Revolutionizing 3,4-Methylenedioxyacetophenone Production via Solid Acid Catalysis for Commercial Scale
Revolutionizing 3,4-Methylenedioxyacetophenone Production via Solid Acid Catalysis for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally compliant synthetic routes for critical intermediates. A significant breakthrough in this domain is detailed in Chinese Patent CN111072625A, which discloses a novel preparation method for 3,4-methylenedioxyacetophenone, a pivotal building block for cardiovascular drugs and flavoring agents like heliotropin. This technology represents a paradigm shift from traditional homogeneous Lewis acid catalysis to a heterogeneous solid acid system utilizing modified diatomite. By leveraging the unique porous structure and surface hydroxyl groups of diatomite, modified with sulfonic acid groups, the process achieves high conversion rates under mild conditions while drastically simplifying downstream processing. For R&D directors and procurement managers alike, this innovation offers a compelling value proposition: the elimination of expensive, corrosive metal catalysts translates directly into reduced operational expenditures and a greener manufacturing footprint, positioning suppliers who adopt this technology as leaders in sustainable chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4-methylenedioxyacetophenone has relied heavily on classical Friedel-Crafts acylation using stoichiometric amounts of strong Lewis acids such as aluminum chloride (AlCl3) or zinc chloride (ZnCl2). While these methods are chemically effective, they suffer from severe industrial drawbacks that impact both cost and environmental compliance. The use of metal halides generates substantial quantities of acidic wastewater and hazardous sludge during the quenching and workup phases, creating a significant burden on waste treatment facilities. Furthermore, the corrosive nature of these catalysts necessitates the use of specialized, expensive reactor linings and increases maintenance costs due to equipment degradation. The post-reaction separation is notoriously difficult, often requiring complex extraction and purification steps to remove trace metal residues that could contaminate the final API or flavor product, thereby compromising purity specifications and increasing the overall cost of goods sold.
The Novel Approach
In stark contrast, the methodology outlined in the patent utilizes a diatomite-based solid acid catalyst, which fundamentally alters the reaction landscape by introducing heterogeneity to the acylation process. This approach allows for the catalyst to be easily separated from the reaction mixture via simple hot filtration, eliminating the need for aqueous quenching and the associated generation of heavy metal waste. The reaction conditions are notably milder, typically operating between 60°C and 120°C, which reduces energy consumption and minimizes the risk of thermal degradation of sensitive substrates. By replacing corrosive liquid acids with a stable solid support, the process not only enhances operator safety but also extends the lifespan of standard stainless steel reactors. This technological leap ensures that the production of high-purity 3,4-methylenedioxyacetophenone becomes more economically viable and environmentally sustainable, addressing the critical pain points of modern chemical manufacturing.
Mechanistic Insights into Diatomite-Solid Acid Catalyzed Acylation
The efficacy of this synthesis lies in the sophisticated modification of the diatomite support, which transforms a naturally occurring siliceous sedimentary rock into a potent solid acid catalyst. The process begins with the thermal activation of diatomite to remove adsorbed water, followed by treatment with a sulfonating agent such as chlorosulfonic acid. This modification grafts strong acidic sulfonic acid groups (-SO3H) onto the extensive surface area of the diatomite, creating active sites capable of generating the acylium ion necessary for the electrophilic aromatic substitution. The porous structure of the diatomite provides a high surface-to-volume ratio, ensuring excellent dispersion of the active acid sites and facilitating efficient mass transfer between the liquid reactants and the solid catalyst surface. This heterogeneous mechanism prevents the aggregation of active species and allows for a controlled reaction environment that favors the formation of the desired para-substituted product while minimizing polyacylation side reactions.
From an impurity control perspective, the solid acid mechanism offers distinct advantages over homogeneous catalysis. In traditional systems, the strong coordination between the product ketone and the metal Lewis acid often leads to catalyst deactivation and requires excess reagent to drive the reaction to completion, which in turn promotes side reactions. The diatomite solid acid, however, interacts with the substrate primarily through surface protonation, allowing the product to desorb more readily once formed. This dynamic equilibrium reduces the residence time of the product in the reactive zone, thereby suppressing the formation of oligomeric byproducts and tars. Additionally, the absence of metal ions eliminates the risk of metal-catalyzed oxidation or rearrangement of the methylenedioxy ring, a common degradation pathway in harsh acidic environments. Consequently, the crude product obtained from this process exhibits a cleaner profile, requiring less intensive purification to meet the stringent quality standards demanded by the pharmaceutical and flavor industries.
