Advanced Synthesis of Pyrazine-Containing Triazole Derivatives for Antifungal Drug Development
Advanced Synthesis of Pyrazine-Containing Triazole Derivatives for Antifungal Drug Development
The pharmaceutical landscape is continuously evolving with the demand for novel antifungal agents that can overcome resistance mechanisms inherent in pathogenic fungi. Patent CN103373987A presents a significant advancement in this domain by disclosing a comprehensive class of pyrazine ring-containing triazole derivatives. These molecules represent a strategic fusion of two potent pharmacophores: the pyrazine ring, known for its diverse biological activities including antibacterial and antitubercular properties, and the 1,2,4-triazole moiety, which is a cornerstone in modern antifungal therapy found in drugs like fluconazole and voriconazole. The structural integrity and synthetic accessibility of these derivatives make them highly attractive candidates for lead optimization programs. By integrating a pyrazine unit directly onto the triazole scaffold, the patent outlines a pathway to create libraries of compounds with potentially enhanced bioavailability and target affinity. This technical insight report analyzes the synthetic methodology and commercial implications of this technology for global pharmaceutical manufacturers.
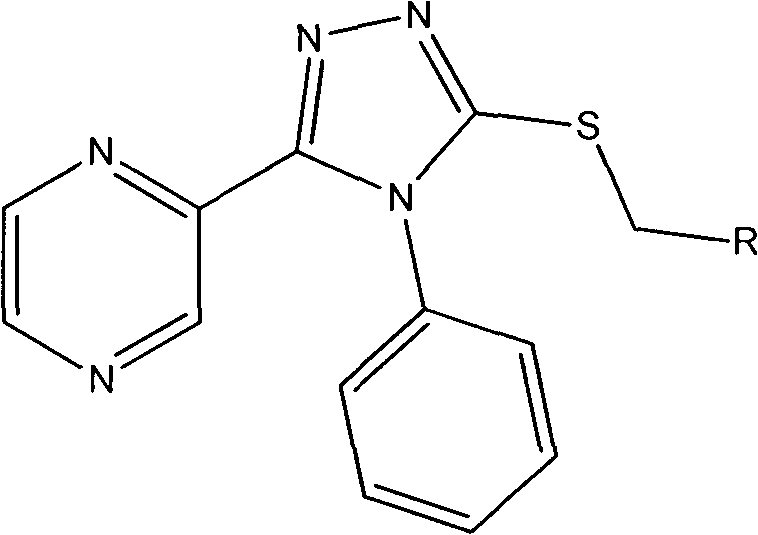
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex heterocyclic systems combining nitrogen-rich rings like pyrazine and triazole has been fraught with challenges regarding regioselectivity and reaction conditions. Conventional methods often rely on harsh acidic or basic environments that can degrade sensitive functional groups, leading to lower overall yields and difficult purification processes. Furthermore, traditional routes to functionalized triazoles frequently involve multi-step sequences with poor atom economy, generating substantial chemical waste. The introduction of substituents at specific positions on the triazole ring often requires protecting group strategies, which add unnecessary steps, time, and cost to the manufacturing process. In the context of antifungal drug development, where rapid iteration of chemical structures is required to optimize potency and safety profiles, these inefficiencies create significant bottlenecks. The reliance on expensive transition metal catalysts in some cross-coupling approaches also introduces the risk of heavy metal contamination, necessitating rigorous and costly downstream purification to meet stringent regulatory standards for pharmaceutical intermediates.
The Novel Approach
The methodology described in patent CN103373987A offers a streamlined and robust alternative that addresses many of these historical pain points. The novel approach utilizes a modular synthetic strategy that builds the triazole ring in situ from readily available precursors, specifically leveraging the reaction between a pyrazine-derived hydrazide and phenyl isothiocyanate. This cyclization strategy is highly efficient, proceeding under relatively mild reflux conditions in common solvents like ethanol. A key advantage of this route is the late-stage diversification capability; the final alkylation step allows for the facile introduction of various substituted benzyl groups without disturbing the core heterocyclic system. This modularity enables medicinal chemists to rapidly generate a focused library of analogs by simply changing the benzyl bromide reagent. The process avoids the use of precious metal catalysts, relying instead on inexpensive bases like sodium hydroxide, which drastically simplifies the workup procedure and reduces the environmental footprint of the synthesis. This combination of operational simplicity and structural flexibility makes the novel approach superior for both laboratory-scale discovery and potential commercial scale-up.
