Scalable Electrochemical Synthesis of Vilacatide: A Green Route for Commercial API Manufacturing
The pharmaceutical landscape for treating secondary hyperparathyroidism in chronic kidney disease patients has been significantly advanced by the introduction of calcimimetic agents like Vilacatide (also known as Etelcalcetide). As demand for this intravenous polypeptide drug grows, the industry faces a critical challenge: developing a manufacturing process that balances high purity with environmental sustainability and cost-efficiency. Patent CN112062811A presents a groundbreaking solution by introducing a solid-phase electrochemical oxidation method for constructing the critical disulfide bond. This innovation moves away from traditional, hazardous chemical oxidants, offering a pathway that drastically improves atom economy and simplifies downstream purification. For R&D directors and procurement strategists, this patent represents a pivotal shift towards greener, more robust peptide manufacturing protocols that align with modern regulatory and economic pressures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex peptides like Vilacatide has been plagued by the difficulty of forming specific disulfide bonds without generating structural impurities. Conventional methods relying on air oxidation, iodine/acetic acid systems, or hydrogen peroxide often lack the necessary specificity, inevitably leading to mismatched disulfide bonds and scrambled peptide sequences that are notoriously difficult to separate. Furthermore, previous patents such as CN106928320 utilized 2,2'-dipyridyl disulfide activation; while effective for coupling, this approach generates stoichiometric amounts of mercaptopyridine byproducts. These sulfur-containing impurities not only complicate the purification process, requiring extensive chromatography, but also pose significant environmental disposal challenges. Additionally, liquid-phase synthesis strategies often require pseudo-dilution conditions to prevent inter-chain polymerization, resulting in massive solvent consumption and low volumetric productivity, which is economically unsustainable for large-scale commercial operations.
The Novel Approach
The methodology outlined in CN112062811A revolutionizes this process by integrating electrochemical oxidation directly into the solid-phase synthesis workflow. Instead of adding external chemical oxidants, the system utilizes a controlled electric current to drive the formation of the disulfide bond between the resin-bound peptide and free L-Cysteine. This approach ensures that the oxidation occurs specifically at the active site on the resin, minimizing the risk of inter-chain mismatches that plague solution-phase reactions. By avoiding the use of 2,2'-dipyridyl disulfide, the process eliminates the generation of toxic mercaptopyridine waste entirely, adhering to the principles of green chemistry. The result is a streamlined workflow where the disulfide bridge is constructed with high fidelity on the solid support, leading to a crude product with significantly higher purity and a much simpler purification profile compared to legacy methods.
Mechanistic Insights into Solid-Phase Electrochemical Oxidation
The core of this technological advancement lies in the precise control of the electrochemical environment to facilitate thiol-disulfide exchange. In this mechanism, the polypeptide-resin, with the Cysteine side-chain protecting group removed, acts as the substrate in an electrolytic cell containing a supporting electrolyte such as tetrabutylammonium tetrafluoroborate. When a constant current (optimized between 5-20 mA) is applied across platinum electrodes, the thiol groups are electrochemically activated. This activation promotes the nucleophilic attack by the free L-Cysteine in the solution onto the resin-bound thiol, forming the heterodisulfide bond. Unlike chemical oxidation where reagent concentration gradients can lead to uneven reaction rates, the electrochemical potential provides a uniform driving force across the resin bed. This uniformity is crucial for ensuring that every peptide chain on the resin undergoes the coupling reaction simultaneously, thereby maximizing the utilization rate of the expensive D-amino acid backbone fragments and preventing the accumulation of unreacted starting materials.
From an impurity control perspective, this mechanism offers superior selectivity. Traditional chemical oxidants are often indiscriminate, potentially oxidizing other sensitive residues like Methionine or Tryptophan if present, or causing over-oxidation to sulfinic or sulfonic acids. The electrochemical method allows for fine-tuning of the oxidation potential, keeping it within the window required for disulfide formation while sparing other functional groups. Moreover, because the reaction happens on the solid phase, any unreacted L-Cysteine or electrolyte salts can be simply washed away with DMF or DCM before the final cleavage step. This "wash-away" capability inherent to solid-phase synthesis means that the final cleavage mixture contains far fewer organic impurities, directly translating to higher crude purity (reported up to 89.7% in examples) and reducing the burden on the final preparative HPLC purification step.
