Scalable Electrochemical Construction of Thio(seleno)phosphoric Acid Thioester Compounds for Advanced Drug Synthesis
Introduction to Next-Generation Electrochemical Phosphorus Chemistry
The landscape of organophosphorus synthesis is undergoing a significant transformation driven by the urgent need for greener, more sustainable manufacturing processes. Patent CN114525529B introduces a groundbreaking electrochemical dehydrogenative oxidative coupling method for constructing thio(seleno)phosphoric acid thioester compounds. This technology represents a paradigm shift from traditional stoichiometric oxidation to catalytic electrochemical activation, utilizing simple starting materials such as thiophenol compounds, elemental sulfur or selenium, and diphenylphosphine. The significance of this innovation lies in its ability to access valuable P-S and P-Se bond-containing scaffolds, which are critical building blocks in the development of novel antibiotics, enzyme inhibitors, and fluorescent probes, without relying on toxic reagents. By leveraging electricity as a traceless reagent, this method aligns perfectly with modern green chemistry principles while maintaining high efficiency and operational simplicity.
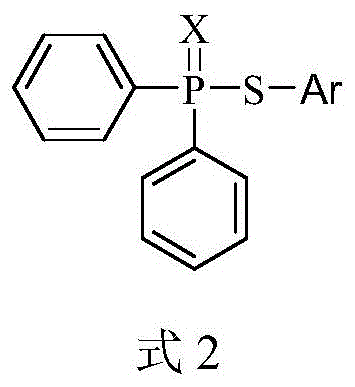
For research directors and process chemists, the structural versatility offered by this patent is particularly compelling. The general formula allows for extensive derivatization on the aromatic ring (Ar), accommodating halogens, alkyl groups, and alkoxy substituents at ortho, meta, or para positions. This flexibility ensures that the methodology is not limited to a single molecule but serves as a robust platform for generating diverse libraries of high-purity pharmaceutical intermediates. The electrochemical approach mitigates the safety risks associated with handling air-sensitive phosphorus halides, thereby streamlining the workflow in both laboratory and pilot plant settings.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of oxyphosphorothioate and related thioester compounds has been plagued by significant technical and environmental hurdles. Traditional routes often necessitate the use of hazardous chlorinating agents such as carbon tetrachloride, which poses severe toxicity risks and creates substantial waste disposal challenges. Furthermore, many established protocols require harsh reaction conditions, including elevated temperatures exceeding 100°C and strict inert atmospheres to prevent the decomposition of sensitive intermediates. The reliance on stoichiometric chemical oxidants not only increases the raw material costs but also complicates the downstream purification process due to the formation of inorganic salt byproducts. These factors collectively hinder the economic viability and environmental sustainability of large-scale production, making the search for alternative synthetic strategies a priority for forward-thinking chemical enterprises.
The Novel Approach
In stark contrast, the electrochemical method disclosed in the patent offers a streamlined, atom-economical alternative that directly addresses these pain points. By employing an undivided cell setup with a carbon rod anode and a platinum sheet cathode, the reaction proceeds smoothly at mild temperatures ranging from 25°C to 45°C. The use of elemental sulfur or selenium as the chalcogen source eliminates the need for pre-functionalized, unstable reagents. Crucially, the system utilizes potassium iodide (KI) as a catalytic redox mediator rather than a stoichiometric oxidant, significantly reducing reagent consumption. This novel approach not only simplifies the operational procedure but also enhances the overall safety profile of the synthesis, making it an attractive option for cost reduction in fine chemical manufacturing.
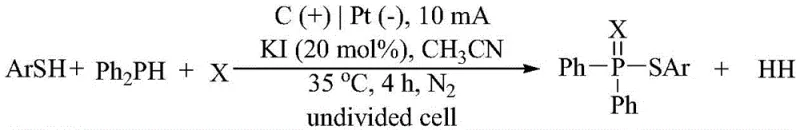
Mechanistic Insights into Electrochemical Dehydrogenative Oxidative Coupling
Understanding the underlying reaction mechanism is vital for optimizing process parameters and ensuring reproducibility at scale. The proposed catalytic cycle initiates at the anode, where iodide ions (I⁻) from the electrolyte are oxidized to generate iodine radicals (I•). These highly reactive species abstract a hydrogen atom from diphenylphosphine (HPPh₂), yielding the key diphenylphosphino radical (•PPh₂). Simultaneously, the thiophenol substrate reacts with electro-generated iodine species to form a transient S-I intermediate, which undergoes homolytic cleavage to produce thiyl radicals. The convergence of the phosphorus-centered radical and the sulfur-centered radical leads to the formation of the S-P(III) intermediate. Finally, this intermediate reacts with elemental sulfur (S₈) or selenium to furnish the final S-P(V) or Se-P(V) product, completing the constructive bond formation.