Mechanistic Insights into Base-Mediated Triazole Cyclization
The core chemical transformation in this synthesis is the formation of the 1,2,4-triazole ring via a base-mediated cyclization of a thiosemicarbazide intermediate. Mechanistically, the process begins with the nucleophilic attack of the hydrazide nitrogen on the electrophilic carbon of the phenyl isothiocyanate, forming a stable thiosemicarbazide linkage. Upon treatment with sodium hydroxide in an aqueous or alcoholic medium, the thioamide proton is abstracted, generating a nucleophilic anion. This anion then undergoes an intramolecular cyclization, attacking the adjacent carbonyl carbon (originally from the hydrazide), followed by dehydration to aromatize the triazole ring. This specific pathway is advantageous because it ensures the correct regiochemistry of the triazole substituents, placing the pyrazine ring at the 3-position and the phenyl group at the 4-position with high fidelity. The robustness of this mechanism is evidenced by the consistent yields reported across different examples in the patent, suggesting that the electronic effects of the pyrazine ring do not significantly hinder the cyclization efficiency. Understanding this mechanism is crucial for process chemists aiming to optimize reaction parameters such as temperature and base concentration for large-scale production.
Following the formation of the triazole core, the final step involves a nucleophilic substitution reaction where the triazole nitrogen acts as a nucleophile to displace the bromide ion from various substituted benzyl bromides. This SN2 type reaction is highly sensitive to steric hindrance and the electronic nature of the benzyl ring. The patent demonstrates that a wide range of substituents, including electron-withdrawing halogens like fluorine, chlorine, and bromine at ortho, meta, and para positions, are well-tolerated. 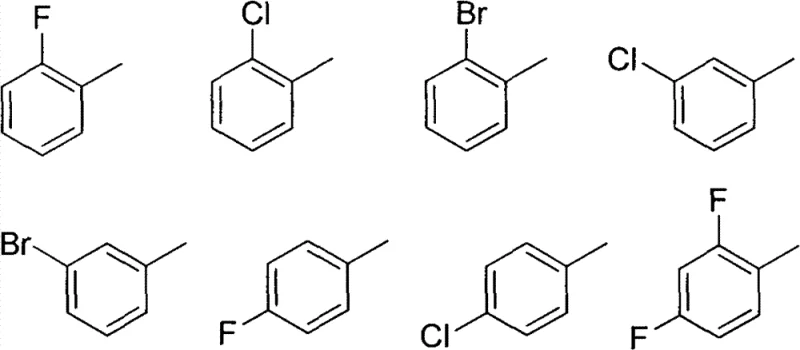 As shown in the structural variations, the presence of halogen atoms not only provides handles for further chemical modification but also enhances the lipophilicity of the final molecule, which is a critical parameter for membrane permeability and oral bioavailability in antifungal drugs. The ability to incorporate these diverse groups without compromising the yield of the alkylation step highlights the versatility of the synthetic route. Impurity control in this step is managed by careful stoichiometry and the use of anhydrous acetonitrile, which minimizes hydrolysis of the benzyl bromide and ensures high purity of the final sulfide-linked product.
As shown in the structural variations, the presence of halogen atoms not only provides handles for further chemical modification but also enhances the lipophilicity of the final molecule, which is a critical parameter for membrane permeability and oral bioavailability in antifungal drugs. The ability to incorporate these diverse groups without compromising the yield of the alkylation step highlights the versatility of the synthetic route. Impurity control in this step is managed by careful stoichiometry and the use of anhydrous acetonitrile, which minimizes hydrolysis of the benzyl bromide and ensures high purity of the final sulfide-linked product.
