Advanced Synthetic Route for Famciclovir Impurity C: Enhancing Quality Control and Supply Stability
Advanced Synthetic Route for Famciclovir Impurity C: Enhancing Quality Control and Supply Stability
The pharmaceutical industry's relentless pursuit of purity and safety has placed immense pressure on the availability of high-quality reference standards for impurity profiling. A pivotal development in this domain is documented in patent CN107488176B, which discloses a robust and scalable method for synthesizing Famciclovir Impurity C, chemically identified as 4-(2-amino-9H-purin-9-yl)-2-methylbutyl acetate (BRL45145). This specific impurity is a critical quality attribute for Famciclovir, a widely prescribed antiviral prodrug used to treat herpes simplex and herpes zoster infections. The ability to reliably produce this impurity allows manufacturers to strictly monitor its levels during the production of the active pharmaceutical ingredient (API), ensuring compliance with stringent global regulatory guidelines. The disclosed technology offers a significant breakthrough by utilizing readily available starting materials to construct the complex side chain and purine core efficiently.

Understanding the structural relationship between the parent drug and its impurities is fundamental for R&D teams focused on method validation. As illustrated in the structural comparison, Famciclovir (S-1) contains a diacetate ester side chain, whereas Impurity C (S-2) features a monoacetate structure resulting from a specific dehydroxylation side reaction. The absence of a reliable synthetic route for this impurity has historically hindered the ability of quality control laboratories to accurately quantify its presence in bulk drug substances. By addressing this gap, the patent provides a vital tool for the pharmaceutical supply chain, enabling precise quantification and ensuring that the final medicinal preparations are safe for patient use. This technological advancement underscores the importance of having dedicated synthetic pathways for even minor process-related impurities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the disclosure of this patent, the scientific literature, including reports such as Reference 1 (Bryant et al., Journal of Chromatography A, 1996), had identified the existence of Famciclovir Impurity C and its chromatographic behavior. However, a significant void existed in the available art regarding its actual synthesis. Conventional approaches to obtaining such specific impurities often involved isolating them from crude reaction mixtures of the main API synthesis, a process that is notoriously inefficient, low-yielding, and incapable of producing the quantities required for routine analytical calibration. Furthermore, attempting to synthesize such molecules using non-optimized routes often leads to complex mixtures of regioisomers, particularly when alkylating the purine ring, making purification a costly and time-consuming bottleneck. The lack of a defined, reproducible synthetic protocol meant that many manufacturers struggled to source authentic reference standards, potentially compromising the accuracy of their impurity profiling and risking regulatory non-compliance.
The Novel Approach
The methodology presented in patent CN107488176B introduces a streamlined, five-step linear synthesis that circumvents these historical challenges by starting from (+/-)-alpha-methyl-gamma-butyrolactone. This novel approach constructs the requisite carbon skeleton first, installing the bromine handle necessary for subsequent coupling, before introducing the purine base. This strategy ensures high regioselectivity during the alkylation step, favoring the desired N9-substitution over the N7-isomer. The route is designed for operational simplicity, avoiding cryogenic conditions or exotic reagents, and utilizes common solvents like methanol, dichloromethane, and DMF. By decoupling the side-chain synthesis from the purine coupling until the later stages, the process achieves high overall yields and simplifies the purification of intermediates. This logical disconnection of the molecule allows for the efficient production of not only the target impurity but also the chlorinated intermediate (BRL48951), adding further value to the process.
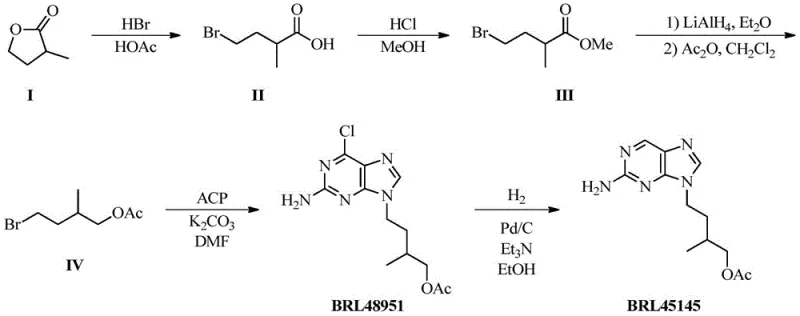
Mechanistic Insights into the Multi-Step Synthesis
The synthetic pathway relies on a series of well-understood organic transformations that have been optimized for maximum efficiency. The sequence initiates with the ring-opening of the lactone using hydrobromic acid in acetic acid, a reaction that proceeds via nucleophilic attack of the bromide ion on the protonated carbonyl, yielding the bromo-acid intermediate with excellent conversion rates. Following esterification to protect the carboxylic acid functionality, the critical reduction step employs lithium aluminum hydride (LiAlH4) to convert the ester into a primary alcohol. This reduction is immediately followed by acetylation using acetic anhydride and a catalytic amount of DMAP, which installs the acetate group while preserving the bromine handle for the subsequent nucleophilic substitution. This tandem reduction-acetylation sequence is crucial for establishing the correct oxidation state of the side chain prior to coupling.
