Advanced Synthesis of Stable Gingerol Derivatives for Pharmaceutical and Flavor Applications
The pharmaceutical and fine chemical industries have long recognized the immense therapeutic potential of gingerol, a bioactive compound renowned for its anti-inflammatory, antioxidant, and anticancer properties. However, the commercial exploitation of natural gingerol has been severely hindered by its inherent chemical instability and the complexities associated with extracting it from natural sources. Addressing these critical challenges, the patent CN103159599A, titled "Synthesis process of gingerol derivative," introduces a groundbreaking methodology that transforms the production landscape for these valuable molecules. This intellectual property outlines a robust, scalable, and economically viable route to synthesize novel gingerol derivatives, specifically targeting the limitations of natural extraction. By leveraging a streamlined condensation reaction followed by catalytic reduction, this technology enables the production of stable, high-purity intermediates that retain the biological efficacy of the parent compound while offering superior handling characteristics for downstream formulation.
For research and development directors seeking reliable sources of bioactive scaffolds, this patent represents a significant leap forward. It moves away from the unpredictable yields of agricultural extraction towards a controlled, synthetic environment where parameters can be tightly managed. The ability to produce derivatives such as Gingerol-a-1 and Gingerol-b-1 through a concise one-to-two-step process not only accelerates the timeline for preclinical studies but also lays the foundation for commercial-scale manufacturing. As a reliable gingerol derivative supplier, understanding the nuances of this synthetic pathway is essential for securing a consistent supply chain for next-generation nutraceuticals and pharmaceutical agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the acquisition of gingerol has relied heavily on the extraction and purification of the compound from fresh or dried ginger rhizomes. This conventional approach is fraught with significant inefficiencies and technical bottlenecks that render it unsuitable for large-scale industrial applications. Firstly, the content of 6-gingerol, the most biologically active component, is naturally low within the plant matrix, necessitating the processing of massive quantities of raw biomass to obtain trivial amounts of the target molecule. Furthermore, the chemical nature of natural gingerol is extremely unstable; it is highly susceptible to decomposition when exposed to heat or light, often dehydrating spontaneously into shogaol or undergoing other degradation pathways. This instability complicates storage, transportation, and formulation, leading to substantial batch-to-batch variability. Additionally, the separation and purification of single-component gingerol from the complex mixture of essential oils and oleoresins found in ginger is technically demanding and costly, often requiring extensive chromatographic steps that drive up the final price prohibitively for widespread clinical or commercial use.
The Novel Approach
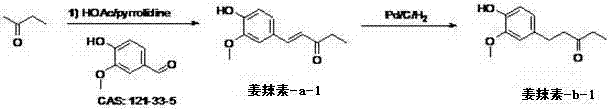
In stark contrast to the arduous extraction methods, the novel approach detailed in the patent utilizes a direct synthetic strategy starting from readily available commodity chemicals. The core innovation lies in the condensation of vanillin with aliphatic aldehydes or ketones within a specifically tuned acid-base solvent system. This reaction efficiently constructs the carbon skeleton of the gingerol derivative in a single operational step, bypassing the need for complex multi-step syntheses typical of older methodologies. The resulting intermediate, a shogaol derivative, is then subjected to a mild catalytic hydrogenation to yield the final saturated gingerol derivative. This synthetic route is characterized by its simplicity, mild reaction conditions ranging from 0 to 50 degrees Celsius, and the use of inexpensive reagents like pyrrolidine and acetic acid. The process dramatically reduces the generation of hazardous waste and eliminates the reliance on agricultural cycles, ensuring a continuous and predictable supply of high-quality material regardless of seasonal variations in crop yields.
Mechanistic Insights into Aldol Condensation and Hydrogenation
The chemical elegance of this process is rooted in the mechanism of the aldol condensation reaction, which serves as the cornerstone for building the characteristic carbon chain of the gingerol derivative. In this transformation, vanillin acts as the electrophilic partner, while an aliphatic ketone, such as butanone, serves as the nucleophile. The reaction is facilitated by a dual solvent system comprising both acidic and basic components, such as acetic acid and pyrrolidine. This unique solvent environment plays a critical role in activating the alpha-carbons of the ketone and stabilizing the transition states, thereby promoting the formation of the carbon-carbon double bond with high regioselectivity. The careful control of the solvent ratio, which can range from 1:1 to 1:100, allows chemists to fine-tune the reaction kinetics, suppressing the formation of unwanted by-products and maximizing the yield of the desired enone intermediate. This level of control is paramount for maintaining the purity profile required for pharmaceutical applications, as it minimizes the presence of structural isomers that could complicate downstream purification.
Following the condensation, the subsequent hydrogenation step is equally critical for defining the final properties of the product. The intermediate shogaol derivative contains an alpha,beta-unsaturated ketone moiety, which is reduced using a palladium on carbon (Pd/C) catalyst under a hydrogen atmosphere. This catalytic hydrogenation selectively targets the carbon-carbon double bond while preserving the carbonyl functionality and the aromatic ring integrity. The mechanism involves the adsorption of hydrogen onto the metal surface of the catalyst, followed by the sequential addition of hydrogen atoms to the alkene bond. This step converts the unstable, conjugated system of the shogaol derivative into the more stable, saturated beta-hydroxy ketone structure of the gingerol derivative. The use of mild conditions during this reduction ensures that no over-reduction of the carbonyl group occurs, thereby preserving the pharmacophore essential for the compound's biological activity. This precise control over the reduction process exemplifies the sophistication of the patented method, delivering a product with enhanced stability compared to its natural counterparts.
