Advanced Metal-Free Synthesis of Gamma-Keto Sulfones for Commercial API Production
Introduction to Breakthrough Gamma-Keto Sulfone Technology
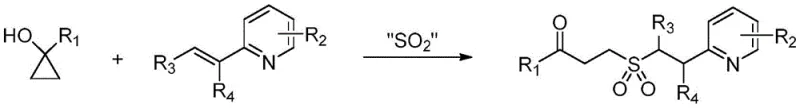
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the need for greener, more efficient, and cost-effective methodologies. A significant advancement in this field is documented in Chinese Patent CN111925319A, which discloses a novel synthetic method for gamma-keto sulfone compounds. These structural motifs are increasingly recognized for their utility in medicinal chemistry, serving as versatile building blocks for bioactive molecules and active pharmaceutical ingredients (APIs). The core innovation lies in a three-component coupling reaction that elegantly merges 1-substituted cyclopropanols, alkenyl pyridines, and a sulfur dioxide substitute. Unlike conventional approaches that often struggle with harsh conditions or toxic reagents, this patented process operates under remarkably mild thermal conditions and, crucially, requires no metal catalyst. For R&D directors and process chemists, this represents a paradigm shift towards cleaner synthesis, offering a pathway to high-purity products with simplified workup procedures that align perfectly with modern regulatory standards for residual solvents and metals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of gamma-keto sulfone scaffolds has been fraught with synthetic challenges that hinder efficient commercial production. Traditional strategies predominantly rely on the oxidation of thioethers, utilizing strong oxidants such as meta-chloroperoxybenzoic acid (mCPBA) or hydrogen peroxide. These oxidative conditions are not only hazardous to handle on a large scale due to exothermic risks but also lack chemoselectivity, often leading to over-oxidation or degradation of sensitive functional groups present in complex drug candidates. Furthermore, alternative radical pathways described in prior art frequently necessitate the use of stoichiometric amounts of transition metal catalysts, particularly copper salts, alongside peroxides. The reliance on heavy metals introduces a significant bottleneck in the manufacturing workflow, as it mandates rigorous and expensive purification steps to reduce metal residues to parts-per-million levels compliant with ICH Q3D guidelines. These legacy methods collectively result in lower overall yields, higher waste generation, and increased production costs, making them less attractive for the cost-sensitive manufacturing of high-volume pharmaceutical intermediates.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology outlined in CN111925319A offers a streamlined, catalyst-free solution that fundamentally reshapes the synthesis of gamma-keto sulfones. By leveraging the ring-strain energy of 1-substituted cyclopropanols, the reaction initiates a radical cascade that seamlessly incorporates sulfur dioxide from a solid surrogate, such as DABCO·(SO2)2, directly into the molecular framework. This approach eliminates the need for external oxidants or transition metal catalysts entirely, thereby removing the associated safety hazards and purification burdens. The reaction proceeds efficiently in common organic solvents like acetonitrile at temperatures as low as 40°C, demonstrating exceptional functional group tolerance across a broad range of substrates including those with electron-withdrawing halogens or electron-donating methyl groups. Reported yields ranging from 59% to 93% underscore the robustness of this transformation, providing process chemists with a reliable tool for constructing these valuable intermediates with high atom economy and minimal environmental impact.
Mechanistic Insights into Metal-Free Radical Coupling
The mechanistic elegance of this transformation lies in its ability to harness the inherent reactivity of strained rings without external activation by metals. The reaction is believed to proceed via a thermal radical initiation where the 1-substituted cyclopropanol undergoes homolytic cleavage of the carbon-carbon bond adjacent to the hydroxyl group. This ring-opening event generates a nucleophilic carbon-centered radical species that is stabilized by the adjacent oxygen atom. This key intermediate then traps the sulfur dioxide molecule released in situ from the solid surrogate, forming a sulfonyl radical. Subsequently, this sulfonyl radical adds across the double bond of the alkenyl pyridine in a regioselective manner, followed by hydrogen abstraction or termination steps to yield the final gamma-keto sulfone product. The absence of metal catalysts ensures that the reaction trajectory is governed purely by the electronic and steric properties of the organic substrates, leading to a cleaner impurity profile that is highly desirable for pharmaceutical applications where trace metal contamination is strictly regulated.
Understanding the impurity control mechanisms in this metal-free system is critical for ensuring consistent product quality during scale-up. Since the reaction does not involve transition metals, the primary impurities typically arise from incomplete conversion of starting materials or minor side reactions involving the radical intermediates. However, the mild conditions (40°C to 60°C) significantly suppress thermal decomposition pathways that are common in high-temperature processes. Furthermore, the use of a solid SO2 surrogate allows for precise stoichiometric control, minimizing the formation of bis-sulfonated byproducts that can occur with excess gaseous SO2. The resulting crude reaction mixtures are generally cleaner, allowing for straightforward purification via standard silica gel chromatography using dichloromethane and methanol gradients. This predictability in impurity formation and removal greatly facilitates the development of robust control strategies, ensuring that the final high-purity pharmaceutical intermediates meet stringent specifications required for downstream drug substance manufacturing.
