Revolutionizing Isoindolinone Production: A Green Heteropolyacid Catalytic Strategy for Commercial Scale
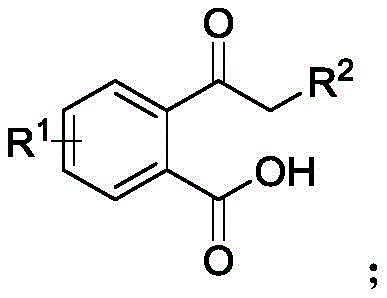
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex nitrogen-containing heterocycles, particularly those featuring quaternary carbon centers which are prevalent in bioactive molecules. Patent CN115894563A introduces a groundbreaking approach for the synthesis of 3,3'-disubstituted isoindolinone compounds, utilizing a dehydration coupling reaction driven by heteropolyacid (HPA) catalysts. This innovation represents a significant paradigm shift from traditional transition metal catalysis, offering a pathway that is not only chemically efficient but also aligns with the stringent environmental and safety standards required by modern regulatory bodies. By employing simple substrates such as 2-acylbenzoic acids, diphenylphosphine oxides, and aliphatic amines, this method achieves high yields under mild conditions, producing water as the sole byproduct. For R&D directors and process chemists, this patent data suggests a viable route to access valuable scaffolds used in subcellular imaging agents and receptor modulators without the baggage of toxic metal residues.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted isoindolinone skeletons has relied heavily on the use of precious transition metal catalysts such as palladium, ruthenium, rhodium, and iridium to facilitate intermolecular or intramolecular cyclization reactions. While these methods can be effective in a laboratory setting, they present substantial drawbacks when evaluated through the lens of commercial manufacturing and supply chain sustainability. The primary concern for procurement and quality assurance teams is the inevitable presence of heavy metal residues in the final active pharmaceutical ingredient (API) or intermediate, which necessitates expensive and time-consuming purification steps to meet strict ppm limits. Furthermore, the market volatility of noble metals introduces significant cost uncertainty, and the toxicity associated with these catalysts complicates waste disposal and environmental compliance, often requiring specialized infrastructure for handling hazardous materials.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes heteropolyacids (HPAs) as a green, solid acid catalyst to drive the three-component coupling reaction, effectively bypassing the need for expensive transition metals. This novel approach leverages the unique acidic properties of HPAs to activate the carbonyl groups of the 2-acylbenzoic acid and facilitate the nucleophilic attack by the amine and phosphine oxide components. The result is a streamlined process that operates at moderate temperatures between 60-120°C and uses environmentally benign solvents like dimethyl carbonate (DMC). By eliminating the reliance on scarce resources and generating only water as a byproduct, this strategy drastically simplifies the downstream processing workflow. For supply chain managers, this translates to a more reliable sourcing model where catalyst availability is not subject to geopolitical mining constraints, ensuring continuous production capabilities.
Mechanistic Insights into Heteropolyacid-Catalyzed Dehydration Coupling
The core of this synthetic breakthrough lies in the mechanistic efficiency of the heteropolyacid catalyst, specifically species like H4SiW12O40, which acts as a strong Brønsted acid to promote dehydration. The reaction mechanism likely involves the protonation of the carbonyl oxygen of the 2-acylbenzoic acid by the HPA, increasing its electrophilicity and making it more susceptible to nucleophilic attack by the amine. Subsequent interaction with the diphenylphosphine oxide facilitates the formation of the critical C-P and C-N bonds required to close the isoindolinone ring and establish the quaternary carbon center at the C3 position. This cascade occurs with high atom economy, as the only atoms lost from the starting materials form water molecules, which can be easily removed to drive the equilibrium forward. Understanding this mechanism is crucial for process engineers, as it highlights the tolerance of the system to various substituents on the aromatic rings, allowing for the synthesis of a diverse library of derivatives without compromising yield or selectivity.
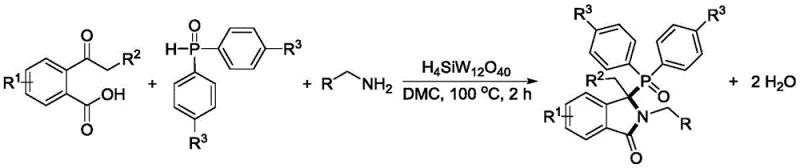
From an impurity control perspective, the use of HPAs offers a distinct advantage over base-mediated or transition-metal catalyzed routes which often generate complex side products or isomeric mixtures. The specificity of the acid-catalyzed dehydration minimizes the formation of oligomeric byproducts or over-oxidized species that are common in radical-based cyclizations. This cleanliness of reaction is paramount for R&D directors who must validate the purity of intermediates before they enter the GMP supply chain. The patent data indicates that yields can reach up to 87% with optimal catalyst loading, suggesting that the reaction kinetics are favorable and that the catalyst remains stable throughout the process duration. This stability reduces the risk of catalyst decomposition products contaminating the final batch, thereby reducing the burden on analytical teams during quality control testing and release.
