Advanced Catalytic Synthesis of N-Substituted-3-Aminoacrolein for Commercial Pharmaceutical Manufacturing
Introduction to Breakthrough Catalytic Technology
The landscape of pharmaceutical intermediate manufacturing is undergoing a significant transformation driven by the urgent need for greener, safer, and more cost-effective synthetic routes. A pivotal development in this domain is detailed in patent CN102603546B, which discloses a novel synthetic method for N-substituted-3-aminoacrolein, a critical building block for numerous bioactive molecules. This technology replaces hazardous traditional reagents with a benign oxidative amination strategy, utilizing a specialized silicomanganese composite catalyst (SixMnO2(1+x)) and molecular oxygen from air. By shifting away from stoichiometric oxidants and toxic chlorinating agents, this process addresses major pain points for R&D directors concerned with impurity profiles and supply chain heads focused on regulatory compliance. The core innovation lies in the unique synergy between silicon and manganese, which enhances catalytic activity to levels unattainable by standard manganese dioxide, enabling high-yield production under relatively mild conditions.
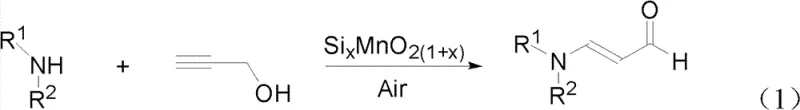
This reaction pathway represents a paradigm shift for the reliable pharmaceutical intermediate supplier market, offering a route that is inherently safer and more scalable. The ability to use air as the sole oxidant not only reduces raw material costs but also simplifies the engineering controls required for the reaction vessel, eliminating the need for complex handling systems for dangerous gases like chlorine or phosgene. Furthermore, the versatility of the catalyst allows for the synthesis of a wide range of derivatives by simply varying the secondary amine substrate, making it a highly adaptable platform technology for custom synthesis projects. As we delve deeper into the technical specifics, it becomes clear that this method offers substantial advantages in terms of atom economy and environmental footprint, aligning perfectly with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-substituted-3-aminoacrolein has relied heavily on two primary methodologies, both of which present severe drawbacks for modern industrial application. The first approach involves the formation of Vilsmeier reagents through the reaction of N-substituted formamides with highly toxic and corrosive agents such as phosgene, oxalyl chloride, phosphorus oxychloride, or bis(trichloromethyl)carbonate. These reagents pose extreme safety risks, requiring specialized containment infrastructure and generating significant quantities of hazardous waste that are costly to treat and dispose of. Moreover, the multi-step nature of this sequence, often involving subsequent reaction with vinyl ethers and alkaline hydrolysis, leads to lower overall yields and complex purification challenges due to the formation of chlorinated byproducts. The second conventional method utilizes the direct reaction of secondary amines with propiolaldehyde or the oxidation of propargyl alcohol using stoichiometric amounts of active manganese dioxide or chromium trioxide. While operationally simpler, these methods suffer from poor atom economy because they require large excesses of heavy metal oxidants, resulting in massive volumes of solid manganese waste that complicate downstream processing and increase the environmental burden.
The Novel Approach
In stark contrast, the novel approach described in the patent leverages a silicomanganese composite catalyst to facilitate the aerobic oxidation of propargyl alcohol in the presence of secondary amines. This method fundamentally alters the reaction economics by replacing expensive and dangerous chemical oxidants with free air, thereby drastically reducing the cost of goods sold (COGS). The use of the SixMnO2(1+x) catalyst allows the reaction to proceed with high selectivity and efficiency, avoiding the over-oxidation side reactions common with harsher oxidants like chromium trioxide. By operating in a closed high-pressure system with air, the process ensures that the oxidation potential is carefully controlled, leading to cleaner reaction profiles and easier isolation of the target aldehyde. This shift from stoichiometric consumption of metals to a truly catalytic cycle represents a major advancement in cost reduction in pharma intermediates manufacturing, as it minimizes raw material consumption and waste generation simultaneously. Additionally, the avoidance of halogenated reagents means the final product has a much cleaner impurity profile, reducing the burden on quality control laboratories.