How to Synthesize Gamma-Keto Sulfones Efficiently
The practical implementation of this synthesis route is designed for ease of operation, making it accessible for both laboratory discovery and pilot plant operations. The protocol involves a simple one-pot procedure where the three key components are mixed in an inert atmosphere, eliminating the need for complex addition sequences or specialized pressure vessels. Detailed standardized synthesis steps are provided below to guide process development teams in replicating these results effectively.
- Combine substituted alkenyl pyridine, 1-substituted cyclopropanol, and a solid sulfur dioxide substitute (such as DABCO·(SO2)2) in an organic solvent like acetonitrile under an inert nitrogen atmosphere.
- Heat the reaction mixture to a mild temperature range of 40°C to 60°C and stir for 18 to 24 hours to allow the three-component coupling and ring-opening to proceed to completion.
- Monitor reaction progress via TLC, then concentrate the solution under reduced pressure and purify the crude residue using column chromatography with a dichloromethane and methanol mobile phase.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this metal-free technology translates into tangible strategic advantages that extend beyond mere chemical efficiency. The elimination of expensive transition metal catalysts such as palladium or copper directly impacts the bill of materials, reducing raw material costs significantly. Moreover, the removal of metal catalysts obviates the need for specialized scavenging resins or complex extraction protocols designed to lower metal content, which further drives down processing costs and shortens cycle times. The use of stable, solid sulfur dioxide surrogates instead of hazardous gases simplifies logistics and storage requirements, mitigating supply chain risks associated with the transportation of compressed toxic gases. This operational simplicity enhances supply continuity, ensuring that production schedules are not disrupted by regulatory hurdles or safety incidents related to hazardous reagent handling.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the downstream processing workflow. By avoiding the use of transition metal catalysts, manufacturers save substantially on the costs associated with catalyst procurement and, more importantly, the extensive purification steps required to meet regulatory limits for heavy metals in APIs. The mild reaction conditions also contribute to energy savings, as heating to 40°C requires significantly less utility consumption compared to high-temperature reflux conditions often seen in traditional methods. Additionally, the high reaction yields reported minimize the loss of valuable starting materials, improving the overall mass balance and reducing the cost per kilogram of the final intermediate. These factors combine to create a highly cost-competitive manufacturing route that supports margin improvement in price-sensitive generic drug markets.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commercially abundant and stable raw materials. The sulfur dioxide surrogate, DABCO·(SO2)2, is a shelf-stable solid that can be sourced from multiple chemical suppliers, reducing dependency on single-source vendors for hazardous gases. The substrates, including substituted cyclopropanols and vinyl pyridines, are readily available building blocks in the fine chemical industry, ensuring a steady flow of inputs for continuous production. The robustness of the reaction to varying substrate electronics means that supply chain disruptions for specific derivatives can often be managed by switching to alternative analogs without re-optimizing the entire process. This flexibility allows for agile response to market demands and reduces the lead time for high-purity pharmaceutical intermediates, enabling faster time-to-market for new drug formulations.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this technology aligns perfectly with green chemistry principles and increasingly strict global regulations. The absence of heavy metals reduces the burden on wastewater treatment facilities and lowers the cost of hazardous waste disposal. The mild operating temperatures and atmospheric pressure conditions make the process inherently safer and easier to scale from gram to multi-ton quantities without requiring exotic high-pressure reactors. The high atom economy of the three-component coupling minimizes waste generation, supporting sustainability goals and reducing the carbon footprint of the manufacturing process. These environmental advantages not only ensure compliance with current regulations but also future-proof the supply chain against tightening environmental standards, making it a sustainable choice for long-term commercial production of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent data and are intended to clarify the operational benefits and technical feasibility for potential partners and licensees.
Q: How does this metal-free method improve impurity profiles compared to traditional copper-catalyzed routes?
A: Traditional methods often rely on transition metal catalysts like copper, which can leave trace residues requiring complex and costly removal steps to meet ICH Q3D guidelines. This novel protocol operates without any metal catalyst, inherently eliminating the risk of heavy metal contamination and significantly simplifying the downstream purification process for high-purity API intermediates.
Q: What are the safety advantages of using DABCO·(SO2)2 over gaseous sulfur dioxide?
A: Gaseous sulfur dioxide is toxic, corrosive, and difficult to handle on an industrial scale, requiring specialized pressurized equipment. The use of solid DABCO·(SO2)2 as a surrogate provides a safe, stable, and easy-to-weigh solid source of SO2, drastically reducing operational hazards and facilitating safer commercial scale-up of complex pharmaceutical intermediates.
Q: Does this synthesis method support a wide range of substrate functional groups?
A: Yes, the patent data demonstrates excellent substrate adaptability, tolerating various electron-withdrawing and electron-donating groups on the pyridine ring, as well as different aryl and alkyl substitutions on the cyclopropanol. This broad scope allows for the efficient generation of diverse chemical libraries for drug discovery without needing to optimize conditions for each new derivative.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gamma-Keto Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for the next generation of therapeutic agents. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of gamma-keto sulfone intermediates meets the highest quality standards required by global regulatory bodies. We understand that consistency and reliability are paramount in the pharmaceutical supply chain, and our dedicated technical team is committed to delivering products that exceed expectations.
We invite you to collaborate with us to leverage this advanced technology for your specific drug development programs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your project's unique volume and purity requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your timeline while optimizing your manufacturing costs.
