Advanced Synthesis of Ritonavir Intermediate BDH via Stereoselective Reduction
The pharmaceutical industry continuously seeks robust synthetic routes for critical antiviral intermediates, particularly for protease inhibitors like ritonavir and lopinavir. Patent CN111393329A, published in July 2020, discloses a highly efficient preparation method for the key intermediate BDH, chemically known as (2S,3S,5S)-2-amino-3-hydroxy-5-tert-butoxycarboxamido-1,6-diphenylhexane. This specific intermediate serves as the central hydroxyethylene scaffold essential for the biological activity of these HIV protease inhibitors. The disclosed technology addresses long-standing challenges in stereochemical control and process scalability, offering a pathway that leverages chiral pool synthesis combined with advanced asymmetric reduction techniques. For R&D directors and process chemists, this patent represents a significant evolution from traditional resolution-based methods, promising higher optical purity and streamlined operations. By anchoring the synthesis on L-phenylalanine, the route inherently transfers chirality, reducing the burden on downstream purification and enhancing the overall economic viability of producing high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex peptidomimetic intermediates like BDH has been plagued by inefficient stereocontrol and excessive step counts. Prior art, such as the methods referenced in CN101967130A, often relied on resolving racemic mixtures or employing multiple protection-deprotection sequences that eroded overall yield. These conventional approaches frequently suffered from low diastereomer excess (de%) values, necessitating rigorous and costly chromatographic separations or repeated crystallizations to meet the stringent purity specifications required for API manufacturing. Furthermore, the use of harsh reaction conditions in older methodologies often led to epimerization at sensitive chiral centers, generating difficult-to-remove impurities that compromised the safety profile of the final drug substance. The accumulation of these inefficiencies resulted in prolonged production cycles and elevated waste generation, creating substantial bottlenecks for supply chain managers aiming to secure reliable volumes of clinical-grade material.
The Novel Approach
In stark contrast, the methodology outlined in CN111393329A introduces a streamlined, convergent strategy that maximizes atom economy and stereochemical fidelity from the outset. The process initiates with the alkylation of L-phenylalanine, effectively locking in the first chiral center before chain elongation occurs. 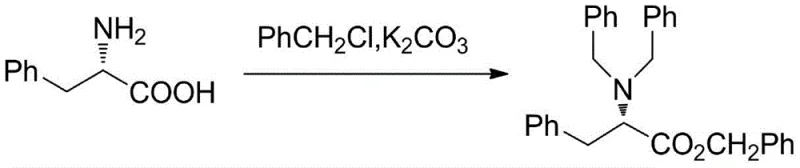 . This foundational step utilizes inexpensive and readily available benzyl chloride under mild alkaline conditions, avoiding the need for exotic reagents. The subsequent chain extension via acetonitrile condensation and Grignard addition builds the carbon backbone efficiently. Crucially, the novel approach integrates a sophisticated dual-reduction system using sodium borohydride modified with methanesulfonic and trifluoroacetic acids in the presence of a chiral CBS catalyst. This specific modification allows for the simultaneous reduction of enamine and carbonyl functionalities with exceptional stereocontrol, directly establishing the critical 3-hydroxy and 5-amino stereocenters in a single operational sequence, thereby drastically simplifying the synthetic tree.
. This foundational step utilizes inexpensive and readily available benzyl chloride under mild alkaline conditions, avoiding the need for exotic reagents. The subsequent chain extension via acetonitrile condensation and Grignard addition builds the carbon backbone efficiently. Crucially, the novel approach integrates a sophisticated dual-reduction system using sodium borohydride modified with methanesulfonic and trifluoroacetic acids in the presence of a chiral CBS catalyst. This specific modification allows for the simultaneous reduction of enamine and carbonyl functionalities with exceptional stereocontrol, directly establishing the critical 3-hydroxy and 5-amino stereocenters in a single operational sequence, thereby drastically simplifying the synthetic tree.
