Advanced Microreactor Bromination Strategy for High-Purity Kinase Inhibitor Intermediates
Advanced Microreactor Bromination Strategy for High-Purity Kinase Inhibitor Intermediates
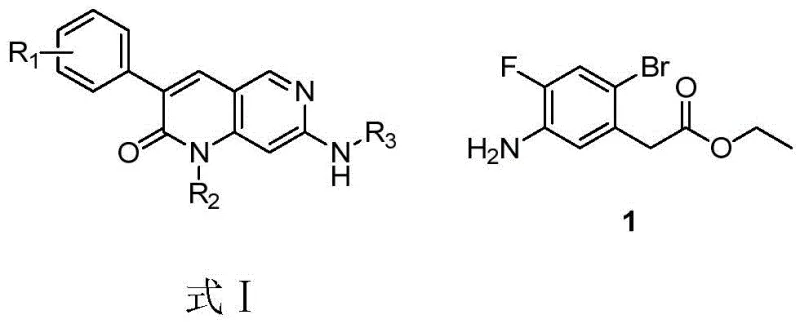
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for complex kinase inhibitor intermediates, particularly those serving as core scaffolds for treating serious conditions like gastrointestinal stromal tumors and acute myeloid leukemia. Patent CN110845337B introduces a groundbreaking methodology for synthesizing ethyl 2-bromo-4-fluoro-5-aminophenylacetate, a critical building block known as Compound 1, which is essential for constructing dihydronaphthyridine derivatives. This innovation shifts the paradigm from hazardous batch nitration processes to a sophisticated continuous flow bromination strategy, leveraging microreactor technology to achieve unprecedented control over reaction selectivity and thermal management. By reordering the synthetic sequence to introduce the halogen substituent at a later stage via bromination rather than early-stage nitration, the process effectively mitigates the severe safety risks associated with exothermic nitration reactions while simultaneously enhancing the purity profile of the final product. For R&D directors and procurement specialists alike, this patent represents a significant leap forward in process chemistry, offering a pathway that is not only chemically elegant but also commercially viable for multi-ton manufacturing campaigns.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Compound 1 has relied on a route starting with 2-bromo-4-fluorophenylacetic acid, necessitating a nitration step to introduce the nitrogen functionality required for subsequent cyclization. This traditional approach, as documented in prior art such as WO2013184119, suffers from inherent and severe drawbacks that make it unsuitable for modern, large-scale industrial applications. The nitration reaction is notoriously violent and highly exothermic, posing significant safety hazards regarding thermal runaway and pressure buildup within standard reactor vessels. Furthermore, the regioselectivity of electrophilic aromatic substitution during nitration is often poor, leading to a complex mixture of isomers that are structurally similar and extremely difficult to separate via standard crystallization or chromatography techniques. These purification challenges result in substantial yield losses and increased waste generation, driving up the cost of goods sold and complicating the supply chain for high-purity active pharmaceutical ingredients. Consequently, the reliance on this legacy route creates a bottleneck for manufacturers aiming to secure a reliable supply of this key intermediate for clinical and commercial drug production.

The Novel Approach
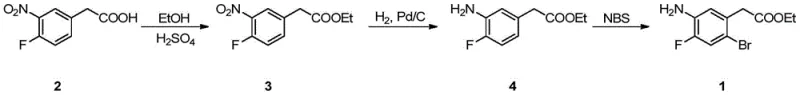
In stark contrast to the perilous nitration-first strategy, the novel process disclosed in CN110845337B ingeniously reverses the synthetic logic by starting with 3-nitro-4-fluorophenylacetic acid and deferring the halogenation to the final step. This strategic pivot allows the use of mild esterification and catalytic hydrogenation conditions to establish the core carbon-nitrogen framework before introducing the sensitive bromine atom. The crown jewel of this new methodology is the implementation of microreactor technology for the critical bromination step, where N-bromosuccinimide (NBS) is used as the brominating agent under precisely controlled flow conditions. This continuous flow setup enables the reaction to proceed at low temperatures with rapid mixing and efficient heat dissipation, effectively suppressing the formation of unwanted polybrominated byproducts that plague batch processes. The result is a streamlined, safer, and highly selective synthesis that transforms a hazardous multi-step sequence into a manageable and scalable operation, directly addressing the pain points of both process safety and product quality assurance.
