Advanced Chiral Synthesis of Posaconazole Intermediates for Commercial Scale Manufacturing
The pharmaceutical industry continuously seeks robust pathways for antifungal agents, particularly for second-generation triazoles like posaconazole. A pivotal breakthrough in this domain is detailed in patent CN113666915B, which discloses a highly efficient preparation method for a critical posaconazole intermediate, specifically the compound of Formula I. This patent addresses long-standing challenges in chiral synthesis by introducing a novel catalytic system that transforms cheap, achiral starting materials into high-value chiral intermediates. For R&D directors and procurement strategists, this technology represents a significant shift away from legacy processes plagued by low yields and hazardous reagents. The disclosed method achieves a total yield exceeding 38% with chemical purity reaching 99% and optical purity greater than 99% ee, establishing a new benchmark for reliable pharmaceutical intermediate supplier capabilities in the antifungal sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Formula I has been hindered by convoluted reaction sequences and prohibitive costs. Prior art methods often relied on Wittig reactions to construct terminal double bonds, a process notorious for generating difficult-to-remove triphenylphosphine oxide waste and requiring strict anhydrous conditions. Furthermore, existing routes frequently utilized expensive stoichiometric chiral substances to establish stereocenters, alongside unstable and hazardous reagents such as sodium hydride (NaH), tert-butyl hydroperoxide, and titanium tetrachloride. These conventional pathways typically spanned 8 to 10 steps with dismal total yields ranging from merely 6.7% to 21.6%. Such inefficiencies not only inflated manufacturing costs but also introduced significant safety risks and environmental burdens, making large-scale cost reduction in pharmaceutical intermediates manufacturing nearly impossible under traditional protocols.
The Novel Approach
In stark contrast, the methodology outlined in CN113666915B streamlines the synthesis into just five robust steps, initiating from the inexpensive and readily available achiral compound of Formula II. A strategic innovation in this route is the sequence of functionalization: introducing the triazole moiety prior to epoxidation. This specific ordering minimizes steric hindrance during the critical substitution phase, allowing the reaction to proceed smoothly under milder basic conditions compared to the harsh environments required by older methods. The process eliminates the need for unstable strong bases during the triazole introduction and avoids the complex purification associated with phosphine oxide byproducts. By simplifying the synthetic tree and utilizing air or oxygen as the oxidant, this approach drastically enhances operational safety and throughput potential.
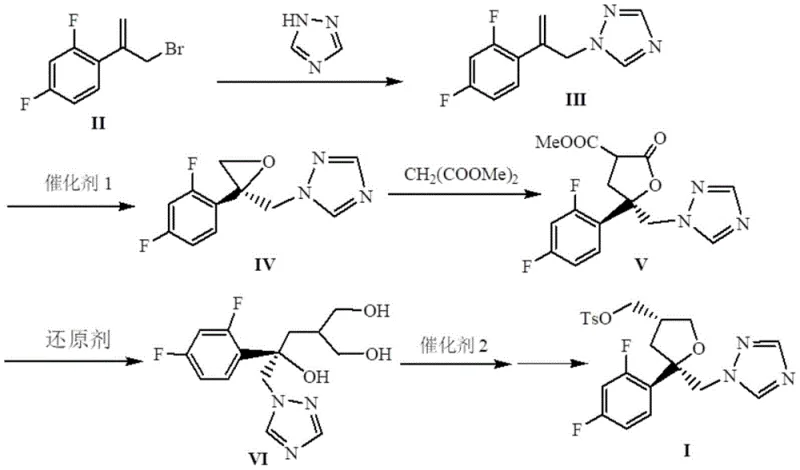
Mechanistic Insights into FeCl3-Catalyzed Asymmetric Epoxidation
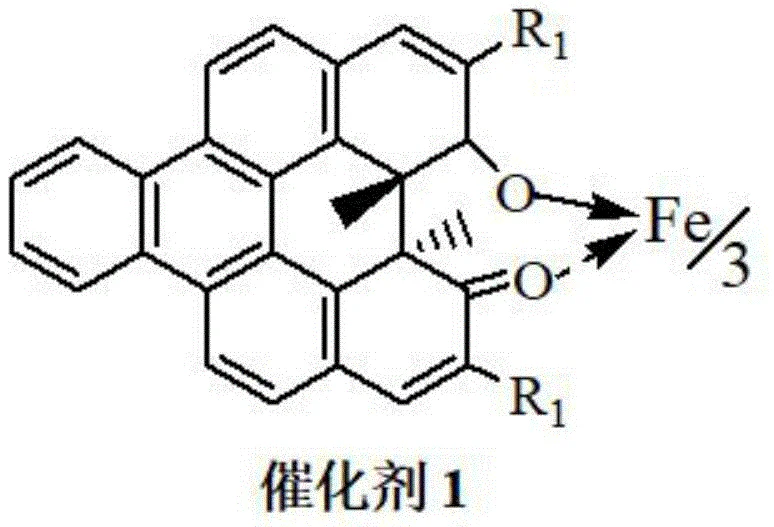
The cornerstone of this technological advancement is the utilization of a Chiral Helicenol Ketone Iron Complex, designated as Catalyst 1. This sophisticated catalyst facilitates the asymmetric epoxidation of the alkene intermediate (Formula III) using molecular oxygen or air, a reaction that traditionally requires expensive peracids or metal-oxo species. The iron center, coordinated within a rigid helical ligand framework, creates a highly defined chiral environment that directs the oxygen transfer with exceptional stereocontrol. This mechanism is crucial for establishing the two chiral centers required for the biological activity of the final drug. The patent data indicates that optimizing the substituents (R1) on the catalyst ligand—such as using isopropyl or methyl groups—can fine-tune the enantioselectivity, achieving ee values as high as 94.3% in the epoxidation step alone, which is subsequently upgraded to >99% in later stages.
