Scalable Synthesis of Novel Indolo Delta-Sultone Compounds for Advanced Drug Discovery
Scalable Synthesis of Novel Indolo Delta-Sultone Compounds for Advanced Drug Discovery
The pharmaceutical industry is constantly seeking novel heterocyclic scaffolds that can serve as potent pharmacophores for next-generation therapeutics. A recent breakthrough detailed in patent CN116217587A introduces a robust and efficient methodology for synthesizing indolo delta-sultone compounds, a unique class of fused heterocycles combining the biological relevance of the indole ring with the metabolic stability of the sultone motif. This innovation addresses a critical gap in medicinal chemistry by providing a straightforward route to access these complex structures, which have shown promising antitumor activity in preliminary screenings against HepG2 and Caco-2 cell lines. The significance of this technology lies not only in the biological potential of the resulting molecules but also in the exceptional operational simplicity of the synthetic protocol, which operates under mild conditions without the need for expensive transition metal catalysts or extreme temperatures.
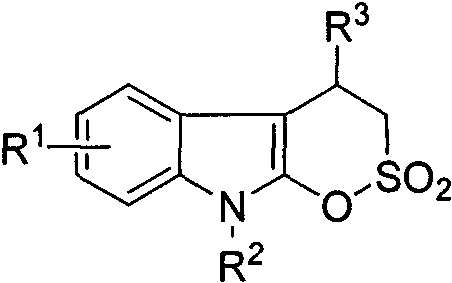
For research directors and process chemists, the ability to rapidly generate diverse libraries of such compounds is paramount for structure-activity relationship (SAR) studies. The general structure disclosed allows for extensive derivatization at the nitrogen atom and the aromatic rings, enabling the fine-tuning of physicochemical properties such as solubility and metabolic stability. By leveraging this patented technology, organizations can accelerate their hit-to-lead campaigns, potentially identifying new candidates for oncology treatments much faster than with conventional synthetic approaches. The fusion of the indole and sultone rings creates a rigid three-dimensional architecture that may offer superior binding affinity to biological targets compared to flexible linear analogues, representing a strategic advantage in modern drug design.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of sultone-fused heterocyclic systems has been fraught with synthetic challenges that hinder their widespread adoption in industrial settings. Traditional pathways often rely on the use of highly reactive and corrosive sulfonating agents like chlorosulfonic acid, which necessitate specialized equipment and rigorous safety protocols to manage the exothermic nature of the reactions and the hazardous byproducts generated. Furthermore, classical methods frequently involve multi-step sequences requiring the installation and subsequent removal of protecting groups to prevent side reactions at sensitive functional sites, leading to reduced overall atom economy and increased waste disposal costs. These legacy processes often suffer from poor regioselectivity, resulting in complex mixtures of isomers that are difficult and expensive to separate, thereby compromising the purity profile required for pharmaceutical applications. Additionally, many existing protocols demand high temperatures or prolonged reaction times, which can lead to the thermal degradation of sensitive substrates and limit the scope of compatible functional groups.
The Novel Approach
In stark contrast to these cumbersome traditional techniques, the method disclosed in CN116217587A utilizes a remarkably elegant tandem cyclization strategy that proceeds efficiently at ambient temperature. By employing indolin-2-one and beta-aryl vinyl sulfonyl fluoride as readily available starting materials, the process bypasses the need for harsh reagents and complex protection-deprotection sequences. The reaction is driven by a mild organic base system, specifically utilizing DBU in combination with sodium bicarbonate, which facilitates the cascade transformation with high precision. This approach not only simplifies the operational workflow by condensing multiple bond-forming events into a single pot but also significantly enhances the safety profile of the manufacturing process. The compatibility of this method with a wide range of substituents, including halogens, nitro groups, and various alkyl chains, demonstrates its versatility and robustness, making it an ideal candidate for the rapid synthesis of diverse compound libraries for biological evaluation.
Mechanistic Insights into Base-Catalyzed Tandem Cyclization
The success of this synthetic transformation hinges on the precise interplay between the nucleophilicity of the indolin-2-one derivative and the electrophilicity of the vinyl sulfonyl fluoride. The mechanism likely initiates with the deprotonation of the indolin-2-one nitrogen or the alpha-carbon by the base, generating a reactive nucleophilic species. This species then undergoes a conjugate addition (Michael addition) to the electron-deficient double bond of the vinyl sulfonyl fluoride. The presence of the sulfonyl fluoride group is crucial, as it acts as a latent electrophile that can participate in the subsequent intramolecular cyclization step. The fluoride ion serves as an excellent leaving group in this context, allowing for the formation of the stable six-membered sultone ring through an intramolecular nucleophilic substitution. This cascade sequence is highly concerted, minimizing the accumulation of unstable intermediates and driving the reaction towards the thermodynamically favored fused ring system.
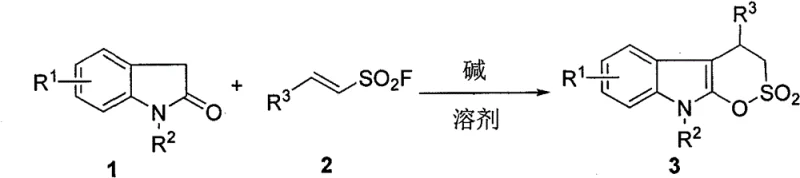
From an impurity control perspective, the choice of the DBU and sodium bicarbonate mixed base system is particularly ingenious. DBU is a strong, non-nucleophilic base that effectively promotes the initial deprotonation without competing in the nucleophilic attack, while sodium bicarbonate acts as a buffer to moderate the reaction environment. This buffering capacity helps to suppress potential side reactions such as the hydrolysis of the sulfonyl fluoride moiety or polymerization of the vinyl species, which are common pitfalls in similar transformations. The mild reaction temperature of 20-25°C further contributes to a clean impurity profile by preventing thermal decomposition pathways. Consequently, the crude reaction mixtures typically contain fewer byproducts, simplifying the downstream purification process and ensuring that the final active pharmaceutical ingredients meet stringent quality specifications with minimal effort.
