Enzymatic Synthesis of High-Purity 3 Beta 7 Beta Dihydroxy 5 Alpha H UDCA Isomer for Commercial Scale
The pharmaceutical industry's relentless pursuit of high-purity reference standards and intermediates has brought significant attention to bile acid derivatives, specifically the isomers of Ursodeoxycholic Acid (UDCA). Patent CN115637281A introduces a groundbreaking biocatalytic method for preparing the 3 beta, 7 beta-dihydroxy-5 alpha-H UDCA isomer, a critical impurity standard required for rigorous quality control in bile acid therapeutics. This innovation addresses the longstanding challenges associated with synthesizing specific steroid isomers, which traditionally suffer from complex multi-step chemical sequences and poor stereocontrol. By leveraging the exquisite specificity of engineered enzymes, this technology enables the production of the target compound with exceptional purity and yield, fundamentally shifting the paradigm from hazardous chemical synthesis to sustainable biomanufacturing. For global supply chains, this represents a pivotal opportunity to secure reliable sources of high-value pharmaceutical intermediates while adhering to increasingly stringent environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
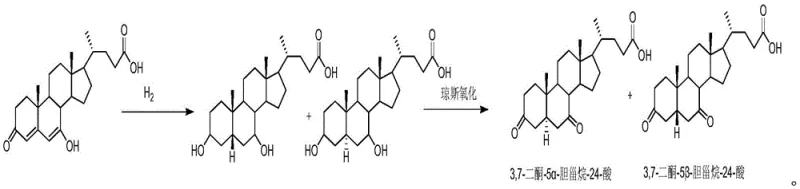
Historically, the preparation of specialized bile acid isomers like the 3 beta, 7 beta-dihydroxy-5 alpha-H variant has relied heavily on classical organic synthesis techniques that are fraught with inefficiencies and safety hazards. Traditional routes often involve the use of aggressive oxidizing agents such as Jones reagent and require multiple protection and deprotection steps to manage the reactivity of hydroxyl groups on the steroid nucleus. These chemical processes frequently necessitate the use of toxic and volatile organic solvents, including dichloromethane, methanol, and acetic anhydride, which pose significant risks to operator safety and create substantial waste disposal burdens. Furthermore, chemical reduction methods often lack the necessary stereoselectivity, leading to the formation of unwanted isomeric byproducts that are difficult to separate, thereby drastically reducing the overall yield and compromising the purity of the final product. The cumulative effect of these factors is a manufacturing process that is not only costly due to low material throughput but also environmentally unsustainable, making it increasingly difficult to justify in modern green chemistry frameworks.
The Novel Approach
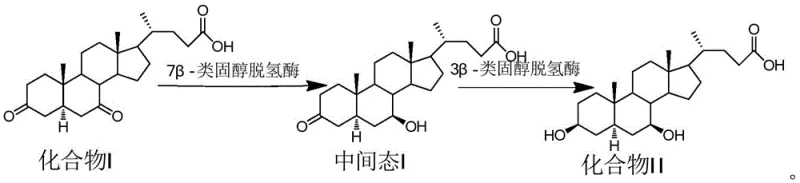
In stark contrast to the cumbersome chemical methodologies, the novel approach detailed in the patent utilizes a streamlined two-step enzymatic cascade that achieves the desired transformation with remarkable precision. The process begins with 3,7-dione-5 α -cholestan-24-oic acid (Compound I) as the starting material, which is subjected to a highly specific reduction catalyzed by 7 β -steroid dehydrogenase. This biocatalyst selectively reduces the ketone at the 7-position to a hydroxyl group with the correct beta stereochemistry, generating a key intermediate without affecting other functional groups. Subsequently, a second enzymatic step employing 3 β -steroid dehydrogenase reduces the remaining ketone at the 3-position, completing the synthesis of the target 3 beta, 7 beta-dihydroxy-5 alpha-H UDCA isomer. This chemo-enzymatic strategy eliminates the need for hazardous reagents and operates under mild physiological conditions, typically around 30-35 °C and neutral pH, ensuring the integrity of the sensitive steroid skeleton throughout the reaction.
Mechanistic Insights into Enzymatic Stereoselective Reduction
The core of this technological advancement lies in the sophisticated application of steroid dehydrogenases, which function as biological catalysts to drive the reduction of ketone groups with absolute stereocontrol. The 7 β -steroid dehydrogenase utilized in the first step is engineered to recognize the specific conformation of the 5-alpha steroid nucleus and deliver a hydride equivalent exclusively to the beta face of the C7 carbonyl. This specificity is crucial because the spatial arrangement of hydroxyl groups on the bile acid scaffold dictates the biological activity and physicochemical properties of the molecule. The enzyme operates via a cofactor-dependent mechanism, utilizing NADH or NADPH generated in situ by a coupled glucose dehydrogenase system, which ensures a continuous supply of reducing equivalents without the need for stoichiometric amounts of expensive chemical reductants. This coupling not only drives the equilibrium towards product formation but also simplifies the reaction workup, as the byproduct is merely gluconic acid, which is easily removed during purification.
Following the initial reduction, the intermediate undergoes a second transformation mediated by 3 β -steroid dehydrogenase, which exhibits complementary specificity for the C3 position. The sequential order of these enzymatic steps is not arbitrary; experimental data within the patent indicates that performing the 7-reduction prior to the 3-reduction results in superior reaction kinetics and product purity compared to the reverse sequence. This suggests a synergistic effect where the introduction of the 7-hydroxyl group potentially alters the substrate's conformation or solubility, making it a more favorable substrate for the subsequent 3-reductase. The result is a clean reaction profile with minimal formation of diastereomeric impurities, such as the 3-alpha isomer, which are common pitfalls in non-enzymatic reductions. This high level of control allows manufacturers to achieve purities exceeding 98 percent directly from the crystallization step, significantly reducing the burden on downstream purification processes.
