Scalable Synthesis of 4-Phenoxyphenylboronic Acid for Next-Generation BTK Inhibitors
Introduction to Advanced Organoboron Synthesis
The pharmaceutical industry's relentless pursuit of potent Bruton's Tyrosine Kinase (BTK) inhibitors, such as Ibrutinib and Tirabrutinib, has placed immense pressure on the supply chain for high-quality organoboron intermediates. Patent CN111072697B introduces a transformative preparation method for 4-phenoxyphenylboronic acid (CAS: 51067-38-0), a critical building block in the synthesis of these life-saving oncology and immunology therapeutics. Unlike traditional routes that struggle with purification bottlenecks, this novel approach leverages a symmetrical dibromo-intermediate strategy to bypass the thermal instability and separation difficulties associated with mono-brominated precursors. By shifting the purification burden to an earlier, more manageable stage of the synthesis, this technology offers a robust pathway for producing high-purity materials essential for clinical and commercial drug manufacturing. The method not only addresses the purity specifications required by stringent regulatory bodies but also aligns with the economic necessities of large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-phenoxyphenylboronic acid has relied heavily on the formation of Grignard reagents or direct lithiation of 4-bromodiphenyl ether. While literature reports yields ranging from 84% to 93%, these methods face significant practical hurdles when translated to industrial settings. The primary bottleneck lies in the procurement and purification of the starting material, 4-bromodiphenyl ether. Direct bromination of diphenyl ether often yields a complex mixture of unreacted starting material, the desired mono-bromo product, and over-brominated byproducts. Separating these components is notoriously difficult because 4-bromodiphenyl ether possesses a high boiling point, making vacuum distillation energy-intensive and risky due to potential thermal decomposition. Furthermore, recrystallization often requires multiple cycles to achieve the necessary purity, leading to substantial material loss and extended processing times. These inefficiencies create a fragile supply chain where minor fluctuations in raw material quality can derail entire production batches, resulting in inconsistent yields and compromised cost structures for downstream API manufacturers.
The Novel Approach
The methodology disclosed in patent CN111072697B fundamentally reengineers the synthetic logic by embracing a "brominate-first, functionalize-later" strategy. Instead of struggling to isolate the elusive mono-bromo species, the process intentionally drives the bromination of diphenyl ether to completion using N-bromosuccinimide (NBS), generating 4,4'-dibromodiphenyl ether. This symmetrical intermediate exhibits superior crystallization properties, allowing it to be purified to greater than 99.8% purity through simple slurry or recrystallization techniques, effectively removing isomeric impurities and unreacted diphenyl ether. Once this high-purity scaffold is established, the process employs a controlled mono-lithiation technique. By meticulously regulating the stoichiometry of n-butyllithium and the boron electrophile, the reaction selectively functionalizes only one of the two bromine positions. This inversion of the traditional workflow eliminates the need for high-temperature distillation of sensitive intermediates and ensures that the subsequent borylation step proceeds with exceptional cleanliness. 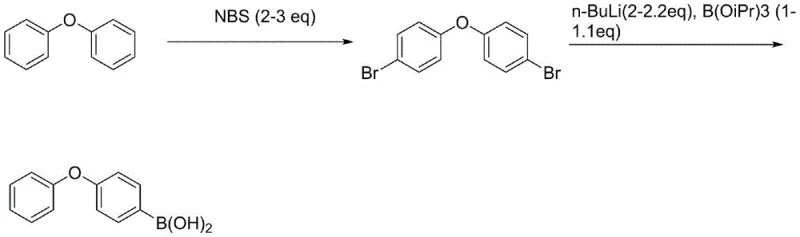
Mechanistic Insights into Selective Mono-Lithiation of Dibromo-Arenes
The core chemical innovation of this process lies in the precise kinetic control exerted during the lithiation phase. When 4,4'-dibromodiphenyl ether is subjected to n-butyllithium at cryogenic temperatures, typically between -70°C and -60°C, the formation of the aryl lithium species is rapid. However, the critical challenge is preventing the second bromine atom from undergoing lithiation, which would lead to double borylation or polymerization side reactions. The patent specifies a molar ratio of n-butyllithium to the dibromo-intermediate of approximately 2.0 to 2.2 equivalents. This specific stoichiometry is counter-intuitive at first glance, as one might expect a 1:1 ratio for mono-substitution; however, in practice, excess base is often required to account for titration errors and moisture scavenging, yet the reaction conditions are tuned such that the mono-lithiated species reacts preferentially with the borate ester before the second lithiation event can occur significantly. The use of triisopropyl borate as the electrophile further enhances selectivity due to its steric bulk and reactivity profile, facilitating the formation of the boronate ester complex which is subsequently hydrolyzed to the free boronic acid.
Impurity control is intrinsically built into this mechanistic pathway through the physical properties of the intermediates. In conventional routes, homocoupling of the aryl lithium species (forming tetraphenyl ether derivatives) is a common side reaction that is difficult to remove. In this novel protocol, the high purity of the incoming 4,4'-dibromodiphenyl ether minimizes the presence of reactive sites that could initiate uncontrolled coupling. Furthermore, the workup procedure involves a carefully controlled acidic quench at temperatures not exceeding 0°C, which prevents the protodeboronation of the sensitive boronic acid product. The final purification via slurring in non-polar solvents like n-heptane effectively removes inorganic salts and residual borate esters, yielding a product with HPLC purity exceeding 99.8%. This level of chemical fidelity is paramount for pharmaceutical applications, where trace metal contaminants or organic impurities can catalyze degradation in the final drug product or complicate the subsequent Suzuki-Miyaura coupling steps.
