Advanced Synthesis of Benzothiazole-2-one Derivatives Using Carbonyl Sulfide and Disulfides
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and economically viable synthetic routes for heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN108101863B, which discloses a highly efficient method for synthesizing benzothiazole-2-ketone derivatives. This technology represents a paradigm shift by utilizing carbonyl sulfide (COS) and stable aromatic disulfides as primary raw materials, catalyzed by inexpensive inorganic sulfides. Unlike traditional approaches that struggle with the instability of thiol precursors, this innovation leverages the thermodynamic stability of disulfides while overcoming their kinetic inertness through a novel dynamic exchange mechanism. For R&D directors and procurement managers alike, this process offers a compelling value proposition: it simplifies the supply chain by using shelf-stable starting materials and reduces operational hazards by avoiding harsh reagents like phosgene. As a reliable pharmaceutical intermediate supplier, understanding such technological advancements is crucial for maintaining a competitive edge in the global market.
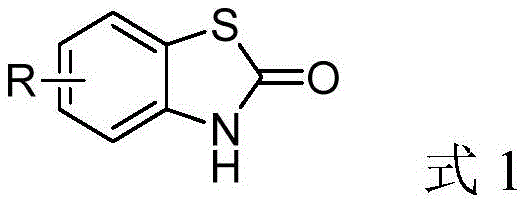
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzothiazole-2-one derivatives has relied heavily on o-aminothiophenols as the foundational building blocks. While chemically logical, o-aminothiophenols suffer from severe practical drawbacks, primarily their inherent instability. These compounds are highly susceptible to oxidative dimerization, spontaneously forming disulfides upon exposure to air, which complicates storage, transportation, and precise dosing in large-scale reactors. Furthermore, conventional carbonylation methods often employ hazardous reagents such as phosgene, triphosgene, or ethyl chloroformate. These reagents not only pose significant safety risks to personnel and facilities but also generate substantial amounts of stoichiometric waste, such as hydrochloric acid or ethanol, necessitating complex downstream neutralization and purification steps. The atom economy of these traditional routes is often poor, and the requirement for strict anhydrous conditions or high temperatures further escalates energy consumption and operational costs, making them less attractive for modern green chemistry standards.
The Novel Approach
The methodology described in the patent introduces a transformative solution by directly utilizing the oxidation product—aromatic disulfides—as the starting material, thereby turning a stability liability into a logistical asset. By employing carbonyl sulfide (COS) as the carbonyl source, the process achieves excellent atom economy, incorporating both carbon and sulfur atoms efficiently into the final heterocyclic ring. The reaction is driven by an inorganic sulfide catalyst, such as sodium hydrosulfide (NaHS), which facilitates the cleavage of the robust S-S bond under remarkably mild conditions. As illustrated in the reaction scheme below, the process operates effectively at ambient temperatures (25°C) and low pressures (0.5 MPa), eliminating the need for energy-intensive heating or high-pressure equipment. This approach not only mitigates the safety risks associated with volatile carbonylating agents but also streamlines the workflow by removing the necessity for additional dehydrating agents, as the reaction inherently proceeds with direct dehydration.
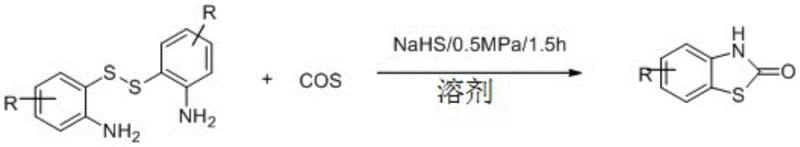
Mechanistic Insights into Inorganic Sulfide-Catalyzed Cyclization
The core of this technological advancement lies in the unique activation mechanism mediated by inorganic sulfides. Thermodynamically, the S-S bond in disulfides is stable, which prevents the spontaneous degradation seen in thiols but typically hinders reactivity. The invention exploits a metal sulfide-disulfide dynamic exchange reaction to overcome this barrier. The inorganic sulfide catalyst acts as a nucleophile or a Lewis base equivalent, attacking the disulfide bond to generate a transient, highly reactive thiolate species in situ. This activated intermediate is then perfectly positioned to react with the electrophilic carbon of the carbonyl sulfide. Unlike carbon dioxide (CO2), which possesses two strong C=O bonds and exhibits significant kinetic inertness, COS contains a C=S bond that is longer and weaker, making it more susceptible to nucleophilic attack. This structural nuance allows COS to serve as a superior carbonyl donor in this specific context, facilitating rapid cyclization.
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity pharmaceutical intermediates. Traditional methods often struggle with side reactions involving the free amine group or over-carbonylation. However, the specific coordination environment created by the inorganic sulfide and the solvent system promotes a highly selective intramolecular cyclization. The reaction proceeds through a concerted pathway where the nucleophilic attack on the COS carbon is immediately followed by ring closure and dehydration. This specificity minimizes the formation of polymeric byproducts or urea-like impurities that are common when using other carbonyl sources. Furthermore, the use of simple inorganic salts as catalysts ensures that the final product is free from toxic transition metal residues, a critical requirement for API intermediates intended for human therapeutic use. The ability to tune the molar ratio of disulfide to inorganic sulfide (preferably 1:0.5 to 1:1) provides process chemists with a robust handle to optimize conversion rates without compromising purity profiles.
