Revolutionizing Anti-Infective Intermediate Production: A Deep Dive into High-Efficiency Hydantoin Synthesis
Revolutionizing Anti-Infective Intermediate Production: A Deep Dive into High-Efficiency Hydantoin Synthesis
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable pathways for the production of critical anti-infective agents. A pivotal advancement in this domain is detailed in Chinese Patent CN101792433B, which discloses a novel method for synthesizing 1-((3-(5-nitro-2-furyl)allylidene)amino)hydantoin. This compound, a potent nitrofuran derivative, serves as a crucial active pharmaceutical ingredient (API) or intermediate for treating urinary tract infections and other bacterial conditions. The patent highlights a transformative shift from traditional, multi-step, and hazardous synthetic routes to a streamlined, one-pot condensation strategy. By leveraging 5-nitrofuran acrylaldehyde, hydrazine hydrate, urea, and ethyl chloroacetate, this methodology not only simplifies the operational workflow but also drastically enhances atom economy. For R&D directors and procurement managers alike, understanding this technological leap is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials while adhering to stringent environmental regulations.
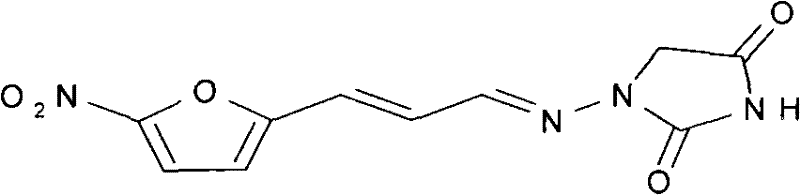
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-((3-(5-nitro-2-furyl)allylidene)amino)hydantoin has been plagued by inefficiencies that hinder large-scale commercial viability. Traditional routes often relied on the direct condensation of 5-nitrofuran acrolein with 1-aminohydantoin, as depicted in earlier literature. However, the preparation of the key 1-aminohydantoin intermediate itself presented significant bottlenecks. One common historical pathway involved reacting hydrazine hydrate with chloroacetic acid, followed by esterification and treatment with potassium cyanate. This multi-step sequence suffered from abysmal yields, typically ranging between 20% and 30%, rendering it economically unfeasible for modern mass production. Another route utilizing benzylidene semicarbazone and ethyl chloroacetate offered slightly better yields but introduced severe purity challenges, particularly during scale-up, where difficult-to-remove impurities compromised the final API quality. Furthermore, many legacy processes necessitated the use of toxic solvents like pyridine or chloroform, creating substantial environmental liabilities and increasing the cost of waste disposal and solvent recovery.
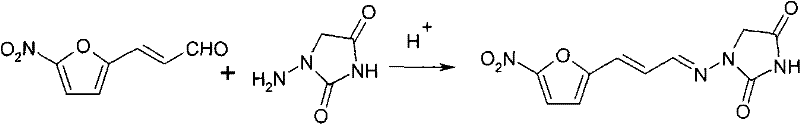
The Novel Approach
In stark contrast to these cumbersome legacy methods, the innovative process outlined in CN101792433B introduces a highly efficient, convergent synthesis strategy. Instead of pre-forming the unstable 1-aminohydantoin, this method generates a semicarbazone intermediate in situ. The process begins with a one-pot reaction between hydrazine hydrate and urea to form semicarbazide, which is then immediately condensed with 5-nitrofuran acrolein without isolation. This telescoped approach eliminates the need for intermediate purification steps such as acid precipitation, concentration, and crystallization, which are both time-consuming and yield-diminishing. The subsequent cyclization with ethyl chloroacetate proceeds under mild conditions in ethanol, a green solvent, avoiding the hazards associated with benzene or chloroform. This strategic reordering of synthetic steps not only boosts the overall yield by approximately 5 percentage points compared to traditional methods but also ensures a cleaner reaction profile, facilitating easier downstream processing and higher final product purity.
Mechanistic Insights into Aldehyde-Amine Condensation and Cyclocondensation
The core chemical innovation lies in the precise control of the aldehyde-amine condensation and the subsequent ring-closing cyclization. The mechanism initiates with the nucleophilic attack of hydrazine on urea to generate semicarbazide, which acts as a protected form of the hydrazine moiety. Upon acidification and reaction with the electron-deficient aldehyde group of 5-nitrofuran acrolein, a stable semicarbazone linkage is formed. This step is critical because it protects the active hydrogens of the hydrazine, preventing unwanted side reactions that often plague direct aminohydantoin synthesis. The second phase involves the nucleophilic substitution of the ethyl chloroacetate by the nitrogen atoms of the semicarbazone backbone. Under basic conditions provided by sodium ethoxide, the molecule undergoes intramolecular cyclization to form the hydantoin ring. This pathway exhibits superior atom economy because it minimizes the generation of stoichiometric by-products. By avoiding the harsh conditions required to deprotect or purify intermediates in older routes, the reaction maintains the integrity of the sensitive nitrofuran double bond, ensuring the biological activity of the final molecule remains uncompromised.

