Advanced Arylamine NH-Containing Tridentate Phosphorane Ligands for Efficient Chiral Synthesis
Advanced Arylamine NH-Containing Tridentate Phosphorane Ligands for Efficient Chiral Synthesis
The landscape of asymmetric catalysis is undergoing a transformative shift driven by the urgent need for sustainable and cost-effective manufacturing processes in the fine chemical industry. Patent CN113024615A introduces a groundbreaking class of tridentate phosphorane ligands containing an arylamine NH group, representing a significant leap forward in ligand design philosophy. Historically, the field was dominated by noble metal complexes such as Rhodium and Ruthenium, famously utilized in the industrial synthesis of L-DOPA as depicted in early catalytic milestones. 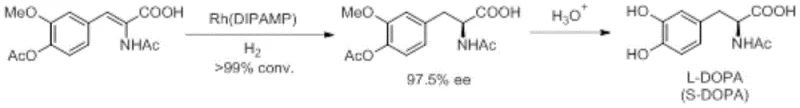 However, the reliance on scarce precious metals poses substantial supply chain risks and cost burdens for large-scale production. This new invention addresses these critical pain points by enabling the use of abundant base metals like Manganese, Iron, and Cobalt without compromising on enantioselectivity or conversion rates, thereby opening new avenues for the reliable pharmaceutical intermediate supplier seeking to optimize their production portfolios.
However, the reliance on scarce precious metals poses substantial supply chain risks and cost burdens for large-scale production. This new invention addresses these critical pain points by enabling the use of abundant base metals like Manganese, Iron, and Cobalt without compromising on enantioselectivity or conversion rates, thereby opening new avenues for the reliable pharmaceutical intermediate supplier seeking to optimize their production portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional asymmetric hydrogenation has long relied on bidentate ligands such as BINAP or Pybox derivatives, which, while effective, often suffer from limitations in substrate scope and catalyst stability. As illustrated by classical tridentate ligands like Pybox and indan-ambox, achieving high turnover numbers (TON) frequently requires precise steric tuning that can be synthetically cumbersome and expensive. 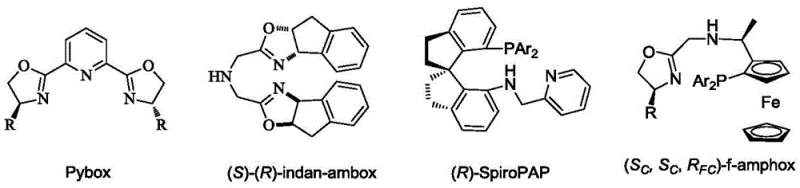 Furthermore, many existing systems struggle with bulky substrates or require harsh reaction conditions that degrade product purity. The electronic properties of standard bidentate ligands often fail to create a sufficiently deep chiral pocket, leading to lower enantiomeric excess (ee) values in complex molecular architectures. For procurement managers, this translates to higher costs associated with recycling expensive metals and additional downstream purification steps to remove trace impurities, complicating the commercial scale-up of complex polymer additives or API intermediates.
Furthermore, many existing systems struggle with bulky substrates or require harsh reaction conditions that degrade product purity. The electronic properties of standard bidentate ligands often fail to create a sufficiently deep chiral pocket, leading to lower enantiomeric excess (ee) values in complex molecular architectures. For procurement managers, this translates to higher costs associated with recycling expensive metals and additional downstream purification steps to remove trace impurities, complicating the commercial scale-up of complex polymer additives or API intermediates.
