Advanced Synthesis of Dual-Function Fluorescent Probe L for Commercial Scale-Up
The chemical industry is witnessing a significant shift towards efficient, dual-function sensing materials, as evidenced by the groundbreaking technology disclosed in patent CN112480080B. This patent introduces a novel fluorescent probe, designated as Probe L, capable of the simultaneous visual detection of silver ions (Ag+) and 2,4,6-trinitrophenol (TNP), a potent nitroaromatic explosive. The innovation lies not only in its dual-application capability but also in its remarkably streamlined synthesis, which overcomes the traditional bottlenecks of multi-step organic synthesis. For R&D directors and procurement specialists alike, this represents a pivotal opportunity to integrate high-performance sensing reagents into environmental monitoring workflows with unprecedented efficiency. The technology promises to redefine standards for detecting hazardous pollutants in water and soil, offering a robust solution for regulatory compliance and safety assurance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of small-molecule fluorescent sensors for environmental pollutants has been plagued by synthetic complexity and economic inefficiency. Traditional routes often involve lengthy multi-step sequences that require rigorous purification between stages, leading to cumulative yield losses and substantial waste generation. Furthermore, many existing probes suffer from poor selectivity, reacting indiscriminately with various metal ions or nitro-compounds, which compromises the reliability of field detection. The reliance on expensive transition metal catalysts or exotic ligands in conventional methods further inflates the cost of goods sold, making widespread deployment in resource-limited settings economically unviable. These factors collectively hinder the commercial scalability of high-sensitivity sensors, creating a gap between laboratory research and practical industrial application.
The Novel Approach
In stark contrast, the methodology outlined in CN112480080B employs a direct, one-pot condensation strategy that drastically simplifies the manufacturing landscape. By utilizing polyphosphoric acid (PPA) as both a solvent and a dehydrating agent, the process facilitates the cyclization of quinaldic acid and 3,3'-diaminobenzidine in a single thermal step. This approach eliminates the need for intermediate isolation and reduces the overall reaction time significantly compared to stepwise protocols. The result is a brown solid product that can be purified through simple recrystallization, achieving yields consistently above 80 percent. This leap in process efficiency translates directly to lower production costs and a reduced environmental footprint, addressing the critical pain points of modern chemical manufacturing.
Mechanistic Insights into PPA-Mediated Cyclization and Sensing
The core of this technological breakthrough is the efficient formation of the benzimidazole-quinoline conjugated system, which serves as the fluorophore backbone. The reaction mechanism involves the nucleophilic attack of the amine groups of 3,3'-diaminobenzidine on the carboxylic acid moiety of quinaldic acid, promoted by the strong acidity and dehydrating power of polyphosphoric acid at temperatures between 155°C and 165°C. This harsh yet effective environment drives the equilibrium towards the formation of the heterocyclic ring, creating an extended pi-conjugated system essential for fluorescence. The structural integrity of Probe L is confirmed by its distinct NMR signatures and its ability to maintain stability under ambient storage conditions, ensuring a long shelf life for commercial distribution.
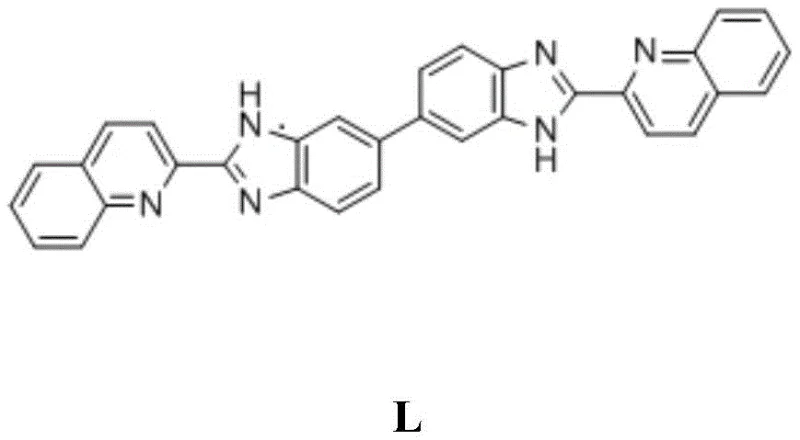
Once synthesized, the sensing mechanism operates through distinct pathways for the two target analytes, ensuring high specificity. For 2,4,6-trinitrophenol (TNP), the detection is driven by a photo-induced electron transfer (PET) process coupled with hydrogen bonding interactions between the probe's N-H groups and the hydroxyl group of TNP. This interaction effectively quenches the fluorescence, resulting in a visible color change from light blue to dark green under UV light. Conversely, the detection of silver ions relies on the strong chelating ability of the nitrogen atoms within the quinoline and imidazole rings. The coordination of Ag+ alters the electronic distribution of the fluorophore, leading to fluorescence quenching without interference from other common metal ions. This dual-mode selectivity is critical for accurate environmental analysis in complex matrices.
