Advanced Metal-Free Synthesis of Optically Active Oxindole Spirodihydrofuran Compounds for Pharmaceutical Applications
Introduction to Novel Metal-Free Synthetic Methodology
The pharmaceutical industry continuously seeks efficient and environmentally benign routes for constructing complex heterocyclic scaffolds, particularly those containing all-carbon quaternary stereocenters which are prevalent in bioactive molecules. Patent CN113200993A introduces a groundbreaking synthesis method for oxindole spirodihydrofuran compounds, addressing the critical challenges associated with steric hindrance and stereocontrol. This innovation leverages a dual-catalytic system comprising an organic small molecule hydrogen bond catalyst and an iodine-based catalyst, operating under remarkably mild conditions ranging from 0 to 25 degrees Celsius. By avoiding the use of toxic transition metals, this protocol not only enhances the safety profile of the manufacturing process but also simplifies the purification steps required to meet stringent pharmaceutical purity standards. The ability to construct these complex spirocyclic systems with high optical activity represents a significant advancement in the field of medicinal chemistry and fine chemical synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of spiro-oxindole frameworks containing quaternary carbon centers has relied heavily on transition metal catalysis, often involving precious metals such as palladium, rhodium, or ruthenium. These conventional methods frequently suffer from significant drawbacks, including the requirement for harsh reaction conditions, high temperatures, and the use of expensive, air-sensitive ligands. Furthermore, the presence of residual heavy metals in the final product poses a severe regulatory hurdle for pharmaceutical applications, necessitating costly and time-consuming purification processes to reduce metal content to parts-per-million levels. The steric congestion around the quaternary carbon center often leads to poor yields and low enantioselectivity when using standard protocols, limiting the practical utility of these routes for large-scale manufacturing. Additionally, many traditional oxidative cyclization methods utilize stoichiometric amounts of hazardous oxidants, generating substantial chemical waste and increasing the environmental footprint of the synthesis.
The Novel Approach
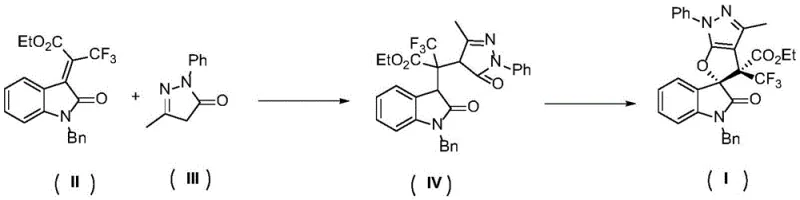
In stark contrast, the methodology disclosed in the patent utilizes a synergistic combination of organocatalysis and halogen catalysis to overcome these historical barriers. The process initiates with a highly stereoselective Michael addition between an oxindole derivative and a pyrazolone, facilitated by a chiral organic small molecule hydrogen bond catalyst. This is followed by an iodine-catalyzed oxidative cyclization using hydrogen peroxide as a green oxidant. This metal-free approach operates at ambient temperatures, drastically reducing energy consumption and eliminating the risk of thermal degradation of sensitive intermediates. The use of elemental iodine or simple iodide salts as catalysts, rather than complex transition metal complexes, significantly lowers the raw material costs and simplifies the supply chain logistics. Moreover, the byproduct of the oxidation step is primarily water, aligning with the principles of green chemistry and reducing the burden on waste treatment facilities. This novel route provides a robust platform for the diverse synthesis of oxindole spirodihydrofurans with excellent functional group tolerance.
Mechanistic Insights into Organocatalytic Michael Addition and Iodine-Mediated Cyclization
The success of this synthetic strategy lies in the precise orchestration of two distinct catalytic cycles that work in tandem to build molecular complexity. The first stage involves the activation of the nucleophilic pyrazolone by the chiral organic small molecule hydrogen bond catalyst, which typically features a thiourea or squaramide moiety capable of forming strong hydrogen bonds with the carbonyl oxygen of the electrophile. This interaction not only increases the electrophilicity of the oxindole double bond but also creates a rigid chiral environment that directs the facial selectivity of the nucleophilic attack. As a result, the Michael addition proceeds with high diastereo- and enantioselectivity, establishing the crucial stereocenters early in the synthesis. The intermediate formed possesses the necessary spatial arrangement to facilitate the subsequent ring-closing step, ensuring that the final spirocyclic product retains the optical purity established in the first step.
Following the formation of the Michael adduct, the reaction transitions to the oxidative cyclization phase mediated by the iodine catalyst. In this step, the iodine species acts as a Lewis acid or generates reactive iodine intermediates that activate the enol or enolate form of the intermediate for intramolecular attack on the oxygen source. The use of hydrogen peroxide as the terminal oxidant regenerates the active iodine species while producing water as the only byproduct, creating a sustainable catalytic cycle. This mechanism effectively overcomes the huge steric hindrance associated with forming the all-carbon quaternary center at the spiro junction. The mild nature of the iodine catalysis prevents side reactions such as over-oxidation or decomposition, which are common pitfalls in traditional oxidative protocols. Understanding this mechanistic pathway allows chemists to fine-tune reaction parameters, such as solvent polarity and catalyst loading, to maximize yield and purity for specific substrate variations, ensuring consistent quality across different batches of production.
