Advanced Pyridine-Based Phosphine Ligands for High-Efficiency Industrial Alkoxycarbonylation
Introduction to Next-Generation Catalytic Systems
The chemical industry is currently witnessing a paradigm shift towards more atom-economical and sustainable processes for synthesizing organic carboxylates, which serve as critical precursors for polymers like PMMA and various pharmaceutical intermediates. Patent CN115772195A introduces a groundbreaking class of pyridine-based diphosphine ligands that address the longstanding challenges of catalyst stability and selectivity in olefin alkoxycarbonylation. Unlike traditional systems that often struggle with catalyst deactivation or poor regioselectivity, these novel compounds leverage a unique pyridine backbone combined with bulky tert-butyl phosphine groups to achieve exceptional performance metrics. The general reaction scheme illustrates how olefins react with carbon monoxide and alcohols under palladium catalysis to yield esters with one additional carbon atom, a transformation of immense industrial value.

This technological advancement is particularly significant for manufacturers seeking a reliable pharmaceutical intermediate supplier or those involved in the production of fine chemicals where purity and efficiency are paramount. By enabling reaction conditions that are both milder and more selective, this innovation reduces the energy footprint and downstream purification costs associated with ester production. The ability to operate with high turnover frequencies ensures that precious metal catalysts are utilized to their maximum potential, aligning perfectly with the economic goals of modern chemical enterprises focused on cost reduction in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the alkoxycarbonylation of olefins has relied heavily on ligands such as dtbpx (1,2-bis(di-tert-butylphosphinomethyl)benzene) developed by Lucite, or ferrocene-based systems like butphos. While these ligands represented significant progress, they are not without limitations regarding long-term stability under harsh acidic conditions and the precise control of linear versus branched product ratios. In many industrial scenarios, the formation of branched by-products necessitates complex and expensive separation processes, thereby eroding profit margins. Furthermore, the synthesis of some advanced ferrocene ligands can be cumbersome, involving multiple steps and sensitive reagents that complicate the supply chain and increase the lead time for high-purity catalyst components. These factors collectively hinder the scalability and economic viability of processes relying on older generation ligand technologies.
The Novel Approach
The invention disclosed in CN115772195A offers a robust alternative by utilizing a pyridine scaffold that inherently provides different electronic and steric properties compared to benzene or ferrocene backbones. As demonstrated in the comparative studies within the patent, ligands designated as A through I exhibit remarkable catalytic activity, with conversion rates reaching up to 99.9% and linear-to-branched selectivity ratios consistently exceeding 99:1. This level of precision effectively eliminates the need for extensive downstream purification, streamlining the entire production workflow. The structural diversity shown in the ligand library allows for fine-tuning the catalyst system to specific substrate requirements, ensuring optimal performance across a range of olefins from ethylene to higher alpha-olefins.
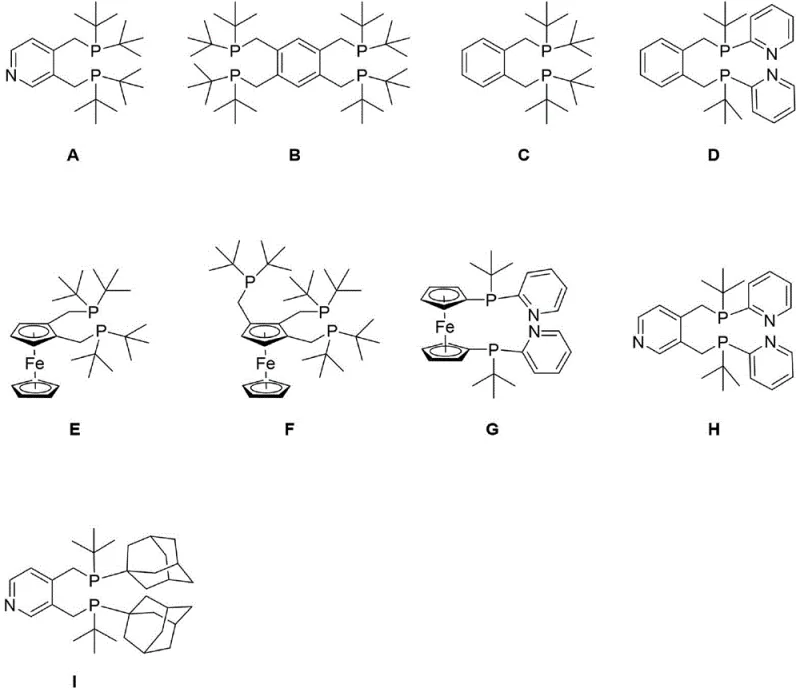
Moreover, the novel approach simplifies the catalyst preparation process itself. The ligands are designed to form stable complexes with palladium precursors rapidly, reducing the induction period often seen with less reactive phosphines. This rapid activation translates directly into higher throughput for reactor vessels, allowing manufacturers to maximize their asset utilization. For procurement managers, this means a more predictable and efficient production schedule, while R&D teams benefit from a versatile platform that can be adapted for hydroformylation and hydroaminomethylation reactions beyond simple carbonylation, expanding the potential application scope of the technology significantly.