Furthermore, the impurity control mechanism inherent in this design is robust. In traditional syntheses, the presence of free hydrazine or unreacted chloroacetic acid often led to polymeric by-products or ring-opened impurities that were notoriously difficult to separate. The new method's use of a pre-formed semicarbazone intermediate effectively masks these reactive sites until the precise moment of cyclization. The choice of ethanol as the solvent further aids in impurity management; it dissolves the reactants effectively while allowing the final product to precipitate cleanly upon acidification. This crystallization-driven purification ensures that the final solid possesses high chemical purity, meeting the rigorous specifications required for pharmaceutical applications. For quality assurance teams, this translates to a more consistent impurity profile, reducing the risk of batch failures and ensuring regulatory compliance across different production runs.
How to Synthesize 1-((3-(5-Nitro-2-Furyl)Allylidene)Amino)Hydantoin Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize the benefits of the one-pot design. The process is divided into two main stages: the formation of the semicarbazone intermediate and the subsequent ring closure. The first stage involves refluxing hydrazine hydrate and urea, followed by controlled acidification and the addition of the aldehyde. Temperature control is vital here, with reflux temperatures around 100°C for the initial step and 80°C for the condensation. The second stage utilizes sodium ethoxide in ethanol to drive the cyclization with ethyl chloroacetate. Detailed operational parameters, including specific molar ratios and timing for reagent addition, are critical for reproducibility. For a comprehensive guide on executing this protocol in a GMP environment, please refer to the standardized synthesis steps below.
- Prepare 5-nitrofuran acrolein semicarbazone via one-pot condensation of hydrazine hydrate, urea, and 5-nitrofuran acrolein.
- Execute condensation ring-closing reaction using ethyl chloroacetate and sodium ethoxide in ethanol solvent.
- Acidify the reaction mixture to precipitate the final high-purity hydantoin product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic advantages beyond mere chemical elegance. The primary benefit is the drastic simplification of the supply chain for raw materials. By utilizing commodity chemicals like hydrazine hydrate, urea, and ethyl chloroacetate, manufacturers can avoid reliance on specialized, expensive, or hard-to-source intermediates like 1-aminohydantoin or nitrofurantoin. This diversification of the raw material base significantly enhances supply chain resilience, mitigating the risk of production stoppages due to single-source supplier failures. Moreover, the elimination of toxic solvents such as chloroform and benzene reduces the regulatory burden and costs associated with hazardous waste disposal. This aligns perfectly with global trends towards green chemistry, potentially lowering insurance premiums and facilitating smoother audits by environmental agencies.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven primarily by the reduction in unit operations. By telescoping the semicarbazone formation and condensation into a single pot without intermediate isolation, the process saves significant energy, labor, and equipment time. The removal of solvent recovery steps for toxic chemicals further lowers operational expenditures. Additionally, the reported yield improvement, while seemingly modest in percentage terms, translates to substantial material savings when scaled to metric tonnage, directly improving the gross margin for the final API. The use of ethanol, a relatively inexpensive and recyclable solvent, compared to pricier specialty solvents, also contributes to a lower cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent delivery schedules, a critical factor for downstream pharmaceutical formulators. Traditional methods often suffered from batch-to-batch variability due to the sensitivity of the 1-aminohydantoin intermediate. The new method's stable semicarbazone intermediate and straightforward workup procedure minimize the risk of batch failures. This reliability allows supply chain planners to maintain leaner inventory levels without the fear of stockouts. Furthermore, the scalability of the reaction, demonstrated in examples up to 2000L reactors, confirms that the process can seamlessly transition from pilot plant to full commercial production without requiring exotic equipment like reactive distillation columns.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this process represents a significant upgrade. The avoidance of carcinogenic solvents creates a safer working environment for plant operators, reducing liability risks. The aqueous workup and ethanol-based system generate waste streams that are easier to treat biologically compared to halogenated waste. This environmental friendliness is increasingly becoming a prerequisite for vendor qualification by major multinational pharmaceutical companies. The ability to produce high-purity material with less waste positions the manufacturer as a preferred partner for sustainability-focused initiatives, opening doors to long-term contracts with top-tier global clients who prioritize green supply chains.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common concerns regarding yield optimization, safety, and scalability. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. We encourage our partners to review these insights to fully appreciate the operational improvements this technology offers over legacy manufacturing methods.
Q: How does the new synthesis method improve yield compared to traditional routes?
A: The patented method eliminates intermediate isolation steps and utilizes a one-pot semicarbazone formation, resulting in a yield increase of approximately 5 percentage points over conventional processes, reaching up to 90% in the first step.
Q: What are the environmental benefits of this manufacturing process?
A: This process avoids the use of carcinogenic solvents like chloroform and benzene, replacing them with ethanol and water, significantly reducing hazardous waste generation and improving workplace safety.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method uses readily available raw materials and standard reaction conditions (reflux at 80-100°C), making it highly scalable without the need for complex reactive distillation equipment required by older nitrofurantoin-based routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-((3-(5-Nitro-2-Furyl)Allylidene)Amino)Hydantoin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires more than just a patent; it demands engineering excellence and unwavering commitment to quality. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific thermal and mixing requirements of this condensation-cyclization sequence, ensuring that the high yields reported in the lab are faithfully reproduced on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1-((3-(5-nitro-2-furyl)allylidene)amino)hydantoin meets the highest international pharmacopoeia standards, providing you with a secure and compliant supply of this critical anti-infective intermediate.
We invite you to collaborate with us to leverage this advanced synthesis technology for your product pipeline. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this greener route can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain with a partner who understands both the chemistry and the commerce of pharmaceutical manufacturing.
