Advanced Manufacturing of 2-Bromo-4-Hydroxyanisole via Mild Acetylation and Bromination Strategy
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical intermediates, particularly those serving as precursors for specialized hormonal therapies. Patent CN112374984A introduces a significant advancement in the preparation of 2-bromo-4-hydroxyanisole, a vital building block for the synthesis of Tiratricol, a compound utilized in treating thyroid hormone resistance syndrome and managing thyroid cancer. This novel methodology addresses longstanding inefficiencies in existing manufacturing protocols by replacing complex electrochemical transformations with a streamlined, three-step chemical synthesis. By leveraging standard organic transformations such as acetylation, electrophilic bromination, and hydrolysis, the process achieves exceptional purity and yield without relying on exotic catalysts or energy-intensive equipment. For global procurement teams and R&D directors, this represents a pivotal shift towards more reliable pharma intermediates supplier networks, ensuring consistent quality and availability for downstream drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-bromo-4-hydroxyanisole has been hindered by reliance on directing-group-free remote C-H oxygenation techniques, often employing electrochemical modes to achieve structural transformation. As illustrated in the prior art, these conventional pathways typically necessitate the use of expensive platinum electrodes and specific electrolyte systems such as nBu4NPF6 in DCE/TFA/TFAA mixtures. 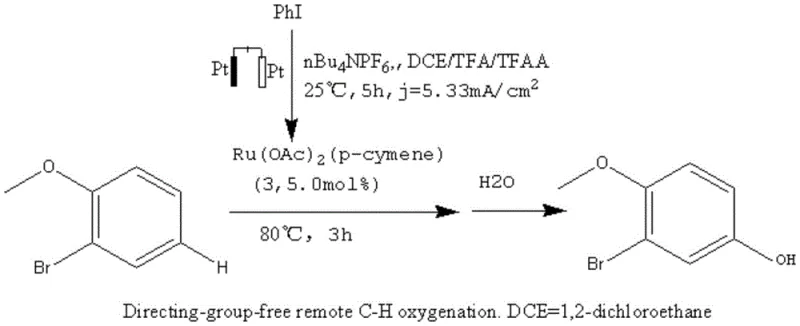 The inherent instability of the platinum electrode under prolonged reaction conditions, combined with the complexity of maintaining precise electrochemical parameters, frequently results in inconsistent batch quality. Furthermore, literature indicates that these electrochemical methods often suffer from yields less than 50%, creating substantial material loss and driving up the cost per kilogram of the final active pharmaceutical ingredient. The requirement for specialized electrochemical reactors also limits the ability to scale these processes to multi-ton production levels, posing a significant bottleneck for supply chain heads looking to secure long-term volume commitments.
The inherent instability of the platinum electrode under prolonged reaction conditions, combined with the complexity of maintaining precise electrochemical parameters, frequently results in inconsistent batch quality. Furthermore, literature indicates that these electrochemical methods often suffer from yields less than 50%, creating substantial material loss and driving up the cost per kilogram of the final active pharmaceutical ingredient. The requirement for specialized electrochemical reactors also limits the ability to scale these processes to multi-ton production levels, posing a significant bottleneck for supply chain heads looking to secure long-term volume commitments.
The Novel Approach
In stark contrast, the invention disclosed in CN112374984A utilizes a classical yet highly optimized organic synthesis strategy that prioritizes operational simplicity and high throughput. The new route begins with the protection of the phenolic hydroxyl group via acetylation, followed by regioselective bromination using N-bromosuccinimide (NBS), and concludes with a mild hydrolysis step. This approach completely eliminates the need for electrochemical cells and precious metal electrodes, substituting them with readily available chemical reagents like acetyl chloride and potassium hydroxide. The transition from an electrochemical paradigm to a thermal chemical paradigm allows for the use of standard glass-lined or stainless steel reactors found in most fine chemical facilities. This fundamental shift not only simplifies the engineering requirements but also drastically improves the reaction yield, with experimental data demonstrating conversion rates exceeding 98%, thereby maximizing raw material utilization and minimizing waste generation.