How to Synthesize Oxindole Spirodihydrofuran Efficiently
The practical implementation of this synthesis involves a straightforward two-step one-pot or sequential procedure that is amenable to standard laboratory and pilot plant equipment. Initially, the oxindole substrate and pyrazolone coupling partner are dissolved in a suitable organic solvent such as dichloromethane, along with a catalytic amount of the chiral organocatalyst. The mixture is stirred at room temperature for a period ranging from 12 to 48 hours, allowing the Michael addition to reach completion as monitored by thin-layer chromatography. Once the intermediate is formed, an iodine source and hydrogen peroxide are introduced directly to the reaction mixture to trigger the cyclization. The reaction is typically complete within 1 to 4 hours, after which standard aqueous workup and column chromatography yield the pure spirocyclic product. For detailed operational parameters and specific stoichiometry ratios optimized for different substrates, please refer to the standardized synthesis guide below.
- Mix oxindole compound, pyrazolone compound, and organic small molecule hydrogen bond catalyst in an organic solvent at 0-25°C for 12-48 hours to form the intermediate.
- Add an iodine source additive and an oxidant (preferably hydrogen peroxide) to the intermediate mixture and react at 0-25°C for 1-4 hours.
- Perform post-treatment including column chromatography purification to isolate the final optically active oxindole spirodihydrofuran product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this metal-free synthesis route offers compelling advantages that directly address the pain points of procurement managers and supply chain directors in the fine chemical sector. The elimination of precious transition metal catalysts removes a significant cost driver from the bill of materials, as these metals are subject to volatile market prices and geopolitical supply risks. Furthermore, the reliance on commodity chemicals like iodine and hydrogen peroxide ensures a stable and reliable supply chain, reducing the likelihood of production delays due to raw material shortages. The mild reaction conditions translate to lower energy costs and reduced wear and tear on reactor equipment, contributing to overall operational efficiency. By simplifying the purification process and avoiding heavy metal scavenging steps, manufacturers can achieve faster turnaround times and higher throughput, ultimately enhancing the competitiveness of the final pharmaceutical intermediate in the global market.
- Cost Reduction in Manufacturing: The substitution of expensive transition metal catalysts with inexpensive iodine sources and organic catalysts leads to a drastic reduction in raw material costs. Additionally, the use of hydrogen peroxide as a green oxidant minimizes waste disposal expenses associated with hazardous chemical byproducts. The simplified downstream processing, which avoids complex metal removal protocols, further reduces labor and consumable costs, resulting in substantial overall cost savings for the manufacturing process.
- Enhanced Supply Chain Reliability: Sourcing high-purity transition metal catalysts often involves long lead times and complex logistics, whereas the reagents required for this novel method are widely available commodity chemicals. This shift significantly reduces the risk of supply chain disruptions and allows for more flexible inventory management. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, providing greater consistency in production scheduling and delivery reliability for downstream customers.
- Scalability and Environmental Compliance: The metal-free nature of this synthesis aligns perfectly with increasingly stringent environmental regulations regarding heavy metal discharge in pharmaceutical manufacturing. The process generates minimal hazardous waste, primarily water and benign organic salts, facilitating easier compliance with environmental protection standards. The mild temperatures and atmospheric pressure conditions make the reaction inherently safer and easier to scale up from gram to ton quantities without requiring specialized high-pressure or high-temperature equipment, ensuring a smooth transition from R&D to commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this method. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios and assess the potential impact on product quality and regulatory compliance.
Q: What are the key advantages of this metal-free synthesis method over traditional transition metal catalysis?
A: This method eliminates the need for toxic and expensive transition metal catalysts, utilizing inexpensive iodine and hydrogen peroxide instead. It operates under mild conditions (0-25°C) and avoids heavy metal residue issues common in API manufacturing.
Q: How does the process ensure high enantioselectivity for the quaternary carbon center?
A: The process employs chiral organic small molecule hydrogen bond catalysts (such as cinchona alkaloid derivatives) which effectively control the stereochemistry during the initial Michael addition step, resulting in high ee values up to 99%.
Q: Is this synthesis route scalable for industrial production of pharmaceutical intermediates?
A: Yes, the use of readily available reagents like elemental iodine and hydrogen peroxide, combined with mild reaction temperatures and simple workup procedures, makes this route highly suitable for commercial scale-up and industrial application.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxindole Spirodihydrofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthetic methodology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a manufacturing setting. Our state-of-the-art facilities are equipped to handle the specific requirements of organocatalytic reactions, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards. We are committed to delivering consistent quality and reliability, making us the ideal partner for companies seeking to secure their supply of complex spirocyclic building blocks.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how we can optimize your supply chain while reducing your overall manufacturing costs through innovative chemistry.