Mechanistic Insights into Pyridine-Backbone Stabilized Palladium Catalysis
The exceptional performance of these ligands can be attributed to the specific electronic environment created by the pyridine nitrogen and the steric bulk of the di-tert-butylphosphino groups. The general structure, defined as Formula (I), features a central pyridine ring substituted at the 3 and 4 positions with methylene-linked phosphine groups. This arrangement facilitates a bite angle and electronic density at the palladium center that favors the migratory insertion of the olefin in a linear fashion. The nitrogen atom in the pyridine ring, while not necessarily coordinating strongly to the metal in the active species, exerts a significant inductive effect that modulates the electron richness of the phosphorus donors, enhancing the stability of the Pd-H and Pd-Acyl intermediates crucial for the catalytic cycle.
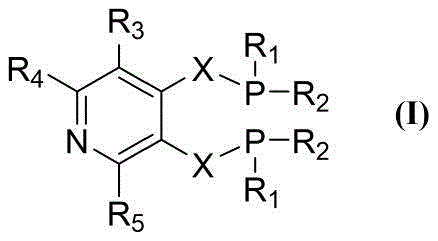
Furthermore, the steric protection provided by the tert-butyl groups on the phosphorus atoms prevents the formation of inactive palladium black or cluster species, which are common deactivation pathways in acidic carbonylation media. This resistance to decomposition is evidenced by the high Turnover Numbers (TON) reported, often exceeding 50,000 and reaching nearly 60,000 in optimized examples. Such durability implies that the catalyst remains active for extended periods, reducing the frequency of catalyst replenishment. From a mechanistic standpoint, this stability ensures a consistent impurity profile throughout the batch, which is critical for pharmaceutical applications where strict regulatory limits on heavy metals and organic impurities must be maintained.
The versatility of the substitution pattern on the pyridine ring (R3, R4, R5) and the phosphine groups (R1, R2) allows chemists to further optimize the solubility and electronic properties of the ligand. For instance, introducing electron-withdrawing or donating groups can fine-tune the nucleophilicity of the phosphorus, adapting the catalyst for more challenging substrates like internal olefins or functionalized alkenes. This modularity makes the technology a powerful tool for process development teams aiming to expand their portfolio of accessible chemical building blocks without investing in entirely new catalyst infrastructure.
How to Synthesize 3,4-bis(di-tert-butylphosphinomethyl)pyridine Efficiently
The synthesis of these high-performance ligands is designed to be straightforward and scalable, avoiding the use of exotic reagents or cryogenic conditions that are difficult to maintain in large reactors. The patent outlines a practical two-step sequence that begins with the activation of a dimethylpyridine precursor, followed by phosphorylation. This route is amenable to standard chemical manufacturing equipment, ensuring that the transition from laboratory bench to pilot plant is seamless. Detailed standardized synthesis steps are provided below to guide process engineers in implementing this technology.
- Preparation of the metalated pyridine intermediate via lithiation with n-BuLi or Grignard formation with Magnesium turnings in anhydrous solvents.
