Advanced Base-Catalyzed Deprotection Strategy for Commercial Abacavir Manufacturing
Advanced Base-Catalyzed Deprotection Strategy for Commercial Abacavir Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes that balance efficiency with regulatory compliance, particularly for antiretroviral agents like Abacavir. Patent CN101522680B introduces a transformative methodology for the preparation of Abacavir and its salts, specifically addressing the critical deprotection step of N-2-acylated intermediates. Unlike traditional approaches that rely on harsh acidic environments, this innovation leverages specific alkaline conditions within a water-alcohol mixture to achieve rapid hydrolysis. This technical breakthrough is pivotal for manufacturers aiming to optimize their production lines for high-purity API intermediates. By shifting the paradigm from acid-mediated to base-mediated deprotection, the process not only accelerates reaction kinetics but also fundamentally alters the impurity profile, resulting in a cleaner crude product that requires less intensive downstream processing. For R&D directors and process chemists, understanding the nuances of this base-catalyzed mechanism is essential for implementing scalable and cost-effective manufacturing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Abacavir has been hindered by the inefficiencies associated with removing amino protecting groups under acidic conditions. Prior art, such as that disclosed in EP 434450-A, typically necessitates the use of vigorously corrosive mineral acids like hydrochloric acid at room temperature for extended periods, often spanning up to two days. This reliance on strong acids presents significant operational hazards and equipment corrosion risks, complicating the engineering controls required for safe industrial operation. Furthermore, these acidic protocols frequently demand high dilution rates to manage exotherms and side reactions, leading to massive solvent volumes that strain facility capacity and increase waste disposal costs. Perhaps most critically, the isolation of the free base often requires complex chromatographic purification, a technique that is notoriously difficult to scale and economically prohibitive for large-scale commercial production. The combination of long reaction times, hazardous reagents, and low-throughput purification creates a bottleneck that severely impacts the overall cost of goods and supply chain reliability for this vital HIV treatment.
The Novel Approach
In stark contrast, the methodology described in CN101522680B utilizes inorganic bases, such as sodium hydroxide, in a mixed solvent system to effect deprotection with remarkable speed and selectivity. This novel approach allows the reaction to proceed at elevated temperatures, specifically at the reflux temperature of the solvent mixture, reducing the reaction time from days to merely one hour without compromising product integrity. The use of a water and alcohol mixture, preferably isopropanol, creates a homogeneous reaction environment that facilitates efficient mass transfer while maintaining the stability of the sensitive purine ring system. Crucially, this basic hydrolysis minimizes the formation of degradation by-products that are commonly observed when heating acidic solutions, thereby preserving the yield and optical purity of the final compound. The process enables the direct isolation of the product salt through crystallization rather than chromatography, representing a monumental shift towards greener and more economically viable manufacturing practices for complex pharmaceutical intermediates.
Mechanistic Insights into Base-Catalyzed Amide Hydrolysis
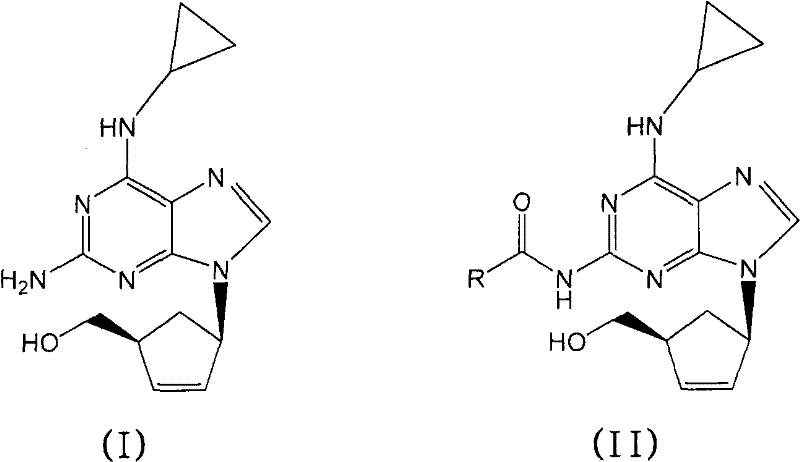
The core of this technological advancement lies in the mechanistic pathway of amide bond cleavage under nucleophilic attack by hydroxide ions. In the presence of an inorganic base like sodium hydroxide, the hydroxide ion acts as a potent nucleophile, attacking the carbonyl carbon of the N-2-acyl group on the purine intermediate. This attack forms a tetrahedral intermediate which subsequently collapses to expel the amine leaving group, regenerating the free amino functionality at the 2-position of the purine ring. Unlike acidic hydrolysis, which relies on protonation of the carbonyl oxygen to activate the electrophile, the basic mechanism avoids the generation of highly acidic species that could potentially protonate other nitrogen atoms within the purine scaffold, leading to ring opening or decomposition. The choice of solvent plays a synergistic role; the aqueous component provides the medium for the ionic base, while the alcoholic component, such as isopropanol, ensures the solubility of the organic intermediate. This balanced solvation environment is critical for maintaining reaction homogeneity and preventing the precipitation of intermediates that could lead to incomplete conversion or localized hot spots.