How to Synthesize 3 Beta 7 Beta Dihydroxy 5 Alpha H UDCA Isomer Efficiently
The implementation of this biocatalytic route requires careful optimization of reaction parameters to maximize enzyme activity and substrate solubility. The process typically employs a biphasic or co-solvent system involving isopropanol and water, which balances the solubility of the hydrophobic steroid substrate with the aqueous requirements of the enzymes. Maintaining the pH within the narrow range of 6.5 to 7.5 is critical for preserving enzyme stability and catalytic efficiency throughout the extended reaction periods of 5 to 12 hours for the first step and 2 to 8 hours for the second. Temperature control is equally important, with optimal performance observed between 30 and 35 °C, preventing thermal denaturation of the biocatalysts while ensuring sufficient reaction rates. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture with Compound I, isopropanol, water, glucose, and adjust pH to 6.5-7.5 at 30-35°C.
- Add glucose dehydrogenase, 7β-steroid dehydrogenase, and coenzymes to catalyze the first reduction to Intermediate I.
- Introduce 3β-steroid dehydrogenase and additional glucose to complete the second reduction, yielding Compound II.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from chemical synthesis to this enzymatic platform offers profound strategic benefits that extend beyond simple unit cost metrics. The elimination of toxic solvents and hazardous reagents fundamentally alters the cost structure of manufacturing by removing the need for expensive containment systems, specialized waste treatment protocols, and regulatory compliance measures associated with volatile organic compounds. This simplification of the process infrastructure translates directly into reduced operational expenditures and a lower total cost of ownership for the production facility. Furthermore, the high specificity of the enzymes minimizes the generation of difficult-to-separate impurities, which dramatically improves the overall mass balance and yield of the process. This efficiency means that less raw material is wasted, and the throughput per batch is maximized, providing a more predictable and robust supply of the critical intermediate.
- Cost Reduction in Manufacturing: The shift to biocatalysis removes the dependency on precious metal catalysts and stoichiometric chemical reducing agents, which are subject to significant price volatility in the global market. By utilizing renewable biocatalysts and inexpensive co-substrates like glucose, the variable costs associated with reagents are substantially lowered. Additionally, the simplified downstream processing, which relies on straightforward crystallization and filtration rather than complex chromatographic separations, reduces both the time and energy consumption required to bring the product to specification. These cumulative efficiencies create a leaner manufacturing model that is resilient to supply chain shocks and raw material price fluctuations.
- Enhanced Supply Chain Reliability: The robustness of the enzymatic process under mild conditions enhances the reliability of supply by reducing the risk of batch failures due to runaway reactions or equipment corrosion. The use of stable enzyme preparations and readily available starting materials ensures that production can be scaled up or down rapidly in response to market demand without the long lead times associated with sourcing specialized chemical reagents. This agility is crucial for pharmaceutical supply chains, where continuity of supply is paramount to prevent disruptions in the production of finished dosage forms. The ability to produce high-purity material consistently also reduces the risk of regulatory delays caused by impurity profile variations.
- Scalability and Environmental Compliance: From an environmental perspective, this process aligns perfectly with the principles of green chemistry, significantly reducing the carbon footprint and E-factor of the synthesis. The aqueous nature of the reaction medium and the absence of halogenated solvents simplify wastewater treatment and allow for easier recycling of process streams. This environmental compatibility not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the corporate sustainability profile of the organization. The scalability of the process is further supported by the fact that enzymatic reactions can be easily transferred from laboratory to pilot to commercial scale with minimal re-optimization, facilitating rapid commercialization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specific UDCA isomer. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this intermediate into their analytical or development workflows. The answers provided are derived directly from the technical specifications and experimental data disclosed in the patent literature, ensuring accuracy and relevance for decision-makers.
Q: What are the advantages of the enzymatic method over chemical synthesis for this UDCA isomer?
A: The enzymatic method offers superior stereoselectivity, eliminating the need for toxic solvents like dichloromethane and acetic anhydride. It operates under mild conditions (30-35°C, neutral pH), resulting in higher purity (over 98%) and yield compared to traditional chemical routes.
Q: Why is the order of enzyme addition critical in this synthesis?
A: The patent specifies that reducing the 7-position ketone first (using 7β-steroid dehydrogenase) followed by the 3-position ketone (using 3β-steroid dehydrogenase) yields significantly higher purity and shorter reaction times compared to the reverse order.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process uses aqueous systems with isopropanol co-solvent and avoids hazardous reagents, making it safer and easier to scale up. The simplified purification steps (crystallization and pulping) further enhance its viability for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3 Beta 7 Beta Dihydroxy 5 Alpha H UDCA Isomer Supplier
As the global demand for high-quality bile acid intermediates continues to rise, partnering with a manufacturer that possesses deep technical expertise in biocatalysis is essential for maintaining a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this technological shift, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which ensure that every batch of 3 beta, 7 beta-dihydroxy-5 alpha-H UDCA isomer meets the exacting standards required for pharmaceutical applications. We understand that consistency is key in the supply of reference standards and intermediates, and our state-of-the-art facilities are designed to deliver that reliability year after year.
We invite potential partners to engage with our technical procurement team to discuss how this innovative enzymatic route can be tailored to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to our biocatalytic supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate the superiority of our manufacturing process and help you secure a sustainable supply of this critical pharmaceutical intermediate.