How to Synthesize 3,3'-Disubstituted Isoindolinone Efficiently
To implement this synthesis in a practical setting, operators must carefully control the stoichiometry and reaction environment to maximize the efficiency of the heteropolyacid catalyst. The standard protocol involves weighing the 2-acylbenzoic acid, diphenylphosphine oxide, and aliphatic amine in a molar ratio ranging from 1:1 to 1:1.5, ensuring that the limiting reagent is fully consumed to prevent difficult separations later. The mixture is dissolved in a preferred organic solvent such as dimethyl carbonate, which provides excellent solubility for the organic substrates while being compatible with the solid acid catalyst. Once the catalyst, typically H4SiW12O40 at 1 to 5 mol%, is added, the reaction vessel is sealed and heated to a temperature between 60-120°C, with 100°C identified as the optimal balance between reaction rate and energy consumption. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining 2-acylbenzoic acid, diphenylphosphine oxide, and aliphatic amine in an organic solvent like dimethyl carbonate.
- Add a heteropolyacid catalyst such as H4SiW12O40 (1-5 mol%) to the mixture under stirring conditions.
- Heat the reaction to 60-120°C for 1-3 hours, then isolate the product via column chromatography after water byproduct removal.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this HPA-catalyzed route offers compelling economic and operational advantages that directly impact the bottom line and production reliability. The most significant benefit is the drastic reduction in raw material costs achieved by replacing volatile precious metal catalysts with inexpensive, commercially available heteropolyacids. This substitution not only lowers the direct cost of goods sold (COGS) but also mitigates the financial risk associated with fluctuating metal prices, allowing for more accurate long-term budgeting and pricing strategies for clients. Additionally, the elimination of heavy metals removes the need for specialized scavenging resins or complex filtration systems, further reducing the operational expenditure related to purification and waste management infrastructure.
- Cost Reduction in Manufacturing: The economic argument for this technology is strengthened by the simplified workup procedure, as the only byproduct is water which can be removed via simple phase separation or evaporation. This eliminates the need for extensive chromatographic purification often required to remove metal salts, leading to substantial savings in solvent consumption and labor hours. Furthermore, the high yield reported in the patent data implies less raw material waste per kilogram of product produced, enhancing the overall material efficiency of the plant. By optimizing the catalyst loading to as low as 1 mol%, manufacturers can achieve significant cost savings without sacrificing throughput, making this route highly competitive for large-volume production.
- Enhanced Supply Chain Reliability: From a logistics perspective, relying on heteropolyacids ensures a more stable supply chain compared to noble metals which are often sourced from geopolitically sensitive regions. The raw materials for this synthesis, including 2-acylbenzoic acids and simple amines, are commodity chemicals with robust global availability, reducing the risk of production stoppages due to material shortages. This reliability is critical for maintaining continuous manufacturing schedules and meeting the just-in-time delivery expectations of downstream pharmaceutical partners. Moreover, the solid nature of the HPA catalyst facilitates easier storage and handling, minimizing the safety risks and regulatory burdens associated with transporting hazardous liquid catalysts or pyrophoric reagents.
- Scalability and Environmental Compliance: The mild reaction conditions described in the patent, operating at atmospheric pressure and moderate temperatures, make this process inherently safer and easier to scale from pilot plant to commercial tonnage. The use of green solvents like dimethyl carbonate aligns with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions, helping companies maintain their environmental permits without costly upgrades. The absence of toxic heavy metals in the waste stream simplifies effluent treatment processes, allowing facilities to meet discharge standards more easily and reduce their environmental footprint. This alignment with green chemistry principles not only ensures compliance but also enhances the corporate sustainability profile, which is becoming a key differentiator in B2B negotiations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this heteropolyacid-catalyzed synthesis route for isoindolinone derivatives. These answers are derived directly from the experimental data and beneficial effects outlined in the patent documentation to provide clarity on process feasibility. Understanding these details helps stakeholders assess the fit of this technology within their existing manufacturing portfolios and quality frameworks.
Q: What are the primary advantages of using heteropolyacid catalysts over traditional noble metals?
A: Heteropolyacids offer a non-toxic, inexpensive alternative to palladium or ruthenium, eliminating heavy metal residue concerns and reducing raw material costs significantly.
Q: How does this method impact the impurity profile of the final isoindolinone product?
A: The reaction produces water as the only byproduct, which simplifies purification and ensures a cleaner impurity profile compared to methods generating toxic side products.
Q: Is this synthesis route suitable for large-scale industrial manufacturing?
A: Yes, the mild reaction conditions (60-120°C) and use of stable solid catalysts make this process highly scalable and safe for commercial production environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3'-Disubstituted Isoindolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this heteropolyacid-catalyzed route for producing high-value pharmaceutical intermediates with superior purity profiles. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market supply is seamless and efficient. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of validating the absence of heavy metal residues, guaranteeing that every batch meets the exacting standards required for drug substance manufacturing. We are committed to leveraging this green chemistry innovation to deliver cost-effective solutions that enhance your supply chain resilience.
We invite you to collaborate with our technical team to explore how this synthesis method can optimize your specific project requirements and reduce overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your strategic goals for reliable pharmaceutical intermediates supplier partnerships.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