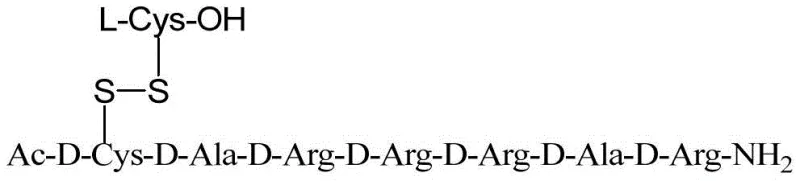
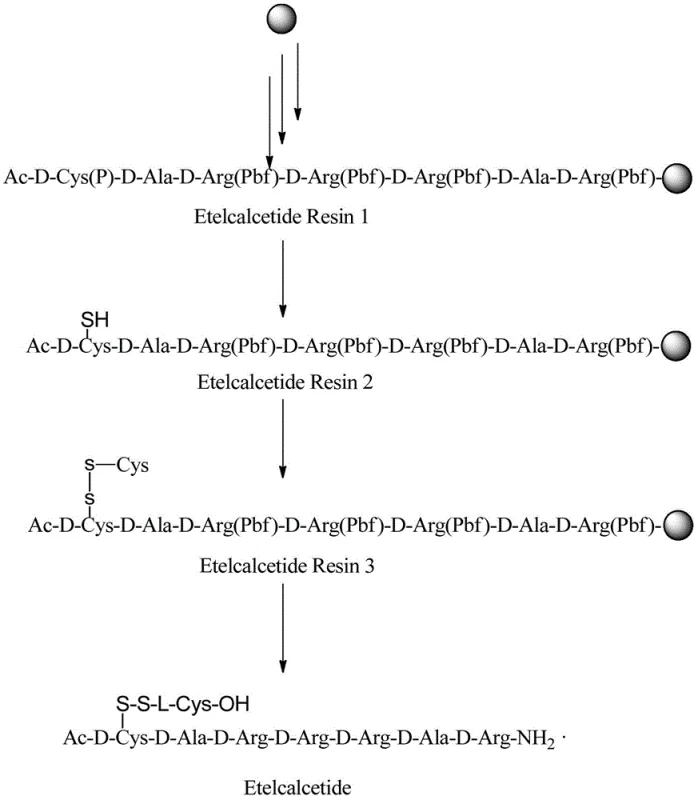
How to Synthesize Vilacatide Efficiently
The synthesis of Vilacatide via this novel route begins with the assembly of the heptapeptide backbone on a Rink Amide resin using standard Fmoc/tBu solid-phase peptide synthesis (SPPS) strategies. The sequence Ac-D-Cys-D-Ala-D-Arg-D-Arg-D-Arg-D-Ala-D-Arg is built from the C-terminus to the N-terminus, utilizing efficient coupling reagents like DIPCDI/HOBt to ensure high coupling yields at each step. Once the backbone is fully assembled and acetylated, the critical side-chain protecting group on the Cysteine residue (such as Mmt or Trt) is selectively removed using mild acidic conditions that leave the rest of the peptide intact. Following this deprotection, the resin is subjected to the electrochemical cell containing L-Cysteine hydrochloride and the electrolyte. After the electrolysis is complete, typically running for several hours at controlled temperatures between 25-65°C, the resin is washed thoroughly. The final step involves global deprotection and cleavage from the resin using a TFA-based cocktail, releasing the crude Vilacatide which is then precipitated and purified. Detailed standardized synthesis steps are provided in the guide below.
- Prepare the fully-protected Vilacatide main chain on solid-phase resin using standard Fmoc chemistry, ending with Acetylation.
- Selectively deprotect the side-chain protecting group (P) of the Cysteine residue on the resin using mild acidic conditions.