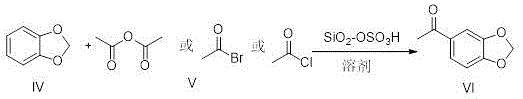
How to Synthesize 3,4-Methylenedioxyacetophenone Efficiently
Implementing this advanced synthetic route requires precise control over catalyst preparation and reaction parameters to maximize yield and efficiency. The protocol involves a two-stage process where the catalyst is first activated and modified before being introduced to the acylation reactor. Operators must ensure that the diatomite is thoroughly dried to prevent hydrolysis of the sulfonating agent, which would otherwise reduce the acidity of the final catalyst. Once the catalyst is prepared, the acylation is conducted in a chlorinated solvent, with dichloroethane being the preferred medium due to its ability to dissolve both the organic substrate and the acylating agent while maintaining stability under reflux conditions. The detailed standardized synthesis steps, including specific molar ratios, temperature ramps, and workup procedures, are outlined in the guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the diatomite solid acid catalyst by drying diatomite at 110-250°C and modifying it with chlorosulfonic acid at room temperature.
- Combine the catalyst, solvent (dichloroethane), 1,2-methylenedioxybenzene, and acylating agent (acetic anhydride) in a reactor.
- Heat the mixture to 60-120°C for 5-10 hours, then filter to recover the catalyst and purify the product via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this diatomite-catalyzed process translates into tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the supply chain for raw materials; diatomite is an abundant, low-cost natural resource, unlike specialized organometallic catalysts which are subject to volatile pricing and geopolitical supply risks. By shifting to a catalyst that can be potentially regenerated or disposed of with minimal environmental impact, manufacturers can significantly lower their total cost of ownership. Furthermore, the elimination of metal halides removes the need for expensive metal scavenging resins or complex distillation trains dedicated to removing trace metals, streamlining the production workflow and reducing utility consumption. This efficiency gain allows for more competitive pricing structures for the final intermediate, providing a buffer against market fluctuations.
- Cost Reduction in Manufacturing: The transition to a solid acid catalyst fundamentally alters the cost structure of 3,4-methylenedioxyacetophenone production by removing the dependency on stoichiometric quantities of expensive Lewis acids. In conventional processes, the catalyst is consumed and becomes part of the waste stream, whereas the solid acid can be filtered and potentially reused, leading to substantial savings in raw material costs. Additionally, the simplified workup procedure reduces the consumption of water, neutralizing agents, and solvents required for extraction, directly lowering variable production costs. The reduction in equipment corrosion also implies lower capital expenditure on maintenance and replacement of reactor components, contributing to long-term financial stability and improved profit margins for the manufacturing entity.
- Enhanced Supply Chain Reliability: Sourcing high-purity intermediates is often bottlenecked by the availability of specialized reagents, but this method relies on commodity chemicals that are widely available globally. Diatomite and common acylating agents like acetic anhydride are produced at massive scales for various industries, ensuring a stable and continuous supply chain that is resilient to disruptions. The robustness of the catalyst preparation also means that production can be ramped up quickly without waiting for long lead-time deliveries of exotic catalysts. This reliability is crucial for maintaining just-in-time inventory levels and meeting the strict delivery schedules required by downstream pharmaceutical clients, thereby strengthening the supplier-client relationship and reducing the risk of production stoppages.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial waste disposal intensify, this green chemistry approach offers a future-proof solution for commercial scale-up. The reduction in hazardous waste generation simplifies the permitting process for new production lines and reduces the liability associated with waste management. The mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors without the need for exotic alloys, facilitating easier technology transfer from pilot plant to full commercial production. This scalability ensures that suppliers can meet increasing demand for high-purity 3,4-methylenedioxyacetophenone without compromising on environmental standards, aligning with the corporate sustainability goals of major multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this diatomite-catalyzed synthesis. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios. The answers highlight the balance between performance, cost, and operational simplicity that defines this innovative approach.
Q: What are the primary advantages of using diatomite solid acid over traditional Lewis acids like AlCl3?
A: The diatomite solid acid catalyst eliminates the need for stoichiometric amounts of corrosive metal halides, significantly simplifying post-treatment by allowing simple filtration instead of complex aqueous quenching, thereby reducing three wastes and equipment corrosion.
Q: What yields can be expected from this novel acylation process?
A: According to patent data, the process achieves high yields ranging from approximately 60% to over 90%, with optimized conditions using acetic anhydride and chlorosulfonic acid-modified diatomite reaching up to 90.3%.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the use of inexpensive, commercially available diatomite and mild reaction conditions (60-120°C) makes this method highly scalable, offering substantial cost savings and improved environmental compliance compared to traditional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Methylenedioxyacetophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is not just a regulatory requirement but a strategic imperative for maintaining competitiveness in the global fine chemical market. Our technical team has extensively analyzed the potential of solid acid catalysis, such as the diatomite-based system described in CN111072625A, and we possess the expertise to adapt and optimize these pathways for industrial application. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of laboratory patents are fully realized in large-scale manufacturing. Our state-of-the-art rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of 3,4-methylenedioxyacetophenone we deliver meets the exacting standards required for pharmaceutical and flavor applications.
We invite forward-thinking partners to collaborate with us to leverage these technological advancements for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this novel production method for your specific volume requirements. We encourage you to reach out today to discuss your project needs,索取 specific COA data, and review our comprehensive route feasibility assessments. Let us help you secure a reliable, cost-effective, and sustainable supply of this critical intermediate, driving value and innovation in your final products.