Mechanistic Insights into Silicomanganese-Catalyzed Aerobic Oxidation
The efficacy of this synthesis relies on the unique electronic and structural properties of the silicomanganese composite catalyst, defined by the formula SixMnO2(1+x) where x ranges from 0 to 10. Mechanistically, the silicon component acts as a structural promoter that stabilizes the active manganese species, preventing their aggregation and deactivation during the oxidative cycle. This stabilization allows the manganese centers to efficiently activate molecular oxygen from the air, generating reactive oxygen species that selectively oxidize the propargyl alcohol to the corresponding aldehyde in situ. The generated aldehyde then immediately undergoes condensation with the secondary amine to form the enamine product, driving the equilibrium forward. Unlike pure manganese dioxide, which often requires stoichiometric quantities to drive the reaction to completion due to surface passivation, the silicomanganese composite maintains high surface activity throughout the reaction duration. This sustained activity is crucial for achieving the reported high yields, as it ensures that the oxidation rate matches the condensation rate, preventing the accumulation of unstable intermediates.
From an impurity control perspective, this mechanism offers distinct advantages over traditional methods. The use of air as a clean oxidant eliminates the introduction of halogen atoms into the reaction matrix, thereby preventing the formation of difficult-to-remove chlorinated or brominated impurities that are common in Vilsmeier-type reactions. Furthermore, the heterogeneous nature of the catalyst allows for easy separation via simple filtration at the end of the reaction, minimizing metal leaching into the product stream. This is particularly important for pharmaceutical applications where residual heavy metal limits are stringent. The reaction conditions, typically maintained between 60°C and 90°C, are mild enough to prevent thermal degradation of the sensitive aminoacrolein structure, yet sufficient to overcome the activation energy barrier for aerobic oxidation. This balance ensures high-purity aminoacrolein derivatives are obtained with minimal byproduct formation, simplifying the recrystallization steps required to meet commercial specifications.
How to Synthesize N-Substituted-3-Aminoacrolein Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize the benefits of the silicomanganese catalyst. The process is designed to be robust and scalable, utilizing standard high-pressure reactor equipment available in most pilot and production facilities. The key to success lies in maintaining the optimal ratio of propargyl alcohol to the secondary amine, typically between 1:1 and 1:2, and ensuring adequate oxygen transfer by maintaining system pressure between 1.5 and 2.5 MPa. The catalyst loading is remarkably low, with a preferred molar ratio of propargyl alcohol to catalyst (based on manganese content) of 1:0.15 to 1:0.2, which is a fraction of the loading required for conventional manganese dioxide. Solvent selection is also critical, with methanol and ethanol proving to be the most effective media for solubilizing reactants while supporting the catalytic cycle. Detailed standardized synthesis steps follow below to guide process engineers in replicating these results.
- Charge a high-pressure reactor with secondary amine, propargyl alcohol, silicomanganese composite catalyst (SixMnO2(1+x)), and an organic solvent such as methanol or ethanol.
- Heat the mixture to 60-90°C and introduce air to maintain a system pressure between 1.5 and 2.5 MPa, reacting for 6 to 8 hours.
- Upon completion, cool the reaction, release pressure, filter off the catalyst, concentrate the filtrate, and recrystallize the residue to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology translates into tangible strategic advantages beyond mere technical performance. The elimination of phosgene, oxalyl chloride, and other highly regulated toxic precursors removes significant logistical hurdles and liability risks associated with the transportation and storage of hazardous chemicals. This simplification of the raw material portfolio enhances supply chain reliability, as the primary inputs—secondary amines and propargyl alcohol—are commodity chemicals with stable global availability. Furthermore, the drastic reduction in catalyst loading compared to traditional manganese dioxide methods leads to substantial cost savings in raw material procurement and waste disposal fees. The ability to use air as the oxidant effectively removes the cost of purchasing chemical oxidants entirely, directly impacting the bottom line. These factors combined create a more resilient and cost-efficient supply chain capable of meeting fluctuating market demands without the bottlenecks associated with hazardous material handling.
