Advanced Manufacturing of N,N-Diethyl-m-acetamidoaniline via Closed-Loop Byproduct Recovery
Introduction to Sustainable Dye Intermediate Manufacturing
The global demand for high-performance dye intermediates necessitates a paradigm shift from linear consumption models to circular chemical economies. Patent CN101723845A introduces a groundbreaking methodology for the production of N,N-diethyl-m-acetamidoaniline, a critical precursor in the synthesis of advanced azo dyes and functional pigments. This technology addresses the persistent challenges of waste management and raw material efficiency that have long plagued the fine chemical sector. By replacing conventional hydrochloric acid pathways with a hydrobromic acid-mediated cycle, the process transforms what was once hazardous waste into valuable feedstock. For R&D directors and supply chain leaders, this represents a significant opportunity to enhance process sustainability while securing a more resilient supply chain for essential aromatic amine derivatives.
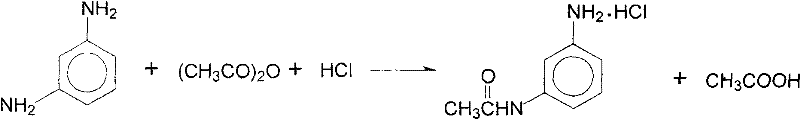
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N,N-diethyl-m-acetamidoaniline has relied on acylation using acetic anhydride followed by salt formation with hydrogen chloride. As illustrated in the reaction scheme above, this traditional pathway generates a complex mixture of inorganic salts, specifically sodium chloride and sodium bromide, during the subsequent ethylation stage. The presence of these mixed salts creates a severe downstream processing bottleneck, as separating them requires energy-intensive purification steps that offer little economic return. Furthermore, the reliance on acetic anhydride introduces higher raw material costs compared to simpler carboxylic acids, and the disposal of mixed halide waste streams imposes a heavy environmental burden and regulatory compliance cost on manufacturers.
The Novel Approach
The innovative process described in the patent fundamentally re-engineers the synthetic route by utilizing glacial acetic acid and hydrobromic acid for the initial acylation step. This strategic substitution ensures that the resulting inorganic byproduct is predominantly sodium bromide, rather than a difficult-to-separate mixture. The purity of the bromide stream is the key enabler for the subsequent recycling loop, where the waste mother liquor is not discarded but instead concentrated and chemically treated. This approach not only simplifies the purification of the target intermediate but also creates a closed-loop system where the byproducts are valorized, significantly reducing the overall ecological footprint and operational expenditure of the manufacturing facility.
Mechanistic Insights into HBr-Mediated Acylation and Ethylation
The core of this technological advancement lies in the precise control of the acylation and alkylation mechanisms. In the first stage, m-phenylenediamine reacts with glacial acetic acid in the presence of hydrobromic acid to form a mono-acylated intermediate stabilized as a hydrobromide salt. This salt formation is crucial as it protects the remaining amino group from unwanted side reactions while enhancing the solubility characteristics required for the next step. The reaction is typically conducted at elevated temperatures between 80°C and 99°C over a period of 20 to 40 hours, ensuring complete conversion of the diamine starting material while minimizing the formation of di-acylated impurities that could compromise the final dye quality.
Following acylation, the intermediate undergoes ethylation with ethyl bromide. The mechanism involves a nucleophilic substitution where the amine nitrogen attacks the ethyl group. Crucially, the process maintains a specific pH window between 2.0 and 7.0 during the addition of ethyl bromide to control the reaction kinetics and prevent over-alkylation or hydrolysis. The reaction generates sodium bromide as the stoichiometric byproduct. Unlike the conventional chloride route, this bromide stream is chemically versatile; it can be reacted with sulfuric acid and ethanol to regenerate ethyl bromide gas, which is then condensed and reused. This regeneration capability effectively decouples the production cost from the volatile market price of alkylating agents.
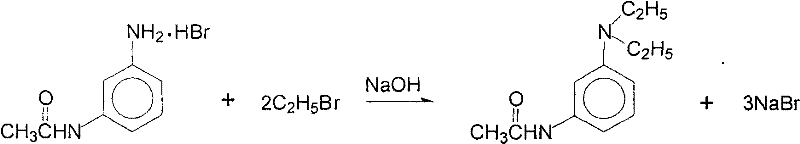
How to Synthesize N,N-Diethyl-m-acetamidoaniline Efficiently
The implementation of this synthesis route requires careful attention to the recycling loops that define its economic advantage. The process begins with the acylation of m-phenylenediamine, followed by the critical ethylation step where reaction conditions must be tightly monitored to ensure high conversion rates. The true innovation, however, is found in the post-reaction processing, where the mother liquor containing sodium bromide is concentrated and subjected to acid-catalyzed esterification-like conditions to regenerate ethyl bromide. This detailed operational sequence allows for the continuous reuse of reagents, transforming a batch process into a semi-continuous cycle of material efficiency. The detailed standardized synthesis steps for implementing this protocol are outlined in the guide below.