Furthermore, the stability of the chiral cyclopentyl moiety is rigorously maintained throughout this basic treatment, ensuring no racemization occurs at the stereocenters. The absence of racemization is a key quality attribute, as the biological activity of Abacavir is strictly dependent on its specific stereochemical configuration {(1S, 4R)}. The reaction conditions are tuned such that the base is strong enough to cleave the amide bond but mild enough to leave the glycosidic-like bond between the purine and the carbocyclic ring intact. Following the hydrolysis, the workup procedure involves phase separation where the product, now in its free base or salt form depending on pH adjustment, partitions favorably into the organic phase or precipitates directly upon acidification. This clean separation profile is indicative of a reaction that generates minimal tarry by-products, simplifying the purification landscape significantly compared to acid-catalyzed routes.
How to Synthesize Abacavir Efficiently
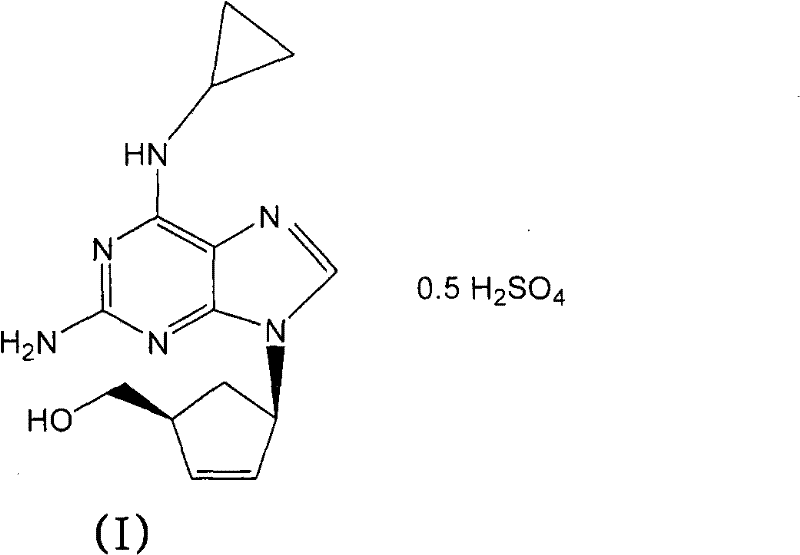
Implementing this synthesis route requires precise control over stoichiometry and thermal parameters to maximize the benefits of the base-catalyzed mechanism. The process begins by suspending the N-acylated intermediate in a defined ratio of isopropanol and aqueous sodium hydroxide, ensuring adequate mixing to initiate the hydrolysis uniformly. Heating the mixture to reflux drives the reaction to completion rapidly, typically within 60 minutes, after which the system is cooled to facilitate phase separation or crystallization. The detailed standardized synthetic steps, including specific molar ratios and temperature ramps, are outlined in the technical guide below to ensure reproducibility and safety during scale-up operations.
