Advanced Photocatalytic Synthesis of Phosphorylated Quinazolinones for Commercial Scale-up
Introduction to Next-Generation Quinazolinone Synthesis
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to access biologically active heterocyclic scaffolds. A groundbreaking development in this arena is detailed in patent CN114644655A, which discloses a novel preparation method for phosphorylated quinazolinone compounds via visible light photocatalysis. This technology represents a significant leap forward in azacycle synthesis, moving away from traditional thermal methods towards a greener, photo-driven paradigm. By leveraging the power of visible light as a clean energy source, this method enables the direct functionalization of terminal non-activated olefins through a radical cycloaddition mechanism. The process is characterized by its mild reaction conditions, operating effectively at room temperature in an air environment, which drastically reduces energy consumption and safety hazards associated with high-pressure or high-temperature reactors. For R&D directors and procurement managers alike, this innovation offers a compelling value proposition: a robust, scalable route to high-value intermediates that aligns perfectly with modern green chemistry principles and supply chain sustainability goals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazoline and its derivatives has relied heavily on classical thermal condensation reactions. The typical preparation method involves heating 2-acyl-N-anilides in the presence of ammonia. While chemically feasible, this traditional approach suffers from several critical drawbacks that hinder its efficiency in a modern commercial setting. Firstly, the requirement for elevated temperatures and ammonia gas introduces significant safety risks, necessitating specialized pressure-rated equipment and rigorous hazard management protocols. Secondly, these thermal processes often generate substantial amounts of chemical waste and by-products, leading to complex downstream purification challenges and increased environmental compliance costs. Furthermore, the harsh conditions can limit the tolerance of sensitive functional groups, restricting the structural diversity of the final products. For a reliable pharmaceutical intermediate supplier, relying on such polluting and dangerous methodologies is increasingly untenable in a regulatory landscape that demands stricter adherence to environmental, social, and governance (ESG) standards.
The Novel Approach
In stark contrast, the novel photocatalytic method described in the patent data offers a transformative solution to these legacy issues. By utilizing visible light irradiation as the driving force, the reaction proceeds under exceptionally mild conditions—specifically at room temperature and under an ambient air atmosphere. This eliminates the need for external heating and inert gas protection, simplifying the operational setup significantly. The core of this innovation lies in the use of a cheap organic photosensitizer, Eosin Y, which facilitates the generation of aryl phosphorus oxygen radicals. These radicals then undergo a cycloaddition with the terminal non-activated olefin of the quinazolinone precursor. This strategy not only achieves high atom economy but also ensures excellent selectivity and yield without the need for toxic transition metal catalysts. The result is a streamlined process that minimizes waste, reduces energy usage, and provides a safer working environment, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Visible Light Photocatalytic Radical Cycloaddition
To fully appreciate the technical sophistication of this synthesis, one must delve into the mechanistic details of the visible light-mediated radical pathway. The reaction initiates when the photosensitizer, Eosin Y, absorbs photons from the visible light source (typically around 450nm wavelength), transitioning from its ground state to an excited state. In this excited state, the catalyst participates in a single electron transfer (SET) process with the aryl phosphine oxide substrate. This interaction generates a highly reactive aryl phosphorus oxygen radical while simultaneously forming a radical anion of the photosensitizer. Crucially, the system utilizes molecular oxygen from the air as the terminal oxidant. The radical anion of the photosensitizer transfers an electron to oxygen, regenerating the ground state catalyst and producing a superoxide anion radical. This catalytic cycle ensures that the photosensitizer is not consumed stoichiometrically, allowing for high turnover numbers and efficient use of the catalyst.
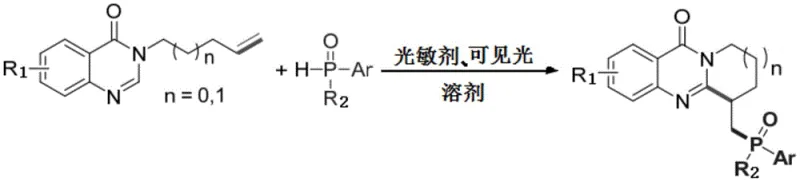
Following the generation of the phosphorus-centered radical, the mechanism proceeds through a radical addition to the unactivated terminal alkene moiety of the 3-(pent-4-en-1-yl)quinazolin-4(3H)-one substrate. This addition creates a new carbon-centered radical intermediate, which subsequently undergoes an intramolecular cyclization or further oxidation to form the stable C-P bond and the final phosphorylated quinazolinone ring system. The beauty of this mechanism lies in its ability to activate inert C-H and C=C bonds under such gentle conditions. The use of air as the oxidant is particularly noteworthy from a process chemistry perspective, as it removes the need for stoichiometric chemical oxidants that often produce hazardous waste streams. This elegant interplay between photo-excitation, radical generation, and aerobic oxidation results in a highly efficient transformation that tolerates a wide array of functional groups, ensuring high purity and minimal impurity profiles in the final high-purity pharmaceutical intermediates.