- Perform acylation of m-phenylenediamine using glacial acetic acid and hydrobromic acid at 80-99°C to form the acylated intermediate.
- Conduct ethylation reaction with ethyl bromide under controlled pH conditions (2.0-7.0) and temperature up to 70°C to yield the target product and sodium bromide mother liquor.
- Recycle the sodium bromide-rich mother liquor by reacting with sulfuric acid and ethanol to regenerate ethyl bromide and recover hydrogen bromide for reuse.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this recycling-centric methodology offers profound strategic benefits beyond simple regulatory compliance. The primary advantage is the drastic reduction in dependency on external suppliers for key alkylating agents like ethyl bromide, which are often subject to price volatility and strict transportation regulations due to their hazardous nature. By generating these reagents in-situ from waste streams, the manufacturing process insulates the supply chain from market fluctuations and logistical disruptions. Additionally, the substitution of acetic anhydride with glacial acetic acid represents a direct material cost saving, as the latter is a commodity chemical with a more stable and lower price point globally.
- Cost Reduction in Manufacturing: The elimination of expensive acetic anhydride in favor of glacial acetic acid immediately lowers the bill of materials. More significantly, the internal regeneration of ethyl bromide means that the facility purchases significantly less of this high-value reagent over time. The process converts a waste disposal cost into a raw material credit, effectively turning the effluent treatment plant into a secondary production unit that recovers value from what was previously considered trash, leading to substantial overall cost optimization.
- Enhanced Supply Chain Reliability: Relying on internal recycling loops reduces the frequency of inbound shipments for hazardous chemicals, thereby simplifying logistics and reducing the risk of supply interruptions caused by regulatory transport bans or supplier shortages. The ability to sustain production runs using recovered materials enhances the continuity of supply for downstream dye manufacturers. This self-sufficiency is particularly valuable in regions with stringent environmental laws where the disposal of bromide-containing waste is becoming increasingly restricted and costly.
- Scalability and Environmental Compliance: The process is inherently scalable as it relies on standard chemical engineering unit operations such as distillation and crystallization which are well-understood at the multi-ton scale. From an environmental perspective, the closed-loop nature of the bromide cycle minimizes the discharge of halogenated organic compounds and inorganic salts into wastewater systems. This aligns perfectly with modern green chemistry principles and facilitates easier permitting for capacity expansion, as the net environmental load per kilogram of product is significantly reduced compared to legacy technologies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this recycling technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and economic rationale for adopting this route. Understanding these details is essential for technical teams evaluating the transition from conventional linear synthesis to this circular manufacturing model.
Q: How does this process reduce environmental impact compared to traditional methods?
A: Traditional methods generate mixed salt waste (NaCl/NaBr) that is difficult to separate and dispose of. This novel process utilizes hydrobromic acid to generate a pure sodium bromide stream, which is chemically converted back into valuable ethyl bromide and hydrogen bromide, effectively closing the material loop and minimizing hazardous waste discharge.
Q: What are the key cost drivers eliminated in this synthesis route?
A: The process eliminates the need for expensive acetic anhydride by using glacial acetic acid. Furthermore, it drastically reduces the consumption of ethyl bromide, a costly alkylating agent, by regenerating it in-situ from the reaction byproducts, leading to substantial raw material cost savings.
Q: Is this method scalable for industrial production of dye intermediates?
A: Yes, the process is designed for scalability. It utilizes standard unit operations such as distillation, filtration, and crystallization. The recycling loops for solvents and reagents enhance process stability and make it highly suitable for large-scale commercial manufacturing of fine chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N-Diethyl-m-acetamidoaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the future of fine chemical manufacturing lies in the integration of high-yield synthesis with sustainable waste management. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex recycling loops like the one described in CN101723845A are implemented with precision and safety. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N,N-diethyl-m-acetamidoaniline meets the exacting standards required for high-performance dye applications, delivering consistency that your production lines depend on.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain resilience and reduce your total cost of ownership.
