Scalable Manufacturing of 4-Boc-Aminopiperidine via Novel Catalytic Hydrogenation Route
Scalable Manufacturing of 4-Boc-Aminopiperidine via Novel Catalytic Hydrogenation Route
The pharmaceutical industry continuously demands more efficient and safer synthetic routes for critical heterocyclic intermediates, particularly those serving as scaffolds for muscarinic M3 receptor antagonists. Patent CN107805218B introduces a transformative methodology for the preparation of 4-Boc-aminopiperidine, a pivotal building block for treating respiratory diseases such as chronic obstructive pulmonary disease (COPD) and asthma, as well as urinary system disorders. This technical disclosure outlines a streamlined two-step process that bypasses the limitations of traditional multi-step syntheses, offering a robust pathway for high-purity API intermediate production. By leveraging acid-catalyzed ketalization followed by direct catalytic hydrogenation, this innovation addresses key pain points in process chemistry, including safety hazards, low yields, and complex purification requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-Boc-aminopiperidine has been plagued by inefficient methodologies that hinder industrial scalability and economic viability. One prevalent conventional route involves the reaction of 1-benzyl-4-piperidone with hydroxylamine hydrochloride to form an oxime, followed by a hazardous reduction step using lithium aluminum hydride or metallic sodium. This approach not only entails a lengthy reaction sequence but also introduces severe safety risks due to the use of pyrophoric reagents, making it unsuitable for large-scale commercial operations. Another existing method utilizes 1-benzyl piperidine-4-formhydrazide, which suffers from a critically low yield of only 43 percent in the initial reduction step, drastically impacting overall process economics. Furthermore, routes starting directly from 4-aminopiperidine often require expensive protected starting materials and complex benzylation sequences, creating supply chain bottlenecks and inflating raw material costs significantly.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the novel approach detailed in the patent utilizes N-benzyl-4-piperidone as a cost-effective starting material, reacting it with orthoformate in an alcohol solution under acid catalysis to form a ketal intermediate. This intermediate subsequently reacts with tert-butyl carbamate to generate an imine species in a highly efficient manner, driven by the continuous removal of alcohol byproducts to shift the chemical equilibrium towards the product. The second stage involves a clean Pd/C catalytic hydrogenation reduction of the imine, which simultaneously achieves deprotection and saturation to yield the final 4-Boc-aminopiperidine. This strategy effectively condenses the synthesis into just two operational steps, eliminating the need for dangerous metal hydrides and significantly shortening the production cycle while maintaining exceptional product quality.
Mechanistic Insights into Acid-Catalyzed Imine Formation and Hydrogenation
The core of this synthetic breakthrough lies in the precise manipulation of reaction equilibria during the formation of the imine intermediate. In the first step, the acid catalyst, typically ammonium chloride or p-toluenesulfonic acid, facilitates the reaction between N-benzyl-4-piperidone and tri-orthoformate to generate a ketal. This ketal then undergoes a condensation reaction with tert-butyl carbamate. Crucially, the process design incorporates the distillation of the alcohol generated during this transformation, which thermodynamically drives the reaction forward to completion, ensuring that residual raw materials are kept below 1 percent as monitored by gas chromatography. This mechanistic control prevents the accumulation of unreacted ketones or carbamates, thereby simplifying the downstream workup and enhancing the overall mass balance of the process.
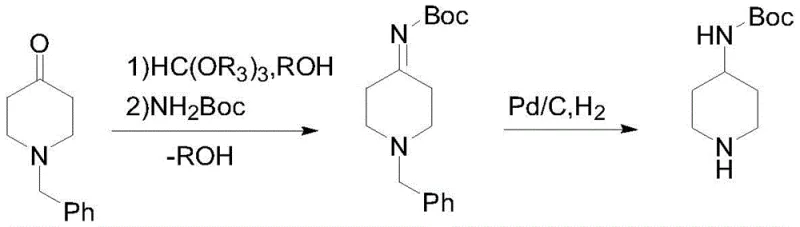
Following the formation of the imine, the second mechanistic phase employs heterogeneous catalysis using Palladium on Carbon (Pd/C) under pressurized hydrogen conditions. The reaction is conducted at elevated temperatures ranging from 60°C to 80°C and pressures between 0.8 MPa and 1.0 MPa. Under these conditions, the catalyst facilitates the hydrogenolysis of the benzyl group while simultaneously reducing the carbon-nitrogen double bond of the imine. This dual-function transformation is highly selective, avoiding over-reduction or side reactions that could compromise the integrity of the piperidine ring. The result is a crude product that requires minimal purification, often needing only a simple recrystallization or pulping step with n-heptane to achieve a GC content exceeding 99 percent, demonstrating superior impurity control compared to older reduction methods.