How to Synthesize Indolo Delta-Sultone Efficiently
The practical implementation of this synthesis is designed for ease of execution, making it accessible for both laboratory-scale discovery and larger-scale production. The protocol requires standard glassware and does not demand inert atmosphere techniques, as the reagents are relatively stable under ambient conditions. The reaction proceeds rapidly, often reaching completion within 20 to 60 minutes, which allows for high-throughput experimentation and quick iteration of structural analogues. Following the reaction, the workup involves simple concentration and standard silica gel chromatography, avoiding the need for complex extraction procedures or specialized scavenging resins. For detailed standardized operating procedures and specific stoichiometric ratios optimized for different substrates, please refer to the step-by-step guide below.
- Combine indolin-2-one, beta-aryl vinyl sulfonyl fluoride, DBU (0.1-0.2 eq), and sodium bicarbonate (1.1-2.0 eq) in dichloromethane.
- Stir the reaction mixture at ambient temperature (20-25°C) for 20 to 60 minutes while monitoring progress via TLC.
- Upon completion, concentrate the mixture and purify the crude residue using silica gel column chromatography to isolate the target indolo delta-sultone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers substantial strategic benefits that extend beyond mere technical feasibility. The reliance on commercially available and inexpensive starting materials, such as indolin-2-one and substituted styrenes, ensures a stable and resilient supply chain that is not vulnerable to the bottlenecks often associated with exotic or custom-synthesized reagents. The elimination of precious metal catalysts removes a significant cost driver from the bill of materials and obviates the need for expensive metal scavenging steps to meet regulatory limits on residual heavy metals in drug substances. Furthermore, the high atom economy and reduced solvent usage inherent in this one-pot process contribute to a lower environmental footprint, aligning with increasingly strict global sustainability mandates and reducing waste disposal expenditures.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the avoidance of cryogenic conditions or high-energy heating requirements, as the reaction proceeds efficiently at room temperature. This drastic reduction in energy consumption translates directly into lower utility costs for large-scale manufacturing facilities. Additionally, the high yields reported, often exceeding 90% for a broad range of substrates, minimize the loss of valuable raw materials and reduce the volume of material that needs to be reprocessed or discarded. The simplified purification workflow further lowers labor costs and increases throughput capacity, allowing manufacturers to produce high-purity intermediates at a fraction of the cost associated with legacy multi-step syntheses.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality or environmental factors, leading to more consistent batch-to-batch performance. This reliability is critical for maintaining continuous production schedules and meeting tight delivery deadlines for downstream clients. Since the key building blocks are commodity chemicals with established global supply networks, the risk of supply disruption due to geopolitical issues or single-source vendor dependencies is substantially mitigated. This stability allows supply chain planners to forecast inventory needs with greater accuracy and optimize logistics strategies for just-in-time delivery models.
- Scalability and Environmental Compliance: Scaling this chemistry from gram to kilogram or tonne scale presents minimal technical hurdles, as the exotherm is manageable and the reaction kinetics are favorable. The use of dichloromethane or other common organic solvents allows for easy integration into existing manufacturing infrastructure without the need for capital-intensive equipment upgrades. Moreover, the generation of benign byproducts and the absence of toxic heavy metals simplify the wastewater treatment process, ensuring compliance with environmental regulations and reducing the administrative burden associated with hazardous waste reporting. This eco-friendly profile enhances the corporate social responsibility standing of the manufacturing entity and facilitates smoother regulatory approvals in key markets.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their pipelines, we have compiled answers to common inquiries regarding the reaction scope, safety, and scalability. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a realistic overview of what can be expected during process development. Understanding these nuances is essential for making informed decisions about resource allocation and project timelines.
Q: What is the primary biological application of these indolo delta-sultone compounds?
A: According to patent CN116217587A, these novel fused heterocyclic structures exhibit significant antitumor activity, specifically demonstrating inhibitory effects against HepG2 and Caco-2 cancer cell lines, making them valuable leads for oncology drug development.
Q: Why is the DBU and sodium bicarbonate system preferred for this cyclization?
A: This dual-base system provides an optimal balance of nucleophilicity and mildness. DBU acts as a strong non-nucleophilic base to initiate the cascade, while sodium bicarbonate buffers the reaction, preventing decomposition of the sensitive sulfonyl fluoride moiety and ensuring high purity profiles.
Q: How does this method improve upon traditional sultone synthesis routes?
A: Traditional methods often require harsh sulfonating agents or multi-step sequences with protecting groups. This patented approach utilizes a direct tandem cyclization at room temperature, significantly reducing energy consumption, waste generation, and operational complexity while achieving yields up to 99%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolo Delta-Sultone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this novel indolo delta-sultone scaffold in the field of oncology drug discovery. As a premier CDMO partner, we possess the technical expertise and infrastructure necessary to translate this academic innovation into a commercially viable reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from early-stage research to full-scale manufacturing. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and potency.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your next drug development program. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to obtain specific COA data for our reference standards and comprehensive route feasibility assessments that will demonstrate how we can optimize your supply chain for maximum efficiency and cost-effectiveness.
