Advanced Microwave-Assisted Synthesis of 2-Amino-5-Bromopyridine for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways for producing critical heterocyclic intermediates, and the technology disclosed in patent CN111187206A represents a significant leap forward in this domain. This patent details a novel method for synthesizing 2-amino-5-bromopyridine, a vital building block for drugs like zopiclone and various kinase inhibitors, by leveraging the synergistic effects of microwave radiation and Lewis acidic ionic liquids. Traditional synthetic routes have long been plagued by either prohibitive reagent costs or cumbersome multi-step procedures involving protection groups, but this innovation offers a streamlined, green chemistry alternative. By utilizing 2-aminopyridine as a starting material and employing a recyclable ionic liquid catalyst under microwave conditions, the process achieves exceptional molar yields exceeding 90% with GC purity levels reaching up to 99.6%. For R&D directors and procurement strategists, this technology signals a shift towards more sustainable and cost-effective manufacturing paradigms that do not compromise on the stringent quality standards required for active pharmaceutical ingredient (API) precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2-amino-5-bromopyridine has relied on two primary methodologies, both of which present significant operational and economic drawbacks for large-scale manufacturers. The first common route utilizes N-bromosuccinimide (NBS) as the brominating agent; while this method offers good selectivity and yields often surpassing 90%, the high market price of NBS makes it economically unviable for cost-sensitive bulk production. The second conventional approach employs liquid bromine directly but necessitates a complex sequence involving amino protection with acetic anhydride, followed by bromination and subsequent hydrolysis with strong bases. This multi-step protection-deprotection strategy not only depresses the overall reaction yield to approximately 65% but also generates substantial volumes of hazardous wastewater, creating severe environmental compliance burdens and increasing waste treatment costs for supply chain managers.
The Novel Approach
In stark contrast to these legacy processes, the method described in patent CN111187206A introduces a direct halogenation strategy that eliminates the need for amino protection entirely. By dissolving 2-aminopyridine in common organic solvents like acetone or acetonitrile and introducing a Lewis acidic ionic liquid catalyst, the system enables highly selective bromination using elemental bromine under microwave irradiation. This approach capitalizes on the unique ability of ionic liquids to absorb microwave energy efficiently, resulting in rapid internal heating and uniform reaction conditions that drive conversion rates significantly higher than conventional conductive heating. The result is a simplified production workflow that reduces unit operations, minimizes solvent usage, and delivers a high-purity product through a straightforward recrystallization step, effectively resolving the cost and complexity issues inherent in prior art.
Mechanistic Insights into Lewis Acid-Ionic Liquid Catalyzed Bromination
The core of this technological breakthrough lies in the sophisticated interplay between the Lewis acidic ionic liquid catalyst and the microwave energy field. The ionic liquid, typically composed of species such as N-butylpyridinium chloride-aluminum trichloride, serves a dual function: it acts as a potent Lewis acid that polarizes the bromine molecule, thereby enhancing its electrophilicity towards the pyridine ring, and simultaneously functions as a microwave absorber due to its high dielectric loss. This dual action ensures that the activation energy for the bromination is lowered while the thermal energy is delivered directly to the molecular level, avoiding the thermal gradients often seen in oil-bath heated reactors. Consequently, the reaction proceeds with remarkable speed and specificity, favoring the formation of the 5-bromo isomer while suppressing the formation of poly-brominated byproducts or other regioisomers that typically complicate purification efforts.
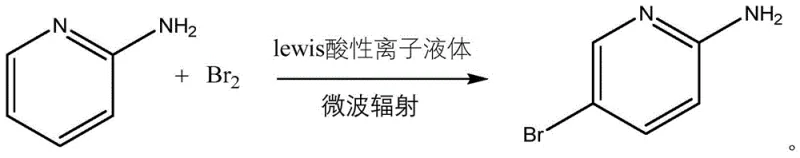
Furthermore, the mechanistic pathway facilitates superior impurity control, which is a critical parameter for R&D directors overseeing API intermediate quality. Because the reaction conditions are mild and the catalyst promotes high regioselectivity, the crude reaction mixture contains fewer side products compared to the harsh acidic or basic conditions of traditional methods. The subsequent workup involves simple reduced-pressure distillation to remove the solvent, followed by recrystallization from alcohols like methanol or ethanol, which effectively removes any residual catalyst or trace impurities. This robustness in impurity profiling ensures that the final 2-amino-5-bromopyridine meets the rigorous specifications required for downstream coupling reactions in drug synthesis, reducing the risk of batch failures and ensuring consistent supply chain reliability for pharmaceutical partners.