Mechanistic Insights into Microreactor-Assisted Bromination
The success of this synthetic route hinges on the unique physicochemical environment created within the microreactor channels during the bromination of ethyl 3-amino-4-fluorophenylacetate. Unlike traditional stirred tank reactors where mixing is limited by macroscopic diffusion and heat transfer is constrained by vessel surface area, microreactors utilize channels with diameters ranging from 10 μm to 3.0 mm to create an immense specific surface area for the reacting fluids. This geometric advantage facilitates near-instantaneous heat exchange, allowing the reaction mixture to be maintained strictly within the optimal temperature window of 0 to 5°C, or even lower if necessary for specific selectivity profiles. Such precise thermal control is paramount because the bromination of electron-rich aniline derivatives is highly susceptible to over-reaction; without immediate heat removal, the local hot spots typical in batch reactors would accelerate the kinetics of secondary bromination, leading to the formation of difficult-to-remove dibromo impurities. By ensuring that every molecule of substrate experiences the exact same residence time and temperature history, the microreactor enforces a uniform reaction trajectory that maximizes the yield of the desired mono-brominated product while kinetically inhibiting side reactions.
Beyond thermal management, the mechanism of impurity control in this process is deeply rooted in the hydrodynamics of laminar flow and rapid mixing inherent to microfluidic systems. In the described protocol, solutions of the amine substrate and the NBS reagent are pumped separately through pre-cooling modules before meeting in a mixing module, ensuring that the concentration of the aggressive brominating species is instantly diluted and consumed upon contact with the substrate. This prevents the local accumulation of excess bromine that typically drives polybromination in batch additions where reagent gradients exist. Furthermore, the short residence time, tunable between 30 to 70 seconds, ensures that the product is ejected from the reaction zone immediately after formation, quenching its reactivity before it can undergo further degradation or substitution. This kinetic quenching effect, combined with the ability to fine-tune the molar ratio of reagents (optimized at 1:1.2), creates a robust defense against impurity generation, yielding a crude product stream that is exceptionally clean and requires minimal downstream processing to meet stringent pharmaceutical specifications.
How to Synthesize Ethyl 2-Bromo-4-Fluoro-5-Aminophenylacetate Efficiently
The execution of this synthesis requires careful attention to the sequential transformation of the starting material, beginning with the acid-catalyzed esterification of 3-nitro-4-fluorophenylacetic acid in absolute ethanol at reflux temperatures around 80°C. Following the removal of solvent and aqueous workup, the resulting nitro-ester undergoes catalytic hydrogenation using palladium on carbon under mild pressure and temperature conditions to afford the corresponding aniline derivative with high fidelity. The final and most critical transformation involves the continuous flow bromination, where the precise coordination of pump flow rates, cooling capacities, and mixing geometries determines the ultimate success of the campaign in terms of yield and purity. While the general principles are straightforward, the specific operational parameters regarding flow rates and residence times are vital for replicating the high selectivity reported in the patent data. For a detailed breakdown of the standardized operating procedures and specific equipment configurations required to implement this chemistry, please refer to the technical guide below.
- Perform esterification of 3-nitro-4-fluorophenylacetic acid with absolute ethanol and sulfuric acid at 80°C to form the ethyl ester.
- Conduct catalytic hydrogenation using Pd/C at 40°C to reduce the nitro group to an amino group, yielding ethyl 3-amino-4-fluorophenylacetate.