How to Synthesize Phosphorylated Quinazolinone Efficiently
Implementing this photocatalytic protocol in a laboratory or pilot plant setting is straightforward, thanks to the simplicity of the reagents and conditions. The process begins by dissolving the quinazolinone substrate and the aryl phosphine oxide coupling partner in a suitable solvent, such as 2-methyltetrahydrofuran (2-MeTHF), which is itself a green solvent derived from renewable resources. A catalytic amount of Eosin Y is added to the mixture, and the reaction vessel is kept open to the air or sparged with oxygen to ensure adequate oxidant supply. The mixture is then subjected to irradiation from a visible light source, such as a blue LED array (450nm), at room temperature. Reaction progress is typically monitored via thin-layer chromatography (TLC) until the starting material is fully consumed. Upon completion, the workup involves simple filtration and solvent removal, followed by purification via silica gel column chromatography. For a detailed breakdown of the specific molar ratios, reaction times, and isolation procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- Dissolve 3-(pent-4-en-1-yl)quinazolin-4(3H)-one and aryl phosphine oxide in 2-methyltetrahydrofuran solvent with Eosin Y catalyst under air atmosphere.
- Irradiate the reaction mixture with 450nm visible light at room temperature for approximately 12 hours to facilitate radical cycloaddition.
- Monitor reaction completion via TLC, then purify the crude product using silica gel column chromatography to obtain the high-purity target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology translates into tangible strategic benefits that go beyond mere chemical novelty. The shift from thermal to photochemical synthesis fundamentally alters the cost structure and risk profile of manufacturing these valuable intermediates. By eliminating the need for expensive transition metal catalysts and harsh reagents, the raw material costs are significantly optimized. Furthermore, the mild reaction conditions reduce the burden on facility infrastructure, allowing for production in standard glass-lined reactors equipped with LED lighting rather than specialized high-pressure autoclaves. This flexibility enhances supply chain resilience, enabling faster response times to market demand fluctuations. The following points detail how this technology drives value across the commercial landscape.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of Eosin Y, an inexpensive and commercially abundant organic dye, replacing costly noble metal catalysts often found in traditional cross-coupling reactions. Additionally, the utilization of atmospheric oxygen as the oxidant eliminates the procurement and handling costs associated with chemical oxidants. The simplified workup procedure, which avoids complex quenching steps for reactive metals, further reduces labor and waste disposal expenses. Collectively, these factors contribute to a substantially lower cost of goods sold (COGS), providing a competitive edge in the pricing of API intermediates.
- Enhanced Supply Chain Reliability: Operating at room temperature and under air atmosphere significantly mitigates safety risks associated with thermal runaways or pressurized gas handling. This inherent safety profile reduces the likelihood of production stoppages due to safety incidents or regulatory inspections. Moreover, the reagents involved, such as 2-methyltetrahydrofuran and aryl phosphine oxides, are readily available from multiple global suppliers, reducing the risk of single-source bottlenecks. This robustness ensures a continuous and reliable flow of materials, which is critical for maintaining uninterrupted production schedules in the fast-paced pharmaceutical sector.
- Scalability and Environmental Compliance: The scalability of photochemical reactions has historically been a challenge, but advancements in reactor design have made large-scale photoredox catalysis a reality. The high atom economy of this specific radical cycloaddition means that fewer atoms end up as waste, aligning with strict environmental regulations. The absence of heavy metal residues simplifies the purification process and ensures that the final product meets stringent purity specifications required for drug substances without the need for extensive metal scavenging steps. This environmental compatibility not only future-proofs the manufacturing process against tightening regulations but also enhances the brand reputation of the supplier as a leader in sustainable chemistry.
Frequently Asked Questions (FAQ)
Understanding the nuances of this new synthetic methodology is essential for stakeholders evaluating its potential for integration into existing supply chains. The following questions address common inquiries regarding the technical feasibility, safety, and versatility of the photocatalytic phosphorylation process. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Whether you are concerned about substrate scope or catalyst recovery, the information below aims to clarify the operational realities of this innovative approach.
Q: What are the primary advantages of this photocatalytic method over conventional thermal synthesis?
A: Unlike conventional methods that require heating 2-acyl-N-anilides in the presence of ammonia—which poses significant safety risks and pollution issues—this photocatalytic approach operates at room temperature using visible light. It utilizes air as a green oxidant and avoids harsh conditions, resulting in a safer, more environmentally friendly process with high atom economy.
Q: Is the photosensitizer used in this process cost-effective for large-scale production?
A: Yes, the process employs Eosin Y (tetrabromofluorescein) as the photosensitizer, which is a commercially available and inexpensive organic dye. Its low cost, combined with the fact that it does not require additional expensive oxidants or transition metal catalysts, makes the overall process highly economically viable for industrial scale-up.
Q: Does this synthetic route support a wide range of substrate variations?
A: The method demonstrates excellent substrate adaptability. It tolerates various substituents on the quinazolinone ring, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups like trifluoromethyl and halogens. Additionally, different aryl phosphine oxides can be utilized, allowing for the diverse functionalization required in drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphorylated Quinazolinone Supplier
As the global demand for complex heterocyclic building blocks continues to rise, partnering with a technically proficient CDMO is crucial for success. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our team of expert chemists is well-versed in adapting cutting-edge academic technologies, such as the visible light photocatalysis described in CN114644655A, into robust industrial processes. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to ensure every batch meets the highest quality standards. Our infrastructure is designed to handle sensitive photochemical reactions safely and efficiently, ensuring that your project moves from bench to plant without compromise.
We invite you to collaborate with us to leverage this advanced synthetic route for your next drug development program. By choosing NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to contact our technical procurement team today to request specific COA data for our quinazolinone portfolio and to discuss route feasibility assessments for your custom synthesis needs. Together, we can accelerate the delivery of life-saving medicines to the market while optimizing costs and maintaining the highest standards of quality and sustainability.