- Slurry the N-acylated intermediate in a mixture of isopropanol and aqueous sodium hydroxide solution.
- Heat the reaction mixture to reflux temperature for approximately 1 hour to effect hydrolysis.
- Separate phases, treat the organic layer with sulfuric acid, and crystallize the abacavir hemisulfate salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this base-catalyzed process offers tangible strategic advantages that extend beyond simple yield improvements. The elimination of corrosive mineral acids reduces the requirement for specialized Hastelloy or glass-lined reactors, allowing for the use of more standard stainless steel equipment in certain stages, which translates to lower capital expenditure and maintenance costs. Additionally, the drastic reduction in reaction time from days to hours significantly increases the throughput of existing manufacturing assets, effectively expanding capacity without the need for new facility construction. The ability to operate at higher concentrations means that solvent consumption per kilogram of product is substantially reduced, lowering both raw material costs and the environmental burden associated with solvent recovery and waste treatment. These factors collectively contribute to a more resilient supply chain capable of responding quickly to market demands for antiretroviral therapies.
- Cost Reduction in Manufacturing: The economic impact of replacing chromatographic purification with crystallization cannot be overstated in terms of operational expenditure. Chromatography is a batch-intensive, solvent-heavy process that often limits production scale and drives up the cost per gram of the active ingredient. By enabling crystallization, this process removes the need for expensive silica gel or resin columns and the vast quantities of eluents associated with them. Furthermore, the use of commodity chemicals like sodium hydroxide and isopropanol instead of specialized acidic reagents ensures stable and predictable raw material pricing. The overall simplification of the workflow reduces labor hours and utility consumption, leading to substantial cost savings in the final manufacturing budget.
- Enhanced Supply Chain Reliability: Shorter cycle times directly correlate to improved supply chain agility. With a reaction time of approximately one hour compared to multi-day protocols, manufacturers can turn around batches much faster, reducing lead times for high-purity pharmaceutical intermediates. This speed allows for more flexible production scheduling and the ability to hold lower inventory levels while still meeting delivery commitments. The robustness of the reaction conditions also implies a lower risk of batch failure due to sensitivity to minor variations in temperature or reagent quality, ensuring a consistent and reliable flow of material to downstream formulation partners.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, moving away from strong mineral acids reduces the generation of acidic wastewater streams that require neutralization before discharge. The process generates fewer organic impurities, which simplifies the waste profile and reduces the complexity of effluent treatment. The high concentration capability of the reaction minimizes the total volume of waste solvent generated per unit of product, aligning with green chemistry principles. This improved environmental footprint facilitates easier regulatory approval and permits for expansion, making the commercial scale-up of complex pharmaceutical intermediates smoother and more sustainable in the long term.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this deprotection strategy. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on reaction parameters and product quality attributes. Understanding these details is crucial for process engineers tasked with technology transfer and validation.
Q: Why is base-catalyzed hydrolysis preferred over acidic conditions for Abacavir synthesis?
A: Base-catalyzed hydrolysis avoids the use of corrosive mineral acids, significantly reduces reaction time from days to hours, prevents product decomposition observed under acidic heating, and eliminates the need for complex chromatographic purification.
Q: What are the critical solvent parameters for this deprotection reaction?
A: The process utilizes a mixture of water and a C1-C6 alcohol, preferably isopropanol. This solvent system facilitates high-concentration reactions, allows for efficient phase separation upon completion, and supports direct crystallization of the product salt.
Q: Does this method impact the stereochemical integrity of the cyclopentyl ring?
A: No, the mild basic conditions employed in this novel process ensure that no racemization occurs at the chiral centers of the cyclopentyl ring, maintaining the high optical purity required for the active pharmaceutical ingredient.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Abacavir Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the rigorous demands of the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the base-catalyzed deprotection of Abacavir are translated into reliable industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of API intermediate meets the highest international standards. Our commitment to continuous improvement allows us to offer clients not just a product, but a optimized supply solution that balances cost, quality, and speed.
We invite potential partners to engage with our technical procurement team to discuss how this enhanced manufacturing route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages tailored to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and compliant chemistry available.