The Novel Approach
The novel approach disclosed in the patent utilizes a unique tridentate architecture incorporating an arylamine NH moiety alongside a chiral ferrocene backbone and a phosphine group. This specific structural motif, represented by general formulas (I) to (III), creates a robust coordination environment that stabilizes base metal centers effectively. 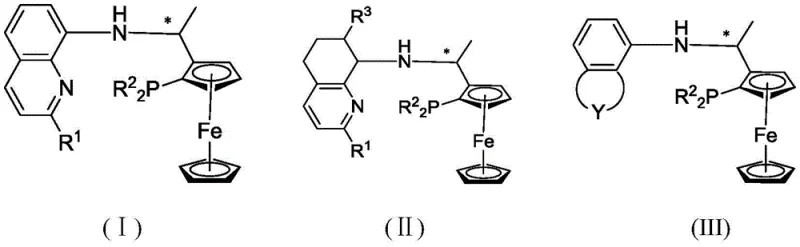 The presence of the NH group is not merely structural; it actively participates in the catalytic cycle through hydrogen bonding interactions with the substrate, a feature absent in many conventional phosphine-only ligands. This design allows for exceptional modifiability, where substituents on the aromatic rings can be tuned to adjust electronic and steric properties for specific reactions. Consequently, this innovation offers a versatile platform for cost reduction in API manufacturing by replacing precious metals with earth-abundant alternatives while maintaining rigorous quality standards required by global regulatory bodies.
The presence of the NH group is not merely structural; it actively participates in the catalytic cycle through hydrogen bonding interactions with the substrate, a feature absent in many conventional phosphine-only ligands. This design allows for exceptional modifiability, where substituents on the aromatic rings can be tuned to adjust electronic and steric properties for specific reactions. Consequently, this innovation offers a versatile platform for cost reduction in API manufacturing by replacing precious metals with earth-abundant alternatives while maintaining rigorous quality standards required by global regulatory bodies.
Mechanistic Insights into Mn-PNN Catalyzed Asymmetric Hydrogenation
The mechanistic superiority of this ligand system lies in its ability to facilitate a double functionalization mechanism during the catalytic cycle. The hydrogen atom in the arylamine NH group possesses enhanced acidity due to the coordination of the amino nitrogen atom to the metal center, making it highly electropositive. This electropositive hydrogen works in concert with the hydride anion generated at the metal center to activate polar double bonds in ketones or imines. The formation of the active catalyst species, such as the Mn-PNN complex shown below, involves the coordination of the tridentate ligand to the manganese center, displacing carbonyl ligands to create an open coordination site for substrate binding. 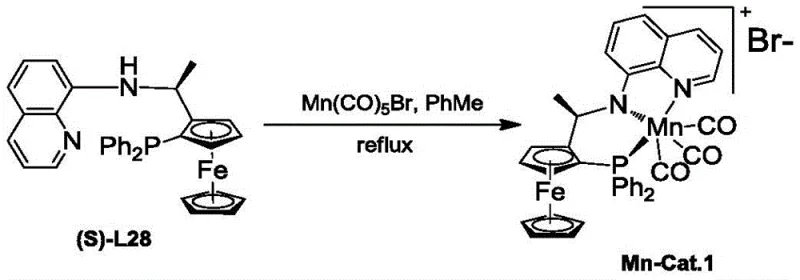 This cooperative activation lowers the energy barrier for the rate-determining step, resulting in high catalytic activity even at moderate temperatures and pressures. For R&D directors, understanding this mechanism is crucial for troubleshooting reaction parameters and optimizing conditions for new substrates, ensuring that the process remains robust against variations in raw material quality.
This cooperative activation lowers the energy barrier for the rate-determining step, resulting in high catalytic activity even at moderate temperatures and pressures. For R&D directors, understanding this mechanism is crucial for troubleshooting reaction parameters and optimizing conditions for new substrates, ensuring that the process remains robust against variations in raw material quality.
Impurity control is inherently built into the design of these ligands due to their high stereoselectivity and the stability of the resulting metal complexes. The rigid ferrocene backbone prevents ligand dissociation under reaction conditions, which minimizes the formation of racemic byproducts often caused by free metal species. Additionally, the specific geometry of the tridentate pocket excludes unwanted transition states that lead to side reactions. The patent data indicates that reactions can proceed with greater than 99% conversion and high enantiomeric excess, significantly reducing the burden on downstream chromatography. This level of control is essential for producing high-purity OLED material or pharmaceutical intermediates where trace impurities can have detrimental effects on final product performance or safety profiles.
How to Synthesize (S)-L28 Ligand Efficiently
The synthesis of the core ligand structures, such as the representative (S)-L28, follows a streamlined pathway that is amenable to large-scale production. The process begins with the lithiation of a chiral ferrocene derivative followed by phosphorylation, and concludes with a condensation reaction with an amino-quinoline derivative. This route avoids the use of exotic reagents and relies on standard organic transformations that are well-understood in industrial settings. The detailed standardized synthesis steps for preparing the ligand and its subsequent metal complex are provided in the guide below, ensuring reproducibility and consistency across different manufacturing batches.