- Perform solid-phase electrochemical oxidation in the presence of L-Cysteine and electrolyte to form the critical disulfide bond.
- Cleave the peptide from the resin and remove all remaining protecting groups using TFA cocktail, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical synthesis route offers compelling economic and operational benefits that extend beyond simple yield improvements. The primary advantage lies in the drastic simplification of the raw material portfolio and waste management protocols. By eliminating the need for specialized and hazardous oxidizing agents like 2,2'-dipyridyl disulfide, companies can reduce their dependency on volatile chemical supply chains and lower the costs associated with handling and disposing of toxic sulfur-containing waste streams. This shift not only lowers the direct cost of goods sold (COGS) but also mitigates regulatory risks associated with environmental compliance, ensuring a more stable and uninterrupted supply of the API for downstream drug product manufacturing.
- Cost Reduction in Manufacturing: The elimination of expensive chemical oxidants and the reduction in solvent usage due to the avoidance of pseudo-dilution conditions lead to substantial cost savings. Furthermore, the improved atom economy means that the expensive D-amino acids, which constitute the bulk of the raw material cost, are utilized much more efficiently. The reduction in downstream purification complexity also translates to lower processing times and reduced consumption of chromatography columns and solvents, driving down the overall manufacturing cost per gram of high-purity Vilacatide.
- Enhanced Supply Chain Reliability: Relying on electricity as the primary "reagent" for the critical bond-forming step decouples the process from the supply volatility of niche chemical oxidants. Electrochemical cells are robust and scalable, allowing for consistent production batches regardless of external chemical market fluctuations. This reliability is crucial for maintaining continuous supply to pharmaceutical partners, especially for life-saving drugs like Vilacatide used in dialysis patients where treatment interruptions are not an option.
- Scalability and Environmental Compliance: The solid-phase nature of the reaction combined with electrochemical control makes this process highly amenable to scale-up. Unlike liquid-phase reactions that require massive reactors to maintain dilution, solid-phase reactors can be scaled by increasing resin volume without proportionally increasing solvent volumes. Additionally, the "green" nature of the process, characterized by the absence of toxic byproducts and reduced solvent waste, aligns perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing facility against tighter emission standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of Vilacatide. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring that stakeholders have accurate information for decision-making. Understanding these nuances is essential for evaluating the feasibility of technology transfer and the potential return on investment for adopting this greener manufacturing platform.
Q: Why is electrochemical oxidation superior to traditional chemical oxidation for Vilacatide?
A: Traditional methods using air, iodine, or 2,2'-dipyridyl disulfide often suffer from poor selectivity, leading to mismatched disulfide impurities and difficult purification. The electrochemical method described in CN112062811A offers precise control over the oxidation potential, significantly reducing mismatched byproducts and eliminating toxic mercaptopyridine waste.
Q: What are the key cost drivers eliminated in this new synthetic route?
A: This route eliminates the need for expensive and hazardous oxidizing agents like 2,2'-dipyridyl disulfide. Furthermore, by performing the coupling on the solid phase, it avoids the pseudo-dilution conditions required in liquid phase synthesis, thereby improving atom economy and reducing the massive solvent volumes typically associated with inter-chain disulfide formation.
Q: Is this electrochemical process scalable for industrial production?
A: Yes, the process utilizes standard solid-phase synthesis equipment adapted with platinum electrodes. The reaction conditions (current intensity 5-20 mA, temperature 25-65°C) are mild and controllable, making the transition from laboratory scale to commercial multi-kilogram production feasible without complex reactor engineering.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vilacatide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing technologies like electrochemical synthesis requires a partner with deep technical expertise and proven scalability. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent examples can be reliably translated to industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped to handle complex peptide analysis, guaranteeing that every batch of Vilacatide meets the highest international pharmacopoeia standards. Our commitment to green chemistry aligns with this new patent, allowing us to offer a sustainable supply chain solution for your peptide API needs.
We invite you to discuss how this innovative electrochemical route can optimize your supply chain and reduce costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term commercial goals for Vilacatide and other complex peptide therapeutics.