How to Synthesize 4-Phenoxyphenylboronic Acid Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters regarding solvent selection and temperature gradients to ensure reproducibility and safety. The process begins with the bromination of diphenyl ether in solvents such as dichloroethane or acetonitrile, followed by a rigorous purification of the dibromo-intermediate to establish a clean baseline for the subsequent organometallic step. The detailed standardized synthesis steps, including specific addition rates, cooling profiles, and quenching protocols necessary for safe operation, are outlined below.
- React diphenyl ether with N-bromosuccinimide (NBS) in a suitable solvent to generate 4,4'-dibromodiphenyl ether, ensuring high purity through crystallization.
- Mix the purified dibromo-intermediate with triisopropyl borate in an ether solvent under nitrogen atmosphere and cool to cryogenic temperatures.
- Add n-butyllithium dropwise while strictly controlling stoichiometry to achieve mono-lithiation, followed by acidic hydrolysis to yield the final boronic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this dibromo-mediated synthesis route represents a significant de-risking of the supply chain for BTK inhibitor intermediates. The primary economic driver here is the drastic simplification of the purification train. By replacing energy-intensive vacuum distillation with crystallization-based purification, the process reduces utility consumption and equipment wear, directly translating to lower manufacturing overheads. Additionally, the raw materials—diphenyl ether and NBS—are commodity chemicals with stable global pricing and abundant availability, insulating the production cost from the volatility often seen with specialized halogenated starting materials. The ability to produce a high-purity intermediate in fewer operational steps also shortens the overall cycle time, allowing manufacturers to respond more agilely to fluctuating market demands without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of high-temperature rectification for mono-bromo intermediates removes a major cost center associated with thermal energy and specialized distillation columns. Furthermore, the high yield of the bromination step (over 93%) combined with the efficient crystallization recovery means that raw material utilization is maximized, minimizing waste disposal costs. The one-pot nature of the lithiation and borylation sequence further reduces solvent usage and labor hours, creating a leaner manufacturing process that inherently supports cost reduction in API manufacturing without the need for expensive catalysts or exotic reagents.
- Enhanced Supply Chain Reliability: Relying on diphenyl ether as the starting feedstock offers a strategic advantage, as it is a widely produced industrial chemical with a robust supply network, unlike custom-synthesized brominated phenols which may have single-source bottlenecks. The process has been verified on a hundred-kilogram scale, proving its viability for commercial scale-up of complex intermediates. This scalability ensures that suppliers can meet large-volume orders consistently, reducing the lead time for high-purity intermediates and preventing production stoppages for downstream pharmaceutical clients who rely on just-in-time delivery models for their clinical and commercial batches.
- Scalability and Environmental Compliance: The use of NBS as a brominating agent, while requiring careful handling, generates succinimide as a byproduct, which is easier to manage and dispose of compared to the heavy metal waste streams associated with alternative catalytic bromination methods. The solvent systems employed, such as ethyl acetate, heptane, and THF, are well-understood and can be efficiently recovered and recycled in modern facilities. This alignment with green chemistry principles facilitates smoother regulatory approvals and environmental compliance audits, ensuring long-term operational continuity and reducing the risk of shutdowns due to environmental violations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 4-phenoxyphenylboronic acid, derived directly from the technical disclosures in the relevant patent literature. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this intermediate into their existing drug development pipelines.
Q: Why is the dibromo-intermediate route preferred over direct mono-bromination for this synthesis?
A: Direct mono-bromination of diphenyl ether often results in difficult-to-separate mixtures due to similar boiling points. The dibromo-intermediate route allows for easy purification via crystallization before the critical lithiation step, significantly improving final product purity.
Q: What are the critical parameters for the lithiation step in this process?
A: Strict temperature control between -40°C to -10°C and precise stoichiometric control of n-butyllithium (2.0-2.2 equivalents relative to the dibromo starting material) are essential to prevent double lithiation and ensure high selectivity for the mono-boronic acid product.
Q: Has this synthesis method been validated for industrial production?
A: Yes, the patent documentation confirms that this method has been successfully verified on a hundred-kilogram scale, demonstrating its robustness and suitability for commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Phenoxyphenylboronic Acid Supplier
As the global demand for BTK inhibitors continues to surge, securing a stable source of high-quality organoboron intermediates is critical for maintaining competitive advantage in the pharmaceutical market. NINGBO INNO PHARMCHEM stands ready to support your development needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art cryogenic reactors and stringent purity specifications monitoring systems, ensuring that every batch of 4-phenoxyphenylboronic acid meets the rigorous QC labs standards required for GMP manufacturing. We understand that consistency is key, and our dedicated technical team works closely with clients to optimize logistics and ensure seamless integration into your supply chain.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how our advanced synthesis capabilities can benefit your project. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall COGS. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our commitment to quality and reliability, ensuring that your transition to commercial production is smooth, efficient, and economically viable.