How to Synthesize 2-Amino-5-Bromopyridine Efficiently
To implement this advanced synthesis route in a laboratory or pilot plant setting, operators must adhere to precise parameters regarding microwave power and catalyst loading to maximize yield and safety. The process begins by dissolving the 2-aminopyridine substrate in a suitable polar aprotic solvent, followed by the careful addition of the Lewis acidic ionic liquid catalyst at a weight ratio of 5-20% relative to the substrate. Once the bromine is introduced, the mixture is subjected to controlled microwave radiation, typically between 50W and 160W, for a duration of 30 to 120 minutes, depending on the specific scale and equipment used.
- Dissolve 2-aminopyridine in an organic solvent such as acetone or acetonitrile within a reaction vessel.
- Add a Lewis acidic ionic liquid catalyst (e.g., N-butylpyridinium chloride-aluminum trichloride) to the solution.
- Introduce bromine and subject the mixture to microwave radiation (50-160W) for 30-120 minutes to effect halogenation.
- Remove the solvent via reduced pressure distillation and recrystallize the residue to obtain high-purity 2-amino-5-bromopyridine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this microwave-assisted ionic liquid technology translates into tangible strategic advantages that extend beyond simple yield improvements. The elimination of expensive brominating agents like NBS and the removal of complex protection-deprotection steps fundamentally alter the cost structure of manufacturing this key intermediate. Additionally, the ability to recycle the Lewis acidic ionic liquid catalyst further drives down variable costs, while the simplified workflow reduces the demand for labor and reactor time, allowing facilities to increase throughput without significant capital expenditure on new infrastructure.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the substitution of high-cost N-bromosuccinimide with elemental bromine, which is significantly cheaper and more readily available on the global market. Moreover, the recyclability of the ionic liquid catalyst means that the effective cost per kilogram of catalyst consumed is drastically reduced over multiple batches, leading to substantial long-term savings in raw material expenditures. By streamlining the process to a single reaction step without protection groups, the consumption of auxiliary reagents like acetic anhydride and strong bases is eliminated, further lowering the total cost of goods sold and improving profit margins for high-volume production runs.
- Enhanced Supply Chain Reliability: Simplifying the synthesis from a multi-step sequence to a direct one-pot reaction inherently reduces the risk of supply chain disruptions associated with sourcing multiple specialized reagents. The reliance on commodity chemicals like bromine and common solvents such as acetone or acetonitrile ensures that raw material availability remains stable even during market fluctuations. Furthermore, the reduced reaction time afforded by microwave heating allows for faster batch turnover, enabling manufacturers to respond more agilely to sudden spikes in demand from downstream pharmaceutical clients without compromising on delivery schedules.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method offers a greener profile by significantly reducing the generation of wastewater and chemical waste associated with hydrolysis and neutralization steps in traditional routes. The absence of heavy metal catalysts and the use of recyclable ionic liquids align with increasingly stringent global environmental regulations, minimizing the liability and cost associated with waste disposal. As the industry moves towards continuous flow microwave reactors, this chemistry is well-positioned for seamless scale-up from kilogram to tonne scales, ensuring a continuous and compliant supply of high-purity intermediates for the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this microwave-assisted synthesis technology, derived directly from the experimental data and beneficial effects outlined in the patent documentation. These insights are intended to clarify the operational feasibility and economic rationale for adopting this novel catalytic system in industrial settings.
Q: What are the primary advantages of using Lewis acidic ionic liquids over NBS for this bromination?
A: Using Lewis acidic ionic liquids significantly reduces raw material costs compared to the expensive N-bromosuccinimide (NBS). Furthermore, the ionic liquid acts as a polarizing agent for bromine, enhancing reaction selectivity and allowing for catalyst recycling, which lowers overall production expenses.
Q: How does microwave irradiation improve the reaction efficiency compared to conventional heating?
A: Microwave irradiation provides internal heating, ensuring uniform temperature distribution throughout the reaction system. This rapid energy transfer, combined with the high polarizability of the ionic liquid, drastically shortens reaction times and improves the conversion rate of raw materials compared to traditional conductive heating methods.
Q: Does this method require amino protection and deprotection steps?
A: No, unlike traditional liquid bromine methods that require complex amino protection and subsequent hydrolysis, this novel approach allows for direct bromination. This elimination of protection/deprotection steps simplifies the workflow, reduces wastewater generation, and increases the overall molar yield to over 90%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-5-Bromopyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the microwave-assisted ionic liquid synthesis method described in patent CN111187206A for producing high-quality 2-amino-5-bromopyridine. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry are fully realized in large-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced processing capabilities to maintain stringent purity specifications, guaranteeing that every batch of intermediate meets the exacting standards required for global pharmaceutical applications.
We invite procurement leaders and R&D teams to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments. Let us help you optimize your production costs and secure a reliable supply of this critical pharmaceutical intermediate through our advanced manufacturing capabilities.