Mechanistic Insights into Stereoselective CBS Reduction
The cornerstone of this patented technology lies in the fourth synthetic step, where the stereoselective reduction of the enaminone intermediate dictates the final optical purity of the BDH molecule. The mechanism involves the activation of sodium borohydride by strong acids like methanesulfonic acid and trifluoroacetic acid, generating highly reactive borane species in situ. When coupled with the chiral inducer (S)-2-methyl-CBS-oxazaborolidine, the reduction proceeds through a rigid, organized transition state that favors the formation of the desired (3S,5S) configuration. 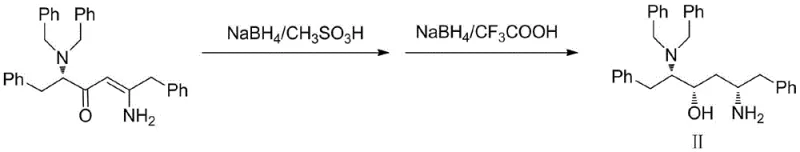 . This chiral environment effectively shields one face of the prochiral ketone and enamine moieties, directing hydride delivery to the opposite face with high precision. The patent data indicates that this specific catalytic system achieves a diastereomer excess (de%) value exceeding 98%, a metric that is critical for minimizing impurity loads in the final API. For process chemists, understanding this mechanistic nuance is vital, as it eliminates the need for classical resolution steps that typically discard 50% of the material, thereby theoretically doubling the potential yield from the chiral starting material.
. This chiral environment effectively shields one face of the prochiral ketone and enamine moieties, directing hydride delivery to the opposite face with high precision. The patent data indicates that this specific catalytic system achieves a diastereomer excess (de%) value exceeding 98%, a metric that is critical for minimizing impurity loads in the final API. For process chemists, understanding this mechanistic nuance is vital, as it eliminates the need for classical resolution steps that typically discard 50% of the material, thereby theoretically doubling the potential yield from the chiral starting material.
Furthermore, the impurity profile generated by this route is significantly cleaner compared to non-catalytic reductions. The use of specific acid modifiers prevents over-reduction or side reactions that could lead to deamination or dehydration byproducts. The subsequent protection step using di-tert-butyl dicarbonate (Boc2O) is highly selective for the primary amine generated in the previous step, leaving the secondary alcohol and tertiary amine untouched. This orthogonality ensures that the intermediate III is formed with high regioselectivity. Finally, the debenzylation step utilizes ammonium formate as a transfer hydrogenation source over a palladium on carbon catalyst. This mild hydrogenolytic condition cleaves the benzyl protecting groups without affecting the acid-labile Boc group or the sensitive hydroxyethylene core, ensuring the structural integrity of the final BDH intermediate is maintained throughout the rigorous deprotection process.
How to Synthesize BDH Efficiently
The synthesis of BDH described in this patent offers a reproducible framework for manufacturing high-purity antiviral intermediates. The process is designed to be scalable, utilizing common organic solvents like tetrahydrofuran, methyl tert-butyl ether, and methanol, which facilitates easy solvent recovery and recycling in a GMP environment. The operational parameters, such as temperature controls ranging from 0°C to 65°C, are well within the capabilities of standard industrial reactors, removing the need for cryogenic specialized equipment.
- React L-phenylalanine with benzyl chloride in alkaline solution to obtain N,N-dibenzylamino-L-benzyl phenylalanine.
- Condense the ester with acetonitrile using sodium amide to form the nitrile intermediate.
- Perform Grignard addition with benzyl magnesium chloride to extend the carbon chain.
- Execute stereoselective reduction using sodium borohydride/methanesulfonic acid and sodium borohydride/trifluoroacetic acid with a chiral inducer.
- Protect the amino group with di-tert-butyl dicarbonate (Boc2O) under basic conditions.