How to Synthesize Benzothiazole-2-one Efficiently
The practical implementation of this synthesis is designed for ease of operation and scalability, making it highly suitable for industrial adoption. The general procedure involves charging a stainless steel high-pressure reactor with the aromatic disulfide substrate, the inorganic sulfide catalyst, and a polar aprotic solvent such as DMF or THF. The system is then pressurized with COS gas, and the mixture is stirred at room temperature for a short duration, typically 1.5 hours. Workup is straightforward, involving standard extraction and purification techniques like column chromatography or recrystallization. This simplicity stands in stark contrast to multi-step sequences required by older methodologies.
- Mix the aromatic o-amino disulfide substrate with an inorganic sulfide catalyst (such as NaHS) in an organic solvent like DMF or THF within a high-pressure reactor.
- Introduce carbonyl sulfide (COS) gas into the reactor to achieve a pressure of 0.1-1 MPa, preferably around 0.5 MPa, and stir the mixture at ambient temperature (5-50°C).
- After reacting for 1-2 hours, concentrate the reaction mixture under reduced pressure and purify the crude product via column chromatography or recrystallization to obtain the high-purity benzothiazole-2-one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic benefits beyond mere chemical elegance. The shift from unstable thiols to stable disulfides fundamentally alters the logistics of raw material management. Disulfides are solids with high melting points and excellent shelf stability, allowing for bulk purchasing and long-term storage without the degradation risks associated with liquid thiols. This stability ensures a consistent supply of quality starting materials, reducing the frequency of quality failures and batch rejections that can disrupt production schedules. Moreover, the elimination of hazardous reagents like phosgene simplifies regulatory compliance and reduces the overhead costs associated with specialized safety infrastructure and waste disposal.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of expensive and hazardous reagents with commodity chemicals. The catalyst system relies on inorganic sulfides like NaHS or K2S, which are abundant and low-cost industrial chemicals, removing the need for precious metal catalysts or complex organic ligands. Additionally, the reaction operates at ambient temperature and low pressure, drastically reducing energy consumption compared to high-temperature reflux or high-pressure hydrogenation processes. The absence of stoichiometric dehydrating agents further lowers the bill of materials, while the high atom economy of the COS fixation minimizes waste treatment costs, collectively driving down the cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the robustness of the raw materials. Since the disulfide starting materials are resistant to oxidation, they can be sourced from a wider range of suppliers without stringent packaging requirements, mitigating the risk of single-source dependency. The mild reaction conditions also mean that the process is less sensitive to minor fluctuations in utility supplies (such as steam pressure or cooling water temperature), ensuring consistent batch-to-batch reproducibility. This reliability is critical for maintaining just-in-time delivery schedules for downstream API manufacturers, preventing costly production delays.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by its inherent safety profile. The low operating pressure (0.1-1 MPa) allows for the use of standard glass-lined or stainless steel reactors without the need for exotic high-pressure vessels, lowering capital expenditure (CAPEX) for capacity expansion. Environmentally, the process aligns with green chemistry principles by utilizing COS, a potential greenhouse gas, as a feedstock, thereby contributing to carbon utilization goals. The simplified workup and lack of heavy metal contaminants reduce the burden on wastewater treatment facilities, ensuring easier compliance with increasingly stringent environmental regulations in major chemical manufacturing hubs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. They are derived from the specific experimental data and beneficial effects outlined in the patent documentation, providing clarity on the practical application of this method for industrial partners.
Q: Why is this method superior to using o-aminothiophenols?
A: Traditional methods often rely on o-aminothiophenols, which are chemically unstable and prone to oxidative dimerization during storage and handling. This novel process utilizes stable aromatic disulfides as starting materials, significantly improving raw material shelf-life and handling safety while maintaining high reaction efficiency.
Q: What are the safety advantages of using COS over phosgene?
A: While phosgene is a highly toxic and hazardous carbonyl source requiring extreme safety measures, carbonyl sulfide (COS) offers a safer alternative with manageable reactivity. The process operates at low pressures (0.1-1 MPa) and ambient temperatures, reducing the risk profile associated with high-energy carbonylation reactions.
Q: Does this catalytic system require expensive ligands or additives?
A: No, one of the key economic advantages of this technology is its simplicity. It utilizes inexpensive inorganic sulfides like sodium hydrosulfide (NaHS) as the activator. The system does not require expensive transition metal ligands, co-catalysts, or additional dehydrating agents, which streamlines the purification process and lowers overall production costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzothiazole-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the COS-mediated cyclization described in CN108101863B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into reliable industrial supply. Our facility is equipped with state-of-the-art high-pressure reactors and rigorous QC labs capable of meeting stringent purity specifications required by global pharmaceutical regulators. We understand that the consistency of heterocyclic intermediates is paramount for the efficacy and safety of the final drug product, and our quality management systems are designed to guarantee batch-to-batch uniformity.
We invite R&D directors and procurement specialists to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data for our benzothiazole derivatives and to discuss route feasibility assessments for your upcoming projects, ensuring a secure and competitive supply chain for your critical intermediates.