- Cost Reduction in Manufacturing: The economic impact of switching to this catalytic system is profound, primarily driven by the reduction in catalyst consumption and the elimination of expensive stoichiometric oxidants. Traditional methods often require equimolar or greater amounts of manganese dioxide, generating tons of solid waste per ton of product, whereas this new method operates with catalytic loadings of roughly 20 mol%. This reduction not only lowers the direct cost of the catalyst but also significantly decreases the cost of waste treatment and disposal, which is a major hidden expense in fine chemical manufacturing. Additionally, the simplified workup procedure, involving simple filtration and recrystallization, reduces labor hours and solvent consumption during purification. These cumulative efficiencies result in a significantly lower cost of goods sold, providing a competitive pricing advantage in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the regulatory restrictions surrounding the transport of toxic reagents like phosgene and oxalyl chloride. By adopting a synthesis route that relies on benign air oxidation and readily available amines, manufacturers can mitigate the risk of supply disruptions caused by regulatory changes or transportation bans. The raw materials for this process are widely produced and sourced from multiple suppliers, reducing dependency on single-source vendors for specialized reagents. This diversification of the supply base ensures that production schedules can be maintained even during market volatility. Moreover, the safer nature of the process reduces the likelihood of plant shutdowns due to safety incidents or environmental compliance audits, ensuring a steady flow of high-quality intermediates to downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes that involve hazardous gases often requires exponential increases in safety infrastructure costs, but this aerobic oxidation method scales linearly with standard pressure vessel technology. The use of air as the oxidant means that the process is inherently greener, producing water as the primary byproduct of oxidation rather than toxic salt wastes. This aligns with increasingly strict environmental regulations globally, future-proofing the manufacturing asset against tighter emission standards. The reduced E-factor (mass of waste per mass of product) makes the process attractive for sustainability reporting and helps companies meet their corporate social responsibility goals. Consequently, this technology facilitates the commercial scale-up of complex pharmaceutical intermediates with minimal environmental impact and regulatory friction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this silicomanganese-catalyzed synthesis. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details is crucial for R&D teams evaluating the feasibility of technology transfer and for procurement teams assessing the long-term viability of the supply source. The clarity provided here aims to remove ambiguity regarding the process capabilities and limitations.
Q: What are the primary advantages of the silicomanganese catalyst over traditional manganese dioxide?
A: The silicomanganese composite catalyst (SixMnO2(1+x)) exhibits significantly higher activity, allowing for catalytic usage (0.05-1 molar ratio) compared to the stoichiometric or near-stoichiometric amounts required for active manganese dioxide. This drastically reduces solid waste generation and improves overall process economics.
Q: Does this synthesis method eliminate the use of hazardous reagents like phosgene?
A: Yes, unlike conventional Vilsmeier-Haack type approaches that rely on phosgene, oxalyl chloride, or phosphorus oxychloride, this patented method utilizes propargyl alcohol and secondary amines with air as the terminal oxidant, fundamentally removing highly toxic and corrosive reagents from the supply chain.
Q: What yields can be expected for N-substituted-3-aminoacrolein derivatives using this technology?
A: Under optimized conditions (60-90°C, 1.5-2.5 MPa air pressure), the process consistently achieves yields exceeding 90% for various derivatives, including N,N-di-n-butyl and N,N-di-n-propyl variants, demonstrating robust scalability for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Substituted-3-Aminoacrolein Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the drug development pipeline. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging advanced catalytic technologies like the one described in CN102603546B, we can offer cost-effective solutions that do not compromise on quality or safety. Our dedication to green chemistry principles ensures that our manufacturing processes are sustainable and compliant with international environmental standards.
We invite you to collaborate with us to optimize your supply chain for N-substituted-3-aminoacrolein derivatives. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your project timelines and budgetary goals. Let us be your partner in bringing innovative therapies to market faster and more efficiently.