- Reaction of the metalated intermediate with di-tert-butylchlorophosphine at controlled temperatures (0°C to 160°C) to form the P-C bonds.
- Quenching, extraction, and recrystallization from methanol at low temperatures (-20°C to -30°C) to isolate the white crystalline ligand product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain directors, the adoption of this pyridine-based ligand technology offers tangible strategic benefits beyond mere technical performance. The primary advantage lies in the drastic simplification of the supply chain for catalyst components. Since the ligands are synthesized from readily available starting materials like lutidine and chlorodi-tert-butylphosphine, the risk of supply disruption due to raw material scarcity is significantly minimized. This reliability ensures continuous operation of downstream esterification plants, preventing costly shutdowns that can occur when specialized catalysts are unavailable. Additionally, the high stability of the ligands allows for longer storage life and easier transportation, reducing logistics costs and inventory write-offs.
- Cost Reduction in Manufacturing: The implementation of these ligands leads to substantial cost savings primarily through the enhancement of catalyst longevity and selectivity. With TON values surpassing 50,000, the consumption of expensive palladium metal per ton of product is drastically reduced, directly lowering the variable cost of production. Furthermore, the near-perfect linear selectivity eliminates the need for energy-intensive distillation columns required to separate branched isomers, resulting in lower utility costs and higher overall yield. The simplified synthesis of the ligand itself also contributes to a lower acquisition cost compared to complex ferrocene-based alternatives, providing a double-layered economic benefit.
- Enhanced Supply Chain Reliability: The robust nature of the synthetic route ensures that production lead times can be kept short and predictable. Unlike multi-step syntheses involving air-sensitive intermediates that require specialized handling, the described methods utilize standard Grignard or lithiation techniques that are well-understood in the fine chemical industry. This familiarity reduces the barrier to entry for contract manufacturing organizations, increasing the number of qualified suppliers capable of producing the ligand. Consequently, buyers gain leverage and security, knowing that the supply of this critical catalyst component is not bottlenecked by a single source or a fragile manufacturing process.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, the process generates minimal waste, aligning with increasingly stringent global emission standards. The patent explicitly notes that post-treatment is simple and the three-waste output is low, which simplifies the permitting process for new production facilities. Scalability is further supported by the use of common solvents like THF, octane, and methanol, which are easily recovered and recycled in industrial settings. This green chemistry profile not only reduces waste disposal costs but also enhances the corporate sustainability image of companies adopting this technology, a factor of growing importance for end-customers in the automotive and consumer goods sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these pyridine-based ligands in industrial settings. The answers are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this technology for potential adoption in their manufacturing workflows.
Q: What represents the primary advantage of these pyridine ligands over traditional dtbpx?
A: The novel pyridine-based ligands demonstrate superior turnover numbers (TON > 50000) and maintain linear-to-branched selectivity ratios exceeding 99:1, significantly outperforming many conventional aryl-bridged diphosphines in ethylene alkoxycarbonylation.
Q: Are these ligands suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly highlights that the synthesis involves simple post-treatment methods and generates minimal three-waste emissions, making the process highly scalable and environmentally compliant for commercial production.
Q: Can these ligands be applied to substrates other than ethylene?
A: Absolutely. While optimized for ethylene, the technology is applicable to terminal olefins with 2 to 10 carbon atoms, including propylene and various butenes, for hydroformylation and hydroaminomethylation reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridine-Based Phosphine Ligand Supplier
As the demand for high-efficiency catalysts continues to grow, partnering with an experienced CDMO becomes essential for securing a competitive edge. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from pilot testing to full-scale manufacturing is smooth and risk-free. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of ligand delivered meets the exacting standards required for sensitive palladium-catalyzed reactions. We understand the critical nature of catalyst consistency and are committed to providing a supply chain that supports your long-term production goals.
We invite you to contact our technical procurement team to discuss how these advanced ligands can optimize your specific process. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to this superior catalytic system. We are ready to provide specific COA data and route feasibility assessments tailored to your project needs, helping you make informed decisions that drive efficiency and profitability in your chemical manufacturing operations.
