Advanced Synthesis of 2,4-Dicumylphenol: Overcoming Selectivity Challenges with Graphene Oxide Catalysis
The global demand for high-performance antioxidants in polymer stabilization and fuel processing continues to surge, driving the need for more efficient synthetic routes for key intermediates like 2,4-dicumylphenol. A recent technological breakthrough detailed in patent CN116283504A introduces a novel catalytic system that addresses long-standing selectivity issues in the Friedel-Crafts alkylation of phenol. This method leverages a synergistic combination of conventional acid catalysts, such as p-toluenesulfonic acid, and advanced nanomaterials, specifically graphene oxide, to achieve superior conversion rates. By integrating these components, the process effectively mitigates the formation of polysubstituted byproducts that have historically plagued industrial production. For R&D directors and procurement specialists, this innovation represents a significant opportunity to enhance supply chain reliability for critical polymer additives. The following analysis dissects the technical merits and commercial implications of this graphene oxide-assisted synthesis.
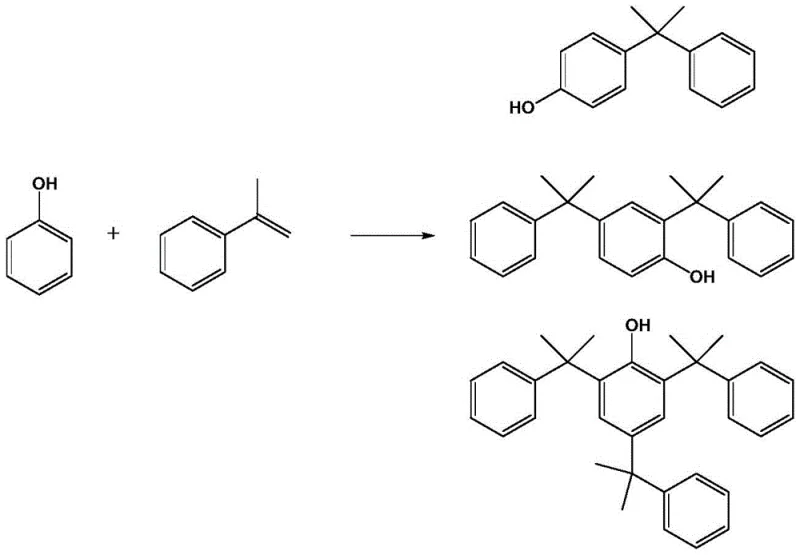
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of 2,4-dicumylphenol typically relies on the reaction between phenol and alpha-methylstyrene under acidic conditions using catalysts like concentrated sulfuric acid or cation exchange resins. However, this classical approach suffers from inherent kinetic challenges due to the activating nature of the alkyl groups on the phenolic ring. Once the first cumyl group is introduced to form 4-cumylphenol, the ring becomes even more susceptible to further electrophilic attack, leading to uncontrolled over-alkylation. Consequently, significant quantities of 2,4,6-tricumylphenol are generated as a persistent impurity, which is difficult to separate and reduces the overall yield of the desired di-substituted product. Furthermore, alpha-methylstyrene is prone to self-polymerization under harsh acidic conditions, generating dimers and trimers that complicate the purification process and increase waste disposal costs. These inefficiencies result in variable product quality and inconsistent batch-to-batch performance, posing risks for downstream applications in sensitive polymer formulations.
The Novel Approach
The methodology disclosed in the patent fundamentally alters the reaction landscape by introducing graphene oxide as a heterogeneous co-catalyst alongside p-toluenesulfonic acid. This dual-catalyst system operates under milder thermal conditions, typically between 90°C and 100°C, which inherently reduces the energy footprint of the process. The unique two-dimensional planar structure of graphene oxide plays a critical role in modulating the reaction pathway, providing a steric environment that favors the formation of the 2,4-disubstituted product while physically hindering the approach of a third alpha-methylstyrene molecule. This spatial constraint effectively suppresses the formation of the tri-substituted impurity, pushing the selectivity towards the target molecule to unprecedented levels. Additionally, the presence of graphene oxide appears to stabilize the reaction medium, minimizing the oligomerization of the olefinic starting material. The result is a cleaner reaction profile that simplifies downstream processing and maximizes the utilization of raw materials.
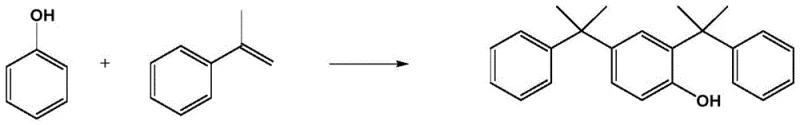
Mechanistic Insights into Graphene Oxide-Assisted Friedel-Crafts Alkylation
The core of this technological advancement lies in the cooperative catalysis mechanism between the Brønsted acid and the functionalized carbon nanomaterial. The p-toluenesulfonic acid provides the necessary protons to activate the alpha-methylstyrene, generating the reactive carbocation intermediate required for the electrophilic aromatic substitution. Simultaneously, the oxygen-containing functional groups on the graphene oxide surface likely interact with the reactants, potentially stabilizing transition states or facilitating proton transfer. More importantly, the physical morphology of the graphene oxide sheets creates a confined reaction space. As the reaction progresses from phenol to 4-cumylphenol and finally to 2,4-dicumylphenol, the increasing steric bulk of the molecule makes it increasingly difficult to access the active sites on the catalyst surface in an orientation favorable for a third substitution. This steric inhibition is the key to the high selectivity observed, ensuring that the reaction stops predominantly at the di-substituted stage. This mechanistic control is crucial for maintaining a narrow impurity profile, which is a primary concern for quality assurance teams in the fine chemicals sector.
