. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and potency. Our facility is equipped to handle the specific safety requirements of organolithium chemistry, guaranteeing a safe and consistent supply of o-chloromethylphenylglycolic acid derivatives.
We invite potential partners to engage with our technical procurement team to discuss how our capabilities align with your project needs. By requesting a Customized Cost-Saving Analysis, you can gain insights into how our optimized processes can reduce your overall manufacturing expenses. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your specific requirements. Let us be your trusted partner in navigating the complexities of agrochemical intermediate supply, ensuring your production lines run smoothly and efficiently.