Furthermore, the process incorporates a solid superacid catalyst, SO42-/ZrO2, for the intramolecular etherification step. This heterogeneous catalysis offers distinct advantages over homogeneous liquid acids, primarily in terms of separation and reusability. The solid acid promotes the cyclization to form the tetrahydrofuran ring efficiently at temperatures between 60°C and 110°C. From an impurity control perspective, the high selectivity of the iron-catalyzed epoxidation minimizes the formation of diastereomers early in the synthesis. This reduces the burden on downstream purification units, ensuring that the final high-purity pharmaceutical intermediate meets stringent regulatory specifications without requiring extensive chromatographic separations that would otherwise erode profit margins.
How to Synthesize Posaconazole Intermediate Efficiently
The synthesis protocol described in the patent provides a clear roadmap for transitioning from laboratory scale to commercial production. The process begins with the nucleophilic substitution of Formula II with 1,2,4-triazole, followed by the pivotal iron-catalyzed epoxidation. Subsequent steps involve condensation with dimethyl malonate, reduction using sodium borohydride and halogens, and final cyclization. The detailed operational parameters, including specific temperature ramps and solvent systems like THF/methanol mixtures, are critical for maintaining the high stereochemical integrity of the product. For process chemists looking to implement this technology, understanding the precise stoichiometry of the reducing agents and the activation of the solid superacid is essential for replicating the reported yields.
- React Formula II with 1,2,4-triazole under basic conditions to form Formula III.
- Perform asymmetric epoxidation on Formula III using Chiral Helicenol Ketone Iron Complex (Catalyst 1) and air/oxygen to yield Formula IV.
- Condense Formula IV with dimethyl malonate, followed by reduction and intramolecular etherification to finalize Formula I.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers transformative benefits regarding cost structure and supply continuity. The shift from multi-step, low-yield processes to a concise 5-step sequence fundamentally alters the cost of goods sold (COGS). By eliminating the need for expensive chiral auxiliaries and hazardous oxidants, the raw material bill is significantly reduced. Moreover, the use of air or oxygen as a terminal oxidant represents a drastic cost saving compared to purchasing stoichiometric oxidizing agents, while simultaneously reducing the chemical waste load that requires costly disposal. This aligns perfectly with modern green chemistry initiatives, potentially lowering regulatory compliance costs associated with hazardous waste management.
- Cost Reduction in Manufacturing: The elimination of expensive reagents like titanium tetrachloride and stoichiometric chiral substances directly lowers variable production costs. Additionally, the ability to recycle the solid superacid catalyst in the final step further drives down operational expenses. The higher overall yield means less starting material is required per kilogram of finished intermediate, maximizing the utility of every dollar spent on raw materials and significantly improving the economic efficiency of the manufacturing campaign.
- Enhanced Supply Chain Reliability: Relying on cheap, achiral starting materials like Formula II mitigates the risk of supply bottlenecks often associated with specialized chiral building blocks. The robustness of the reaction conditions, which tolerate mild temperatures and standard solvents, ensures that production is less susceptible to disruptions caused by sensitive reagent degradation or strict storage requirements. This stability translates to more predictable lead times and a more resilient supply chain capable of meeting fluctuating market demands for antifungal medications.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, avoiding unit operations that are difficult to enlarge, such as cryogenic reactions or handling of pyrophoric materials. The use of molecular oxygen and recyclable heterogeneous catalysts reduces the environmental footprint, facilitating easier permitting and compliance with increasingly strict environmental regulations. This scalability ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved rapidly without compromising on safety or quality standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical application of this technology for industrial partners.
Q: What is the key advantage of the new catalytic system in CN113666915B?
A: The use of a Chiral Helicenol Ketone Iron Complex allows for asymmetric epoxidation using air or oxygen, replacing expensive stoichiometric chiral auxiliaries and hazardous oxidants, resulting in yields over 38% and optical purity exceeding 99% ee.
Q: How does this process improve supply chain stability compared to traditional Wittig routes?
A: Traditional routes rely on unstable reagents like NaH and complex Wittig reactions with low yields (6-21%). This new method uses cheap, achiral starting materials and mild conditions, significantly reducing raw material volatility and processing risks.
Q: Is the catalyst reusable in this manufacturing process?
A: Yes, the solid superacid catalyst (SO42-/ZrO2) used in the final etherification step can be filtered, dried, and activated for reuse, further enhancing the economic viability and waste reduction profile of the process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Posaconazole Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient chiral synthesis in the modern pharmaceutical landscape. Our technical team has extensively analyzed the pathway described in CN113666915B and possesses the expertise to adapt and optimize this route for your specific production needs. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the high optical purity required for posaconazole intermediates, guaranteeing that every batch meets global regulatory standards.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your project's success and reduce your time to market.