Mechanistic Insights into Acetylation-Assisted Regioselective Bromination
The core innovation of this process lies in the strategic use of the acetyl group as a temporary protecting and directing moiety to facilitate precise bromination. In the first stage, 4-hydroxyanisole reacts with acetyl chloride in the presence of an organic base, such as triethylamine, at low temperatures ranging from -5°C to -15°C. This low-temperature control is critical to suppress potential side reactions and ensure the exclusive formation of 4-acetyl anisole. By masking the highly reactive phenolic hydroxyl group, the molecule becomes more stable and manageable for the subsequent electrophilic aromatic substitution. The methoxy group at the para-position then acts as an ortho/para director, but since the para-position is occupied by the methoxy group itself and the hydroxyl is protected, the incoming bromine electrophile is directed specifically to the position ortho to the methoxy group, which corresponds to the 2-position of the anisole ring.
Following the protection step, the introduction of N-bromosuccinimide (NBS) under elevated temperatures (50°C to 100°C) drives the bromination efficiently. 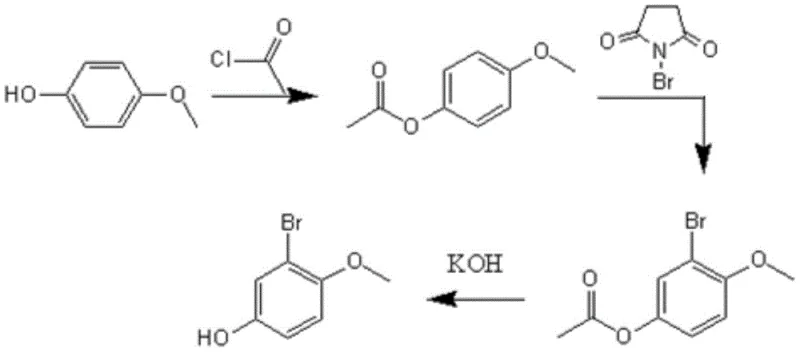 The use of NBS provides a controlled source of bromine radicals or cations, depending on the specific solvent environment, ensuring mono-bromination rather than poly-bromination which is a common impurity challenge in phenol derivatives. The final step involves the removal of the acetyl protecting group using an aqueous potassium hydroxide solution. This saponification reaction is highly efficient and proceeds to completion within 4 to 6 hours at room temperature. The resulting crude product can be easily isolated by filtration after rotary evaporation, and subsequent acidification to pH 3-4 allows for clean extraction into organic solvents. This mechanism ensures that the final product, 2-bromo-4-hydroxyanisole, is obtained with high purity without the need for labor-intensive silica gel column chromatography, a feature that is exceptionally valuable for commercial scale-up of complex pharmaceutical intermediates.
The use of NBS provides a controlled source of bromine radicals or cations, depending on the specific solvent environment, ensuring mono-bromination rather than poly-bromination which is a common impurity challenge in phenol derivatives. The final step involves the removal of the acetyl protecting group using an aqueous potassium hydroxide solution. This saponification reaction is highly efficient and proceeds to completion within 4 to 6 hours at room temperature. The resulting crude product can be easily isolated by filtration after rotary evaporation, and subsequent acidification to pH 3-4 allows for clean extraction into organic solvents. This mechanism ensures that the final product, 2-bromo-4-hydroxyanisole, is obtained with high purity without the need for labor-intensive silica gel column chromatography, a feature that is exceptionally valuable for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 2-Bromo-4-Hydroxyanisole Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible pathway suitable for immediate technology transfer into pilot and production plants. The process is divided into three distinct operational units: protection, functionalization, and deprotection. Each step is designed to minimize solvent exchanges and maximize throughput, with intermediate products often being carried forward directly after simple concentration or filtration. This telescoping capability reduces the overall cycle time and solvent consumption, aligning with green chemistry principles. For R&D teams looking to implement this, the key parameters to monitor are the temperature during the acetyl chloride addition and the stoichiometry of the NBS reagent to prevent over-bromination. The detailed standardized synthesis steps see the guide below.