How to Synthesize Fluorescent Probe L Efficiently
The synthesis protocol described in the patent offers a reproducible pathway for generating high-purity Probe L suitable for analytical applications. The process begins with the precise weighing of quinaldic acid and 3,3'-diaminobenzidine, maintaining a molar ratio of approximately 2.2 to 1 to ensure complete consumption of the diamine. These precursors are suspended in polyphosphoric acid, which acts as the reaction medium, and heated under stirring to facilitate the condensation. Following the reaction period, the mixture is poured into ice water and neutralized carefully to precipitate the crude product.
- Mix quinaldic acid and 3,3'-diaminobenzidine with polyphosphoric acid in a molar ratio of approximately 2.2: 1.
- Heat the mixture to 155-165°C and stir continuously for 48 to 50 hours to facilitate cyclization.
- Cool to room temperature, neutralize with sodium hydroxide to pH 9-10, and recrystallize from ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis route offers tangible strategic benefits beyond mere technical performance. The reliance on commodity chemicals like quinaldic acid and diaminobenzidine ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or controlled precursors. The elimination of transition metal catalysts removes the need for costly and time-consuming heavy metal scavenging steps during downstream processing, which significantly lowers operational expenditures. Furthermore, the high yield and simple workup procedure reduce the volume of organic solvents required for purification, aligning with increasingly stringent environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The one-step nature of the synthesis drastically reduces labor hours and energy consumption compared to multi-step alternatives. By avoiding expensive catalysts and minimizing solvent usage for recrystallization, the overall cost of production is significantly optimized. This efficiency allows for competitive pricing strategies while maintaining healthy margins, making the probe accessible for large-scale environmental monitoring programs.
- Enhanced Supply Chain Reliability: The raw materials utilized in this process are widely available bulk chemicals with established global supply networks. This accessibility ensures consistent production schedules and minimizes the risk of delays caused by raw material shortages. Additionally, the robustness of the reaction conditions allows for flexible manufacturing planning, as the process is tolerant to minor variations in input quality without compromising the final product specifications.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to kilogram or ton-scale commercial production without fundamental changes to the reaction engineering. The use of polyphosphoric acid, while requiring careful handling, simplifies waste treatment compared to processes generating heavy metal sludge. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorescent probe technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the product's capabilities and limitations. Understanding these details is crucial for integrating the probe into existing quality control or environmental safety frameworks.
Q: What are the detection limits of Fluorescent Probe L?
A: According to patent CN112480080B, the probe exhibits high sensitivity with detection limits of 1.36 μM for silver ions and 0.82 μM for 2,4,6-trinitrophenol.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the one-step reaction utilizes readily available raw materials and achieves yields exceeding 80%, making it highly suitable for commercial scale-up without complex purification steps.
Q: How does the probe distinguish between different analytes?
A: The probe relies on distinct mechanisms: electron transfer and hydrogen bonding for TNP detection, and nitrogen-based chelation for silver ion recognition, ensuring high selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorescent Probe L Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-performance sensing materials in modern environmental protection and industrial safety. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to product is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Fluorescent Probe L meets the highest standards of sensitivity and selectivity required for professional applications.
We invite you to collaborate with us to leverage this advanced technology for your specific needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and supply chain objectives.
