Scalable Synthesis of 3,5-Dihydroxyalkylbenzene: A Technical Breakthrough for Commercial Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways for high-value intermediates, particularly those serving the burgeoning cannabinoid and antiviral sectors. Patent CN103058828A presents a significant technological advancement in the synthesis of 3,5-dihydroxyalkylbenzene derivatives, such as the critical intermediate Olivetol (3,5-dihydroxypentylbenzene). Unlike historical methods that relied on the inefficient degradation of lichen acids or the hazardous use of organolithium reagents, this invention introduces a温和 (mild) organozinc-mediated coupling strategy. By shifting the core carbon-carbon bond formation step to a safer organozinc protocol, the patent outlines a route that drastically improves operational safety and cost-efficiency. For R&D directors and procurement managers alike, this represents a pivotal shift from laboratory curiosities to commercially viable manufacturing processes. The methodology not only circumvents the extreme sensitivity associated with traditional Grignard or Lithium reagents but also offers flexible protecting group strategies that facilitate easier purification and higher overall purity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 3,5-dihydroxyalkylbenzenes has been plagued by significant technical and economic bottlenecks that hinder large-scale adoption. The earliest methods involved the degradation of usnic acid extracted from lichens, a process characterized by extremely low yields and complex isolation procedures that are entirely unsuitable for modern industrial demand. Furthermore, synthetic approaches developed in academic literature often depend heavily on benzene-free raw materials or utilize highly reactive organometallic species like organolithium reagents. These traditional synthetic pathways necessitate cryogenic conditions, strictly anhydrous environments, and specialized handling equipment due to the pyrophoric nature of the reagents. Such requirements translate into prohibitive capital expenditures and operational risks, making cost reduction in pharmaceutical intermediate manufacturing nearly impossible. Additionally, the liquid nature of many intermediates in these older routes complicates purification, often leading to broad impurity profiles that fail to meet the stringent specifications required for API synthesis.
The Novel Approach
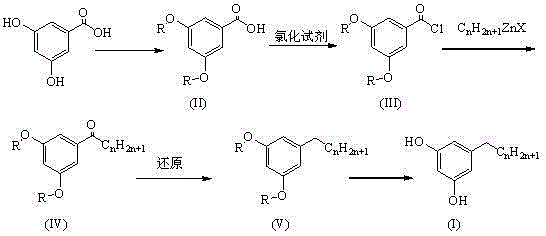
The methodology disclosed in CN103058828A fundamentally reengineers the synthesis by introducing a stable organozinc coupling step that operates under ambient or mild thermal conditions. Instead of forcing a reaction with volatile and dangerous lithium species, the process utilizes alkyl organic zinc reagents ($C_nH_{2n+1}ZnX$) which exhibit superior functional group tolerance and safety characteristics. This novel approach allows the reaction to proceed in non-polar solvents like cyclohexane or toluene, eliminating the need for expensive ether-based solvent systems that pose peroxide formation risks. Moreover, the strategic selection of protecting groups—ranging from simple methyl ethers to benzyl ethers—provides a tunable mechanism for optimizing physical properties. For instance, the benzyl-protected intermediates exist as high-melting solids, enabling purification through recrystallization rather than difficult distillation. This shift from liquid to solid intermediates is a game-changer for quality control, ensuring that the final high-purity pharmaceutical intermediate meets the rigorous standards demanded by global regulatory bodies.
Mechanistic Insights into Organozinc-Mediated Acylation
The core innovation of this synthetic pathway lies in the nucleophilic acyl substitution facilitated by the organozinc reagent. In the third step of the sequence, the activated acyl chloride (III) reacts with the alkyl zinc halide in a non-polar medium. Mechanistically, the zinc-carbon bond is less polarized than the corresponding lithium-carbon bond, which reduces the basicity of the nucleophile while retaining sufficient nucleophilicity to attack the carbonyl carbon of the acid chloride. This subtle electronic difference is crucial because it minimizes side reactions such as enolization or over-addition, which are common pitfalls in ketone synthesis using more aggressive organometallics. The reaction proceeds smoothly at room temperature or with mild heating, generating the diaryl ketone (IV) with high chemoselectivity. Following this, the ketone moiety is subjected to reduction, typically via the Huang Minglong modification (hydrazine hydrate and base) or zinc amalgam reduction. These reduction protocols effectively convert the carbonyl group into a methylene bridge without affecting the aromatic ring or the protecting groups, preserving the structural integrity of the molecule throughout the transformation.
Impurity control is intrinsically built into the mechanistic design of this process, particularly through the choice of the protecting group 'R'. When 'R' is a benzyl group, the resulting intermediates possess crystalline properties that allow for the physical exclusion of impurities. During the synthesis, by-products such as unreacted starting materials or over-alkylated species remain in the mother liquor during recrystallization, thereby enriching the purity of the solid cake. Conversely, when methyl groups are used, the deprotection step utilizes a mixture of hydrochloric acid and pyridine, which efficiently cleaves the ether linkage under reflux conditions. The mechanistic understanding of these deprotection kinetics ensures that the final deprotection step does not induce rearrangement or degradation of the alkyl chain. This level of control over the reaction trajectory ensures that the final 3,5-dihydroxyalkylbenzene product is obtained with a clean impurity profile, significantly reducing the burden on downstream purification processes and enhancing the overall viability of the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 3,5-Dihydroxyalkylbenzene Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step framework for producing 3,5-dihydroxyalkylbenzene derivatives with high reproducibility. The process begins with the protection of 3,5-dihydroxybenzoic acid, followed by activation to the acid chloride, coupling with the organozinc species, reduction of the resulting ketone, and final deprotection. Each stage has been optimized to balance reaction rate with safety, utilizing common industrial reagents like thionyl chloride and zinc chloride. The detailed standardized synthesis steps below provide the specific operational parameters required to replicate these results in a pilot or production setting.