- React 4-hydroxyanisole with acetyl chloride in an organic alkali solution at -5 to -15°C to form 4-acetyl anisole.
- Add N-bromosuccinimide (NBS) to the 4-acetyl anisole solution at 50 to 100°C to generate 2-bromo-4-acetyl anisole.
- Hydrolyze the brominated intermediate using potassium hydroxide, followed by acidification and extraction to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound benefits for cost reduction in pharmaceutical intermediates manufacturing. By eliminating the dependency on platinum electrodes and electrochemical power supplies, the capital expenditure (CAPEX) required for setting up production lines is significantly reduced. Furthermore, the operational expenditure (OPEX) is lowered due to the use of commodity chemicals like acetyl chloride and NBS, which are available in bulk quantities at stable prices globally. The high yield reported in the patent examples implies that less raw material is wasted per kilogram of output, directly improving the gross margin for manufacturers. This efficiency translates into a more competitive pricing structure for buyers seeking a reliable agrochemical intermediate supplier or pharma partner, as the underlying production costs are inherently lower than those of the legacy electrochemical methods.
- Cost Reduction in Manufacturing: The removal of expensive noble metal catalysts and specialized electrochemical equipment dramatically lowers the barrier to entry for production. Since the process relies on standard thermal reactions, facilities do not need to invest in custom-built electrochemical cells, leading to substantial cost savings in infrastructure. Additionally, the avoidance of silica gel column chromatography for purification removes a major cost center associated with stationary phase materials and large volumes of elution solvents. The ability to obtain high-purity products through simple extraction and crystallization means that solvent recovery systems can be optimized further, reducing utility costs and waste disposal fees associated with hazardous solvent mixtures.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including 4-hydroxyanisole, acetyl chloride, and NBS, are widely produced commodities with robust global supply chains. This reduces the risk of supply disruptions that might occur with specialized reagents or custom-synthesized catalysts. The mild reaction conditions also imply that the process is less sensitive to minor fluctuations in utility supply, such as power stability, which is crucial for continuous manufacturing operations. Consequently, suppliers can offer more consistent lead times and maintain higher safety stock levels, ensuring that downstream drug manufacturers face fewer interruptions in their own production schedules.
- Scalability and Environmental Compliance: The process is explicitly designed for mass production, with steps that are easily scalable from laboratory benchtop to multi-ton reactors. The absence of heavy metal catalysts simplifies the environmental compliance profile, as there is no need for rigorous testing and removal of trace platinum or ruthenium residues from the final API. This facilitates faster regulatory approval and reduces the burden on quality control laboratories. Moreover, the simplified workup procedures generate less hazardous waste, aligning with increasingly stringent environmental regulations and sustainability goals adopted by major pharmaceutical corporations worldwide.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this process into existing manufacturing portfolios.
Q: Why is the new acetylation-bromination route superior to electrochemical methods?
A: The new chemical route eliminates the need for expensive platinum electrodes and complex electrochemical setups, achieving yields over 98% compared to less than 50% in prior art, while avoiding harsh reaction conditions.
Q: Does this process require silica gel column chromatography for purification?
A: No, the process is designed for industrial scalability and does not require complicated purification procedures such as silica gel column chromatography or recrystallization, significantly reducing processing time and solvent waste.
Q: What are the critical temperature controls for the acetylation step?
A: The acetylation of 4-hydroxyanisole must be conducted under strict low-temperature conditions, specifically between -5°C and -15°C, to ensure controlled reaction kinetics and prevent side reactions during the formation of 4-acetyl anisole.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Bromo-4-Hydroxyanisole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our technical team has thoroughly analyzed the pathway described in CN112374984A and is fully prepared to execute this synthesis with precision and scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met whether you are in the clinical trial phase or full-scale commercialization. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting impurities at trace levels, guaranteeing that every batch of 2-bromo-4-hydroxyanisole meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized process can enhance your project's economic viability and timeline efficiency.