The coupling reaction represents the heart of the synthesis, where the bromo-acetate intermediate undergoes an SN2-type alkylation with 2-amino-6-chloropurine in the presence of potassium carbonate. The use of anhydrous DMF as the solvent facilitates the dissolution of the polar purine base and enhances the nucleophilicity of the purine nitrogen. The reaction conditions are carefully controlled to favor alkylation at the N9 position of the purine ring, minimizing the formation of the N7 regioisomer which would constitute a difficult-to-remove impurity. The final step involves a catalytic hydrodechlorination using Pd/C under a hydrogen atmosphere. This step cleanly removes the chlorine atom at the C6 position of the purine ring without affecting the acetate ester or the integrity of the purine system, delivering the final target molecule, Famciclovir Impurity C, with high purity suitable for analytical applications.
How to Synthesize Famciclovir Impurity C Efficiently
The execution of this synthesis requires careful attention to reaction stoichiometry and workup procedures to maintain high purity throughout the sequence. The process begins with the ring-opening of the lactone, followed by esterification and reduction-acetylation to generate the key alkylating agent. This agent is then coupled with the purine base, and the resulting chloro-intermediate is finally reduced to the target amine. While the general chemistry is straightforward, the specific conditions regarding temperature, solvent choice, and reagent equivalents are critical for success. For laboratory personnel and process chemists looking to implement this route, adhering to the standardized parameters ensures reproducibility and safety. The detailed standardized synthesis steps are outlined in the guide below.
- Ring Opening: React (+/-)-alpha-methyl-gamma-butyrolactone with 33% HBr in acetic acid to obtain (+/-)-2-methyl-4-bromobutyric acid.
- Esterification & Reduction: Convert the acid to methyl ester using HCl/MeOH, then reduce with LiAlH4 and acetylate to form (+/-)-2-methyl-4-bromobutyl acetate.
- Coupling & Deprotection: Alkylate 2-amino-6-chloropurine with the bromo-acetate intermediate, followed by catalytic hydrogenation (Pd/C) to remove the chlorine atom.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits regarding cost stability and sourcing reliability. The primary advantage lies in the selection of starting materials; (+/-)-alpha-methyl-gamma-butyrolactone is a commodity chemical available from multiple global suppliers, eliminating the risk of single-source bottlenecks often associated with complex nucleoside precursors. This abundance of raw materials translates directly into a more resilient supply chain, as fluctuations in the price or availability of one vendor can be easily mitigated by switching to another. Furthermore, the synthetic route avoids the use of precious metal catalysts in the early stages, reserving the expensive Palladium on Carbon only for the final deprotection step, which optimizes the overall cost of goods sold (COGS) by minimizing the consumption of high-value reagents.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing a convergent strategy that maximizes the yield of each individual step. By avoiding complex protection-deprotection sequences for the side chain and using inexpensive reagents like acetyl chloride and acetic anhydride, the overall material costs are drastically reduced compared to alternative routes that might require more elaborate scaffolding. Additionally, the high conversion rates reported in the patent examples mean that less raw material is wasted, and the burden on downstream purification processes is lessened, leading to substantial savings in solvent usage and processing time.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable intermediates and common solvents ensures that the manufacturing process is robust against logistical disruptions. The synthesis does not require specialized equipment for handling hazardous gases or extreme temperatures, making it adaptable to a wide range of manufacturing facilities. This flexibility allows for rapid scale-up from kilogram to multi-ton production without the need for significant capital investment in new infrastructure, thereby guaranteeing a continuous supply of this critical reference material to meet the demands of quality control laboratories globally.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the route is designed with scalability in mind. The waste streams generated, primarily consisting of aqueous salts and common organic solvents, are manageable using standard wastewater treatment protocols employed in modern fine chemical plants. The elimination of heavy metal contaminants in the early stages simplifies the purification profile, reducing the environmental footprint associated with metal scavenging and disposal. This alignment with green chemistry principles not only facilitates regulatory approval but also positions the manufacturer as a responsible partner in the pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Famciclovir Impurity C. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on the feasibility and advantages of this specific synthetic methodology. Understanding these details is crucial for stakeholders evaluating the integration of this impurity standard into their quality assurance workflows.
Q: What is the primary starting material for synthesizing Famciclovir Impurity C?
A: The synthesis utilizes (+/-)-alpha-methyl-gamma-butyrolactone as the initial raw material, which is commercially abundant and cost-effective compared to complex nucleoside precursors.
Q: How is the chlorine atom removed in the final step of the synthesis?
A: The final step involves a catalytic hydrodechlorination reaction using Palladium on Carbon (Pd/C) under a hydrogen atmosphere in the presence of an acid-binding agent like triethylamine.
Q: Why is synthesizing Impurity C critical for Famciclovir manufacturing?
A: Impurity C (BRL45145) is a known process-related impurity formed during ester group reduction. Having a certified reference standard is essential for HPLC monitoring to ensure the final drug product meets regulatory safety limits.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Famciclovir Impurity C Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your pharmaceutical products depends on the quality of every component, including the reference standards used for testing. Our team of expert process chemists has thoroughly analyzed the synthetic route described in patent CN107488176B and is fully prepared to execute this chemistry at any scale required by our partners. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Famciclovir Impurity C we deliver meets the highest industry standards for analytical accuracy.
We invite you to collaborate with us to secure a stable and cost-effective supply of this critical intermediate. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your specific procurement needs, identifying opportunities to optimize your supply chain further. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us be your partner in maintaining the highest standards of quality and safety in your antiviral drug manufacturing processes.