- Mix the tridentate arylamine NH-containing ligand with Mn(CO)5Br in degassed anhydrous toluene under nitrogen protection at room temperature.
- Heat the mixture to reflux for 12 to 16 hours to ensure complete coordination of the manganese center with the PNN ligand backbone.
- Concentrate the solution under reduced pressure and precipitate the orange solid catalyst powder by adding n-hexane, then filter and wash.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain stakeholders, the adoption of this technology represents a strategic opportunity to de-risk operations and enhance margin profiles. By shifting from noble metals like Rhodium and Iridium to Manganese and Iron, companies can insulate themselves from the volatile pricing and geopolitical supply constraints associated with precious metals. The simplified synthetic route for the ligands themselves, utilizing readily available starting materials, further contributes to a more stable and predictable supply chain. This transition supports the goal of reducing lead time for high-purity pharmaceutical intermediates by eliminating bottlenecks related to catalyst availability and long procurement cycles for specialized reagents.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with inexpensive base metal complexes leads to substantial cost savings in raw material expenditures. Since the ligand enables high turnover numbers with cheap metals, the overall catalyst loading can be optimized to minimize cost per kilogram of product. Furthermore, the high selectivity reduces the need for extensive purification processes, lowering solvent consumption and waste disposal costs. These efficiencies compound over large production volumes, delivering significant economic advantages without sacrificing product quality or yield.
- Enhanced Supply Chain Reliability: Utilizing earth-abundant metals like Manganese ensures a consistent and secure supply of catalytic materials, unaffected by the mining constraints that plague platinum group metals. The ligand library is highly modular, allowing for rapid adaptation if specific raw materials face temporary shortages. This flexibility ensures continuous production schedules and reliable delivery to customers, reinforcing the company's reputation as a dependable partner in the global chemical market.
- Scalability and Environmental Compliance: The catalytic systems described operate under relatively mild conditions, often requiring lower pressures and temperatures than traditional methods, which reduces energy consumption. The high atom economy and selectivity minimize the generation of hazardous waste, aligning with increasingly stringent environmental regulations. This green chemistry profile facilitates easier permitting for new manufacturing facilities and supports corporate sustainability goals, making the technology attractive for long-term investment and commercial scale-up of complex fine chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel ligand technology. These answers are derived directly from the experimental data and theoretical framework presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this chemistry into existing production lines.
Q: What is the key advantage of the arylamine NH group in this ligand design?
A: The introduction of the arylamine NH group enables a double functionalization mechanism. The acidic hydrogen in the NH group facilitates combination with polar double bonds (N/O) in substrates, significantly improving catalyst-substrate interaction, catalytic activity, and stereoselectivity compared to traditional ligands.
Q: Can these ligands effectively replace expensive noble metal catalysts?
A: Yes, the patent demonstrates that complexes formed with cheap metals like Manganese (Mn), Iron (Fe), and Cobalt (Co) exhibit excellent selectivity and high catalytic activity in asymmetric hydrogenation, comparable to traditional Rhodium or Ruthenium systems, thereby drastically reducing raw material costs.
Q: Which pharmaceutical intermediates can be synthesized using this technology?
A: This catalytic system is applicable for synthesizing key intermediates for major drugs including Duloxetine, Aprepitant, Ezetimibe, Dapoxetine, Rivastigmine, Atomoxetine, Eslicarbazepine, and Crizotinib, covering a wide range of therapeutic areas.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arylamine NH Ligand Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting these advanced catalytic technologies to serve the global pharmaceutical and fine chemical sectors. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of ligand or catalyst meets the highest international standards. Our commitment to quality ensures that your downstream processes remain efficient and compliant with all regulatory requirements.
We invite you to collaborate with us to explore how this innovative ligand technology can transform your manufacturing economics. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific product portfolio. We are ready to provide specific COA data and route feasibility assessments to demonstrate the tangible benefits of switching to these high-performance, cost-effective catalytic systems for your next project.