How to Synthesize Pyrazine Triazole Derivatives Efficiently
The synthesis of these high-value pharmaceutical intermediates follows a logical six-step sequence that balances yield and operational ease. The process initiates with the esterification of 2-pyrazinecarboxylic acid, followed by conversion to the corresponding hydrazide. This hydrazide is then condensed with phenyl isothiocyanate to form the key thiosemicarbazide precursor. The subsequent cyclization and alkylation steps finalize the core structure. For detailed operational parameters, safety precautions, and specific stoichiometric ratios required for GMP-compliant manufacturing, please refer to the standardized synthesis guide below.
- Esterify 2-pyrazinecarboxylic acid with ethanol using sulfuric acid catalyst under reflux.
- Convert the ester to hydrazide using hydrazine hydrate, then react with phenyl isothiocyanate.
- Cyclize the intermediate with sodium hydroxide to form the triazole core, followed by alkylation with substituted benzyl bromides.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the synthetic route disclosed in this patent offers compelling advantages that translate directly into cost savings and supply security. The reliance on commodity chemicals rather than specialized reagents mitigates the risk of supply chain disruptions. For procurement managers, this means greater negotiating power with vendors and more stable pricing over time. The elimination of transition metal catalysts removes the need for expensive scavenging resins and complex analytical testing for residual metals, which are significant cost drivers in API manufacturing. Furthermore, the high modularity of the synthesis allows for a "platform" approach to manufacturing, where the same intermediate can be diverted to produce multiple final analogs based on market demand, thereby optimizing inventory management and reducing waste associated with obsolete intermediates.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing inexpensive solvents like ethanol and acetonitrile, which are easily recovered and recycled in a closed-loop system. The avoidance of cryogenic conditions or high-pressure equipment reduces capital expenditure requirements for production facilities. Additionally, the high atom economy of the cyclization step minimizes raw material consumption per kilogram of product. By streamlining the purification process to simple recrystallization and extraction, the method reduces labor hours and solvent usage compared to chromatography-dependent routes. These factors collectively contribute to a substantially lower cost of goods sold (COGS), making the final antifungal intermediates more competitive in the global market.
- Enhanced Supply Chain Reliability: The starting materials, including 2-pyrazinecarboxylic acid and various substituted benzyl bromides, are widely available from multiple global suppliers, ensuring a resilient supply chain. This redundancy prevents single-source bottlenecks that can delay clinical trials or commercial launches. The robustness of the reaction conditions, which tolerate minor variations in temperature and reagent quality, ensures consistent batch-to-batch reproducibility. This reliability is critical for maintaining continuous production schedules and meeting strict delivery timelines required by downstream pharmaceutical partners. The stability of the intermediates also allows for safer storage and transportation, further de-risking the logistics network.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable, having been demonstrated effectively from milligram to multi-gram scales in the patent examples without loss of efficiency. The absence of toxic heavy metals aligns with increasingly stringent environmental regulations and green chemistry principles, simplifying the permitting process for new manufacturing lines. Waste streams are primarily composed of organic solvents and saline solutions, which are easier to treat and dispose of compared to hazardous metal-containing waste. This environmental compatibility not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturing entity, a key consideration for modern supply chain heads evaluating potential partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these pyrazine-triazole derivatives. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals.
Q: What is the primary application of these pyrazine-triazole derivatives?
A: These compounds are designed as potential antifungal agents, leveraging the known biological activity of both pyrazine and triazole pharmacophores.
Q: Does the synthesis require expensive transition metal catalysts?
A: No, the described method utilizes common reagents like sulfuric acid and sodium hydroxide, avoiding costly heavy metal catalysts and simplifying purification.
Q: How is structural diversity achieved in this series?
A: Diversity is introduced in the final alkylation step by varying the substituted benzyl bromide, allowing for fluorine, chlorine, or bromine substitutions on the benzyl ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazine Triazole Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of heterocyclic chemistry, ensuring stringent purity specifications and rigorous QC labs that validate every batch against international pharmacopeia standards. We understand the critical nature of antifungal intermediate supply and are committed to delivering high-quality materials that accelerate your drug development timelines. Our technical team is adept at troubleshooting process deviations and optimizing yields to maximize value for our partners.
We invite you to collaborate with us to leverage this innovative synthetic technology for your next-generation antifungal programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your commercial goals efficiently and reliably.