Impurity control is further enhanced by the suppression of side reactions involving the alpha-methylstyrene monomer. In conventional systems, the strong acidity and elevated temperatures often trigger cationic polymerization of the styrene derivative, leading to tarry residues that reduce reactor efficiency and require aggressive cleaning protocols. The graphene oxide-modified system appears to moderate the effective acidity experienced by the bulk solution, preventing the runaway polymerization of the olefin. This results in a reaction mixture that is easier to handle and work up, with fewer heavy ends to dispose of. For process engineers, this translates to reduced downtime for reactor maintenance and lower consumption of solvents during the extraction and washing phases. The ability to recycle the graphene oxide catalyst after simple filtration and washing further underscores the robustness of this catalytic cycle, offering a sustainable alternative to homogeneous acid systems that generate large volumes of saline wastewater during neutralization.
How to Synthesize 2,4-Dicumylphenol Efficiently
The operational protocol for this synthesis is designed for scalability and ease of execution within standard chemical manufacturing facilities. The process begins with the preparation of the catalytic mixture under an inert atmosphere to prevent oxidation of sensitive components. Precise control of the addition rate of alpha-methylstyrene is critical to manage the exotherm and maintain the optimal temperature window for selectivity. Following the reaction, the workup procedure leverages the heterogeneous nature of the graphene oxide, allowing for its physical separation prior to neutralization, which streamlines the purification workflow. The detailed standardized synthesis steps, including specific molar ratios, temperature ramps, and isolation techniques, are outlined in the guide below for technical reference.
- Under inert gas protection, mix molten phenol, p-toluenesulfonic acid catalyst, and graphene oxide in a reactor, then heat to 50-80°C.
- Slowly add alpha-methylstyrene while maintaining the system temperature between 90-100°C, then continue stirring for 0.5 to 1 hour.
- Cool the mixture, filter to recover graphene oxide, neutralize with base, extract with organic solvent, and purify via distillation and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this graphene oxide-assisted synthesis offers compelling economic and logistical benefits that extend beyond mere chemical yield. The primary value driver is the substantial reduction in raw material waste achieved through improved selectivity. By minimizing the formation of tri-substituted byproducts and olefin polymers, the process ensures that a higher percentage of the expensive alpha-methylstyrene feedstock is converted into the saleable final product. This efficiency gain directly correlates to a lower cost of goods sold, providing a competitive edge in pricing negotiations with downstream polymer manufacturers. Furthermore, the simplicity of the catalyst recovery process eliminates the need for complex filtration systems or extensive wastewater treatment associated with neutralizing large quantities of homogeneous acids. This streamlined workflow reduces utility consumption and labor hours, contributing to a leaner and more cost-effective manufacturing operation.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the ability to recycle the graphene oxide co-catalyst significantly lowers the recurring material costs per batch. Since the catalyst can be recovered via simple filtration and reused multiple times without significant loss of activity, the amortized cost of the catalytic system becomes negligible over time. Additionally, the high purity of the crude product reduces the burden on the distillation and recrystallization units, leading to lower energy consumption for heating and cooling. These cumulative savings allow for a more aggressive pricing strategy while maintaining healthy profit margins, making the supply of high-purity 2,4-dicumylphenol more economically viable in a fluctuating market.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as phenol and alpha-methylstyrene ensures a stable and secure supply of raw materials, as these are produced on a massive global scale with well-established logistics networks. The robustness of the reaction conditions, which tolerate slight variations in temperature and mixing without catastrophic failure, enhances the predictability of production schedules. This operational stability minimizes the risk of batch failures or off-spec production runs that could disrupt delivery timelines to key customers. For supply chain heads, this means a more dependable source of critical antioxidants, reducing the need for excessive safety stock and enabling a more responsive just-in-time inventory model.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively with straightforward equipment configurations that do not require specialized high-pressure or cryogenic infrastructure. The reduction in hazardous waste generation, particularly the minimized saline effluent from neutralization steps, aligns with increasingly stringent environmental regulations and corporate sustainability goals. This compliance advantage reduces the regulatory burden and potential liability associated with waste disposal, facilitating smoother permitting for capacity expansions. The ability to scale from pilot batches to multi-ton commercial production with consistent quality ensures that the supply can grow in tandem with market demand for high-performance polymer additives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this method into existing production lines or sourcing strategies.
Q: What is the primary advantage of using Graphene Oxide in this synthesis?
A: Graphene oxide acts as a co-catalyst that enhances the activity of the acidic system while its planar structure sterically hinders over-alkylation, significantly suppressing the formation of unwanted 2,4,6-tricumylphenol impurities.
Q: Can the graphene oxide catalyst be recycled?
A: Yes, the patent demonstrates that graphene oxide can be recovered by filtration after the reaction, washed, dried, and reused for multiple cycles while maintaining high catalytic activity and selectivity.
Q: What are the typical purity levels achievable with this method?
A: Through optimized reaction conditions and subsequent recrystallization, the process yields 2,4-dicumylphenol with HPLC purity exceeding 99%, minimizing downstream purification costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Dicumylphenol Supplier
The synthesis of 2,4-dicumylphenol via graphene oxide catalysis exemplifies the type of process innovation that drives value in the modern fine chemicals industry. At NINGBO INNO PHARMCHEM, we specialize in translating such advanced laboratory methodologies into robust, commercial-scale manufacturing processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of high selectivity and yield are fully realized in every batch we deliver. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that our antioxidants meet the exacting standards required for polyolefin stabilization and fuel additive applications, providing our partners with unwavering confidence in product quality.
We invite you to collaborate with us to leverage these technological advantages for your supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized process can reduce your total landed costs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can support your long-term strategic goals in the polymer and specialty chemicals sectors.