- Protect the 3,5-hydroxyl groups of 3,5-dihydroxybenzoic acid using alkyl or benzyl protecting groups to form compound (II).
- React the protected acid with a chlorinating agent like thionyl chloride to generate the corresponding acyl chloride (III).
- Couple the acyl chloride with an organozinc reagent (CnH2n+1ZnX) in a non-polar solvent to form the ketone intermediate (IV).
- Reduce the ketone functionality using Huang Minglong reduction or zinc amalgam methods to yield the protected alkylbenzene (V).
- Remove the protecting group via acid catalysis or Pd/C hydrogenation to obtain the final 3,5-dihydroxyalkylbenzene product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this organozinc-based methodology offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the dependency on exotic, high-cost reagents like 3-nonene-2-one or unstable organolithium compounds, manufacturers can source standard, commodity-grade chemicals that are readily available in the global market. This shift significantly mitigates the risk of supply disruptions caused by the limited availability of specialty reagents. Furthermore, the mild reaction conditions reduce the energy load on the manufacturing facility, as there is no longer a need for maintaining cryogenic temperatures or specialized inert atmosphere lines for extended periods. These operational efficiencies translate directly into a more competitive cost structure, allowing for better margin management in the final API production.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents creates a direct pathway for cost optimization without compromising quality. Organolithium reagents require specialized storage and handling infrastructure, whereas organozinc reagents can be generated in situ from widely available alkyl halides and zinc chloride. This substitution removes the premium pricing associated with high-risk chemicals and reduces the waste disposal costs linked to quenching reactive lithium species. Additionally, the ability to purify intermediates via recrystallization (in the benzyl route) reduces solvent consumption compared to repeated chromatographic or distillation steps, further driving down the variable costs per kilogram of production.
- Enhanced Supply Chain Reliability: Reliability is paramount in the pharmaceutical supply chain, and this process enhances stability by relying on robust, well-understood chemical transformations. The use of non-polar solvents like cyclohexane and toluene ensures that the process is compatible with standard stainless steel reactors found in most multipurpose chemical plants, removing the need for specialized glass-lined or Hastelloy equipment often required for corrosive or sensitive reactions. This compatibility means that production can be easily transferred between different manufacturing sites or scaled up rapidly to meet surging demand for cannabinoid precursors or antiviral intermediates without lengthy technology transfer delays.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the process aligns well with modern green chemistry principles. The avoidance of pyrophoric reagents significantly lowers the fire and explosion risk profile of the plant, leading to lower insurance premiums and simpler regulatory compliance. The deprotection steps, whether using acid/pyridine or catalytic hydrogenation, generate manageable waste streams that are easier to treat than the heavy metal sludge often associated with alternative reduction methods. The scalability is further evidenced by the patent's demonstration of high yields (over 50% overall) even with simple workup procedures, indicating that the process is robust enough to handle the variances inherent in multi-ton production campaigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These answers are derived directly from the experimental data and technical specifications provided in patent CN103058828A, ensuring that stakeholders have accurate information for decision-making. Understanding these nuances is critical for evaluating the fit of this technology within your existing manufacturing portfolio.
Q: Why is the organozinc method superior to traditional organolithium routes for this synthesis?
A: Traditional methods often rely on hazardous and expensive organolithium reagents which require strict anhydrous conditions and cryogenic temperatures. The organozinc approach described in patent CN103058828A operates under much milder conditions, significantly reducing safety risks and equipment costs while maintaining high yields.
Q: What are the typical yields achieved with this protective group strategy?
A: The patent demonstrates robust overall yields depending on the protecting group used. For instance, the methyl protection route achieved a total yield of 51.1%, while the benzyl protection route, which allows for recrystallization purification, achieved an improved total yield of 59.3% based on the starting 3,5-dihydroxybenzoic acid.
Q: How does this process address impurity control in pharmaceutical intermediates?
A: The process incorporates strategic purification points. Specifically, when using benzyl protecting groups, the intermediate ketone and reduced products are solids with high melting points that can be purified via recrystallization. This solid-state purification is far more effective at removing trace impurities compared to distilling oily intermediates found in other routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Dihydroxyalkylbenzene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex molecules like 3,5-dihydroxyalkylbenzene requires more than just a patent; it demands deep process engineering expertise. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific requirements of this organozinc chemistry, including the safe handling of zinc reagents and the precise temperature control needed for the reduction and deprotection steps. We maintain stringent purity specifications through our rigorous QC labs, ensuring that every batch of intermediate delivered meets the exacting standards required for downstream API synthesis. Our commitment to quality ensures that the impurity profiles discussed in the mechanistic section are tightly controlled, providing you with a reliable foundation for your drug development programs.
We invite you to leverage our technical capabilities to optimize your supply chain for cannabinoid and pharmaceutical intermediates. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our mastery of this patented synthesis can drive efficiency and reliability in your production schedule, securing your position in the competitive global market.