- Execute the critical bromination step in a microreactor using NBS in DMF at controlled low temperatures (0-5°C) to ensure high mono-bromination selectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this microreactor-based synthesis offers profound strategic benefits that extend far beyond simple chemical yield improvements. By eliminating the need for hazardous nitration infrastructure and the associated safety protocols, manufacturing facilities can significantly reduce their operational overhead and insurance liabilities, translating into a more stable and cost-effective supply base. The inherent safety of the continuous flow process also means that production can be ramped up with greater confidence, reducing the risk of unplanned shutdowns due to safety incidents or regulatory scrutiny regarding hazardous chemical handling. Furthermore, the high selectivity of the bromination step drastically simplifies the purification workflow, reducing the consumption of solvents and chromatography media, which are major cost drivers in the production of high-value intermediates. This efficiency gain allows for a leaner manufacturing model that is more resilient to fluctuations in raw material costs and environmental compliance pressures.
- Cost Reduction in Manufacturing: The transition to this novel route eliminates the expensive and energy-intensive purification steps required to remove nitration isomers, thereby lowering the overall cost of goods sold. By avoiding the use of harsh nitrating agents and the complex waste treatment they necessitate, the process achieves substantial cost savings through simplified effluent management and reduced raw material consumption. The high conversion rates observed in the microreactor also mean that less starting material is wasted, optimizing the atom economy of the entire synthesis and maximizing the return on investment for every kilogram of feedstock purchased.
- Enhanced Supply Chain Reliability: The robustness of the microreactor system ensures consistent product quality batch after batch, minimizing the risk of supply disruptions caused by failed production runs or out-of-specification results. Because the process operates under milder conditions and uses widely available reagents like NBS and ethanol, it is less susceptible to the supply volatility often seen with specialized nitrating mixtures or exotic catalysts. This reliability allows pharmaceutical companies to forecast their inventory needs more accurately and maintain tighter control over their production schedules, ensuring that critical drug development timelines are met without delay.
- Scalability and Environmental Compliance: The modular nature of microreactor technology allows for seamless scale-up through numbering-up strategies rather than risky vessel size increases, facilitating a smooth transition from pilot plant to commercial tonnage production. This approach aligns perfectly with green chemistry principles by reducing solvent usage and energy consumption per unit of product, helping manufacturers meet increasingly stringent environmental regulations and sustainability goals. The ability to operate continuously also reduces the facility footprint required for production, enabling higher output volumes from existing infrastructure without the need for massive capital expenditure on new reactor trains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route, drawing directly from the experimental data and process descriptions found in the patent literature. Understanding these nuances is crucial for technical teams evaluating the feasibility of adopting this technology for their own manufacturing pipelines. The answers provided reflect the specific advantages of the microreactor approach over traditional batch methods, highlighting the practical implications for process safety and product quality.
Q: Why is the microreactor preferred over batch processing for this bromination?
A: The microreactor offers superior heat exchange capabilities and precise temperature control, which are critical for suppressing polybrominated side products that are difficult to separate in traditional batch reactors.
Q: What are the safety advantages of this new synthetic route?
A: By avoiding the violent exothermic nitration reaction required in conventional methods, this route significantly reduces thermal runaway risks and improves overall operational safety for large-scale production.
Q: How does this process impact the purity profile of the final intermediate?
A: The high selectivity of the microreactor bromination minimizes the formation of dibromo impurities, resulting in a crude product with significantly higher purity and reducing the burden on downstream purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl 2-Bromo-4-Fluoro-5-Aminophenylacetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of next-generation kinase inhibitors, and we are uniquely positioned to support your projects with our advanced manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in early-stage clinical trials or full-scale commercial launch. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of ethyl 2-bromo-4-fluoro-5-aminophenylacetate meets the exacting standards required for pharmaceutical applications, giving you peace of mind regarding the integrity of your supply chain.
We invite you to engage with our technical procurement team to discuss how we can tailor this innovative microreactor process to your specific project needs and timeline. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized synthesis route can reduce your overall manufacturing expenses while improving supply security. Please contact us today to obtain specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for complex pharmaceutical intermediate manufacturing.