How to Synthesize 4-Boc-Aminopiperidine Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters to maximize yield and safety. The process begins with the reflux of N-benzyl-4-piperidone with orthoformate and an acid catalyst in methanol or ethanol, followed by the addition of tert-butyl carbamate and toluene to facilitate the azeotropic removal of alcohol. Once the imine intermediate is isolated, it is subjected to hydrogenation in an autoclave using 5 percent or 10 percent Pd/C. The detailed standardized operating procedures, including specific molar ratios, temperature ramps, and safety protocols for handling pressurized hydrogen, are critical for successful technology transfer. For the complete step-by-step technical guide and standardized synthesis protocol, please refer to the structured data section below.
- React N-benzyl-4-piperidone with orthoformate in alcohol under acid catalysis to form a ketal intermediate, then react with tert-butyl carbamate to generate the imine.
- Perform Pd/C catalytic hydrogenation reduction on the imine intermediate under pressurized conditions (0.8-1.0 MPa) at 60-80°C.
- Purify the final product via filtration and crystallization to achieve high purity 4-Boc-aminopiperidine suitable for API synthesis.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial strategic benefits for procurement managers and supply chain directors seeking to optimize their API intermediate sourcing. By replacing expensive and hazardous reagents with commodity chemicals like orthoformates and alcohols, the process inherently lowers the bill of materials. The elimination of lithium aluminum hydride not only reduces raw material costs but also removes the need for specialized quenching and waste disposal infrastructure, leading to significant operational expenditure savings. Furthermore, the shortened synthetic route directly translates to reduced manufacturing lead times, allowing for faster response to market demand fluctuations and improved inventory turnover rates for critical respiratory disease medications.
- Cost Reduction in Manufacturing: The utilization of N-benzyl-4-piperidone as a starting material represents a major cost advantage, as it is significantly cheaper and more readily available than protected 4-aminopiperidine derivatives used in alternative routes. The avoidance of stoichiometric metal reducing agents further decreases reagent costs and waste treatment expenses. Additionally, the high reaction yields observed in the patent examples indicate a highly atom-economical process, minimizing raw material waste and maximizing the output per batch, which collectively drives down the unit cost of production for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on common, bulk-available reagents such as methanol, ethanol, and tert-butyl carbamate ensures a resilient supply chain that is less susceptible to disruptions caused by specialty chemical shortages. The robustness of the Pd/C hydrogenation step, which operates under standard industrial pressure and temperature ranges, allows for flexible manufacturing across multiple facilities without requiring bespoke equipment. This flexibility enhances supply continuity, ensuring that downstream API manufacturers can maintain consistent production schedules for essential medicines treating conditions like irritable bowel syndrome and urinary incontinence.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing straightforward unit operations like reflux, distillation, and filtration that are easily adapted from pilot plant to commercial scale. The absence of heavy metal waste streams associated with traditional reducing agents simplifies environmental compliance and reduces the regulatory burden on manufacturing sites. Moreover, the high purity of the crude product reduces the solvent consumption required for purification, aligning with green chemistry principles and supporting corporate sustainability goals in fine chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process feasibility and product quality. Understanding these details is essential for technical teams evaluating the adoption of this route for commercial production of respiratory and urological drug intermediates.
Q: What are the safety advantages of this new synthesis method compared to traditional routes?
A: Unlike conventional methods that utilize hazardous reducing agents like lithium aluminum hydride or metallic sodium, this patented process employs catalytic hydrogenation with Pd/C. This eliminates significant safety risks associated with pyrophoric reagents and simplifies waste treatment protocols.
Q: How does this route improve yield and purity for industrial scale-up?
A: The process achieves high reaction yields (over 80% in the first step and over 90% in the second step in examples) by driving the equilibrium through the distillation of alcohol byproducts. Furthermore, the final product demonstrates high GC content (>99%), reducing the burden on downstream purification.
Q: Is the starting material N-benzyl-4-piperidone readily available for large-scale production?
A: Yes, N-benzyl-4-piperidone is a common and inexpensive reagent compared to protected aminopiperidines used in other routes. Its availability ensures a stable supply chain and significantly lowers the raw material costs for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Boc-Aminopiperidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic routes play in the global pharmaceutical supply chain. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the CN107805218B process are translated into reliable commercial reality. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that verify every batch meets the highest international standards for API intermediates. We understand that consistency and purity are non-negotiable for your downstream synthesis, and our infrastructure is built to deliver exactly that.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this optimized route can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the manufacturing of complex pharmaceutical intermediates.