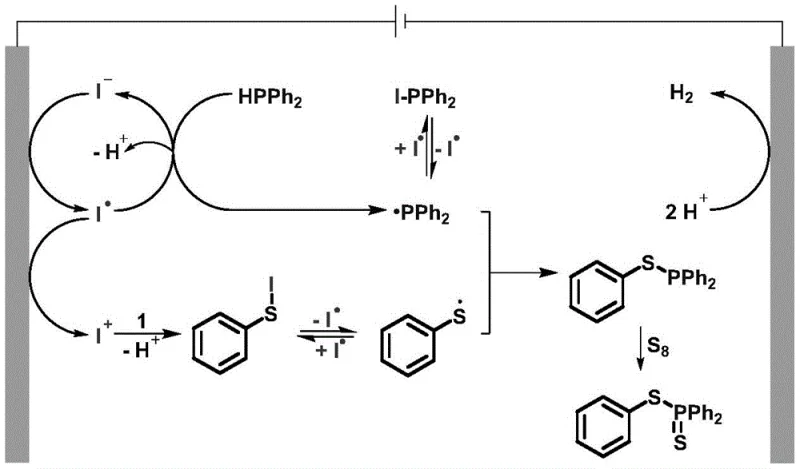
From an impurity control perspective, this radical-mediated pathway offers distinct advantages over ionic mechanisms that might suffer from side reactions like over-oxidation or polymerization. The mild potential applied (constant current of 10 mA) ensures selective activation of the iodide mediator without degrading the organic substrates. Experimental optimization data indicates that maintaining the electrolyte concentration at 20 mol% is critical; lower concentrations lead to incomplete conversion, while excess electrolyte provides no additional benefit. Furthermore, the choice of electrode material plays a pivotal role, with the carbon rod anode demonstrating superior performance compared to carbon cloth or plates, likely due to better mass transfer and surface area characteristics. This mechanistic clarity allows process engineers to fine-tune variables such as current density and temperature to maximize yield and minimize byproduct formation.
How to Synthesize Thio(seleno)phosphoric Acid Thioesters Efficiently
Implementing this electrochemical protocol requires careful attention to reaction setup and parameter control to achieve the reported high yields. The standard procedure involves charging an undivided cell with the thiophenol substrate, elemental chalcogen, diphenylphosphine, and KI in acetonitrile under a nitrogen atmosphere. A constant current electrolysis is then performed using a carbon rod anode and a platinum cathode. The simplicity of the setup belies the sophistication of the transformation, enabling the synthesis of complex molecules like 4-chloro-substituted thioesters with yields reaching up to 78% under optimized conditions. For detailed operational specifics regarding workup and purification, please refer to the standardized synthesis guide below.
- Prepare the electrolyte solution by dissolving thiophenol compound, elemental sulfur or selenium, diphenylphosphine, and KI (20 mol%) in acetonitrile under nitrogen atmosphere.
- Insert a carbon rod anode and a platinum sheet cathode into the undivided cell containing the reaction mixture.
- Apply a constant direct current of 10 mA at 35°C for 4 hours, then purify the crude product via flash chromatography to obtain the target thioester.
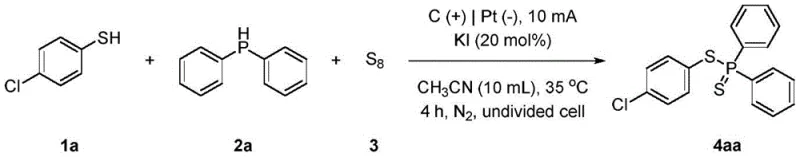
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this electrochemical methodology presents tangible opportunities for optimizing the supply base and reducing total cost of ownership. The elimination of expensive and hazardous chemical oxidants directly translates to lower raw material expenditures and reduced costs associated with hazardous waste treatment. Moreover, the use of commodity chemicals like potassium iodide and elemental sulfur ensures a stable and resilient supply chain, mitigating risks associated with the sourcing of specialized reagents. The mild reaction conditions also imply lower energy consumption for heating and cooling, contributing to a smaller carbon footprint and aligning with corporate sustainability goals.
- Cost Reduction in Manufacturing: The replacement of stoichiometric oxidants with electricity and catalytic amounts of KI drastically reduces the material cost per kilogram of product. Additionally, the simplified workup procedure, which avoids complex quenching steps for strong oxidants, lowers labor and processing time, leading to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: By relying on widely available starting materials such as diphenylphosphine and simple thiophenols, manufacturers can diversify their supplier base and avoid bottlenecks caused by single-source specialty chemicals. The robustness of the reaction across various substrates ensures consistent production output, reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The undivided cell configuration is inherently easier to scale than complex divided cells, facilitating the commercial scale-up of complex organophosphorus compounds. The absence of toxic solvents like carbon tetrachloride and the generation of hydrogen gas as the only byproduct at the cathode simplify environmental compliance and waste management protocols.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this electrochemical synthesis technology. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation, providing clarity on reaction conditions and substrate compatibility.
Q: What are the advantages of this electrochemical method over traditional chemical oxidation?
A: This method eliminates the need for hazardous chemical oxidants like carbon tetrachloride and avoids air-sensitive phosphorus halides, operating under mild conditions (35°C) with high functional group tolerance.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the use of an undivided cell, inexpensive electrode materials (carbon rod/Pt sheet), and simple electrolyte salts like KI makes the process highly amenable to scale-up for commercial manufacturing.
Q: What is the role of Potassium Iodide (KI) in this reaction mechanism?
A: KI acts as a redox mediator where iodide ions are anodically oxidized to iodine radicals, which subsequently generate phosphorus and sulfur radicals necessary for the cross-coupling reaction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thio(seleno)phosphoric Acid Thioester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in the production of advanced pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of thio(seleno)phosphoric acid thioester delivered meets the highest quality standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your supply chain and reduce your manufacturing costs.