- Perform catalytic hydrogenolysis using ammonium formate and Pd/C to remove benzyl groups and yield BDH.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling strategic advantages centered around cost stability and supply continuity. The reliance on L-phenylalanine as the starting material is a significant logistical benefit, as this amino acid is a commodity chemical produced on a massive global scale, ensuring a stable and competitive pricing structure that is less susceptible to the volatility often seen with specialized chiral building blocks. By eliminating the need for expensive chiral resolution agents or complex enzymatic steps, the overall cost of goods sold (COGS) for the intermediate is substantially reduced. The high yield reported in the patent examples, particularly in the final debenzylation step which achieves over 90% conversion, translates directly into lower raw material consumption per kilogram of finished product. This efficiency gain allows suppliers to offer more competitive pricing tiers for long-term contracts, providing a buffer against market fluctuations in the pharmaceutical sector.
- Cost Reduction in Manufacturing: The elimination of resolution steps and the high stereoselectivity of the CBS reduction mean that less material is wasted during purification. Traditional routes often lose significant mass during recrystallization or chromatography to achieve the necessary optical purity; this novel method builds the correct stereochemistry directly into the molecule. Consequently, the demand for expensive solvents and silica gel for purification is drastically minimized. Additionally, the use of ammonium formate for hydrogenolysis avoids the need for high-pressure hydrogen gas infrastructure, reducing capital expenditure requirements for manufacturing facilities and lowering operational safety costs associated with handling compressed flammable gases.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more reliable supply chain. The reagents used, such as benzyl chloride, acetonitrile, and sodium borohydride, are widely available from multiple global vendors, mitigating the risk of single-source dependency. The process tolerance described in the patent suggests that minor variations in reaction parameters do not critically impact the quality of the intermediate, leading to higher batch success rates and fewer production delays. This reliability is crucial for maintaining the continuous flow of materials required for the just-in-time manufacturing schedules of major pharmaceutical companies, ensuring that API production lines remain operational without interruption due to intermediate shortages.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the route is designed for green chemistry principles. The avoidance of heavy metal catalysts in the early stages and the use of heterogeneous palladium catalysts in the final step simplify waste treatment protocols. The aqueous workups described, utilizing simple salts like potassium carbonate and sodium chloride, generate waste streams that are easier to treat compared to those containing complex organic bases or toxic heavy metals. This alignment with environmental regulations facilitates faster regulatory approvals for new manufacturing sites and reduces the long-term liability associated with hazardous waste disposal, making the process highly attractive for large-scale commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of BDH intermediates based on the patented technology. These insights are derived directly from the experimental data and beneficial effects claimed in the patent documentation, providing clarity on the feasibility and advantages of this specific synthetic pathway. Understanding these details helps stakeholders make informed decisions regarding vendor selection and process validation.
Q: How does this patent improve stereoselectivity compared to prior art?
A: The patent utilizes a chiral pool strategy starting from L-phenylalanine and employs (S)-2-methyl-CBS-oxazaborolidine as a chiral inducer during the reduction step, achieving a diastereomer excess (de%) value of over 98%, significantly higher than conventional resolution methods.
Q: What are the key raw materials for this BDH synthesis route?
A: The primary raw material is L-phenylalanine, which is reacted with benzyl chloride. Subsequent steps utilize acetonitrile, benzyl magnesium chloride, and standard protecting group reagents like di-tert-butyl dicarbonate, ensuring原料 availability and cost stability.
Q: Does this method involve hazardous heavy metal catalysts?
A: The final debenzylation step uses palladium on carbon (Pd/C) with ammonium formate as a hydrogen source. While palladium is a precious metal, it is used heterogeneously and can be recovered, minimizing heavy metal contamination risks in the final API intermediate compared to homogeneous transition metal catalysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable BDH Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against viral diseases. Our technical team has extensively analyzed the synthetic route disclosed in CN111393329A and possesses the expertise to implement this advanced stereoselective reduction technology at an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to confirm the >98% de% value essential for downstream API synthesis. We are committed to delivering BDH intermediates that meet the highest international standards for pharmaceutical manufacturing.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific production requirements. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this patented process can optimize your supply chain. By partnering with us, you gain access to a reliable source of complex pharmaceutical intermediates backed by deep chemical engineering expertise and a commitment to sustainable, cost-effective manufacturing solutions. Contact us today to discuss how we can support your antiviral drug development and production goals.