How to Synthesize Gingerol-b-1 Efficiently
The practical execution of this synthesis is designed for ease of operation and scalability, making it highly attractive for industrial adoption. The process begins with the preparation of the reaction vessel, where glacial acetic acid and pyrrolidine are combined and cooled to below 5 degrees Celsius to manage the exothermic nature of the initial mixing. Butanone is then added slowly, followed by the dropwise addition of a vanillin solution, ensuring that the reaction temperature remains controlled throughout the addition phase. After the addition is complete, the mixture is allowed to stir at room temperature for an extended period, typically around two days, to ensure complete conversion of the starting materials. The workup procedure is straightforward, involving neutralization with dilute hydrochloric acid, separation of the organic phase, and treatment with sodium bisulfite to remove unreacted vanillin. The crude product is then purified via recrystallization from petroleum ether or silica gel chromatography to yield the pure intermediate, which is subsequently hydrogenated to obtain the final target molecule. For a comprehensive guide on the specific molar ratios and safety protocols, please refer to the standardized operating procedures below.
- Perform aldol condensation of vanillin and butanone in an acetic acid and pyrrolidine solvent system at low temperature, followed by room temperature stirring.
- Isolate the intermediate shogaol derivative (Gingerol-a-1) through acid neutralization, organic phase separation, and recrystallization from petroleum ether.
- Conduct catalytic hydrogenation of the intermediate using Pd/C catalyst in methanol to reduce the double bond, yielding the final gingerol derivative (Gingerol-b-1).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers transformative advantages over traditional extraction methods. The shift from agriculture-dependent sourcing to chemical synthesis fundamentally alters the risk profile of the supply chain, mitigating the vulnerabilities associated with crop failures, seasonal fluctuations, and geopolitical instability in growing regions. By utilizing commodity chemicals like vanillin and butanone as feedstocks, manufacturers can leverage established global supply networks that offer consistent pricing and availability. This transition not only secures the supply line but also opens up opportunities for significant cost optimization throughout the manufacturing lifecycle. The simplicity of the reaction design means that capital expenditure for specialized equipment is minimized, as standard glass-lined or stainless steel reactors commonly found in fine chemical plants are sufficient for production.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the synthetic route and the use of low-cost raw materials. Unlike extraction processes that require vast amounts of solvent and energy to process tons of biomass for minimal yield, this synthetic method achieves high efficiency with a minimal number of unit operations. The elimination of expensive transition metal catalysts in the condensation step, relying instead on organic amines and acids, further reduces the material costs. Additionally, the purification process is streamlined, avoiding the need for preparative HPLC or other costly separation technologies, which translates directly into lower operational expenditures. The overall result is a manufacturing process that is inherently lean, driving down the cost of goods sold and allowing for more competitive pricing in the final market.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the decoupling of production from agricultural cycles. Natural gingerol supply is subject to the vagaries of weather patterns and harvest seasons, leading to potential shortages and price volatility. In contrast, the synthetic production of gingerol derivatives can be scheduled continuously throughout the year, independent of external environmental factors. The raw materials required are produced on a massive industrial scale globally, ensuring that there is no single point of failure in the upstream supply chain. This reliability allows procurement managers to negotiate long-term contracts with greater confidence, knowing that the manufacturer has the capacity to meet demand consistently without interruption. Furthermore, the stability of the synthetic derivatives simplifies logistics, as the products do not require the stringent temperature-controlled shipping often necessary for natural extracts.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, featuring mild reaction conditions that are easily manageable on a large scale. Operating temperatures between 0 and 50 degrees Celsius reduce energy consumption for heating and cooling, contributing to a lower carbon footprint for the manufacturing facility. The reduction in solvent usage and the avoidance of toxic heavy metals in the primary condensation step align with modern green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations. Waste streams are simpler to treat due to the absence of complex biological residues found in extraction processes. This environmental compatibility not only reduces disposal costs but also enhances the corporate sustainability profile of the supply chain, a factor that is becoming increasingly important for downstream customers in the pharmaceutical and food industries.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this synthesis technology, we have compiled a set of frequently asked questions based on the patent data. These inquiries address common concerns regarding the stability, purity, and applicability of the synthetic derivatives compared to their natural analogues. Understanding these distinctions is crucial for making informed decisions about integrating these materials into your product development pipeline. The answers provided below are derived directly from the experimental data and technical specifications outlined in the intellectual property documentation.
Q: Why is synthetic gingerol preferred over natural extraction?
A: Natural gingerol is chemically unstable, prone to decomposition upon heating or light exposure, and exists in low concentrations within ginger rhizomes, making isolation difficult and expensive. Synthetic derivatives offer superior stability at room temperature and normal pressure, ensuring consistent quality for industrial applications.
Q: What are the key raw materials for this synthesis?
A: The process utilizes vanillin and aliphatic ketones (such as butanone) as primary starting materials. These are commodity chemicals that are cheap, easy to obtain, and allow for a streamlined one-to-two-step reaction sequence.
Q: How is purity ensured in the final product?
A: The process employs simple yet effective purification methods including concentrated washing, drying, silica gel column chromatography, and recrystallization. This avoids tedious purification processes while achieving high purity suitable for pharmaceutical and food grade applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gingerol Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of this patented synthesis route in advancing the development of novel therapeutics and functional food ingredients. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into commercial reality. Our facilities are equipped to handle the specific requirements of this chemistry, from the precise control of acid-base solvent systems to the safe execution of catalytic hydrogenation processes. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from pilot batches to full-scale manufacturing is seamless and efficient. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify the identity and potency of every batch.
We invite you to collaborate with us to unlock the full potential of these stable gingerol derivatives for your specific applications. Whether you are developing a new anti-inflammatory drug or a high-performance nutraceutical, our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a secure, scalable, and cost-effective supply chain that empowers your innovation and accelerates your time to market.
