Advanced Synthesis of Water-Soluble Near-Infrared Quinoline Squaraine Dyes for Bio-Imaging
Advanced Synthesis of Water-Soluble Near-Infrared Quinoline Squaraine Dyes for Bio-Imaging
The pharmaceutical and biotechnology sectors are increasingly demanding high-performance fluorophores capable of deep-tissue imaging with minimal background interference. Patent CN101544844A introduces a groundbreaking class of quinoline-based water-soluble near-infrared luminescent squaraine dyes that address these critical needs. Unlike traditional dyes limited to the visible spectrum, these novel compounds exhibit fluorescence emission wavelengths ranging from 800 nm to 1160 nm, ensuring excellent penetrability through biological tissues. The technology leverages a robust synthetic pathway involving the functionalization of 2-methylquinoline precursors followed by a controlled condensation with squaric acid. This report provides a comprehensive technical and commercial analysis of this innovation, highlighting its potential to revolutionize fluorescent labeling, biological probes, and new drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, squaraine dyes synthesized from pyridine or aniline precursors have faced significant hurdles in biological applications due to their inherent physicochemical limitations. Traditional methods often yield dyes with absorption and emission wavelengths restricted to the visible light range of 500 nm to 650 nm, which suffers from high self-absorption and serious mutual interference between excitation and emission spectra. Furthermore, many conventional squaraine dyes exhibit poor water solubility and small Stokes shifts of less than 30 nm, leading to substantial background noise in complex biological environments. These factors severely limit their sensitivity and utility in high-precision tasks such as in vivo imaging or detecting trace biomarkers, where signal-to-noise ratios are paramount for accurate diagnostic outcomes.
The Novel Approach
The patented methodology overcomes these deficiencies by utilizing a rigid quinoline heterocyclic system conjugated with an aromatic squaric acid four-membered ring. This structural modification extends the conjugated electron cloud, successfully shifting the emission into the near-infrared region while maintaining a large Stokes shift greater than 100 nm. The process involves a straightforward condensation reaction where quinoline quaternary ammonium salts react with squaric acid under azeotropic distillation conditions to remove water efficiently. This approach not only simplifies the purification process through standard silica gel chromatography but also ensures the final product possesses the necessary water solubility and stability for industrial application.
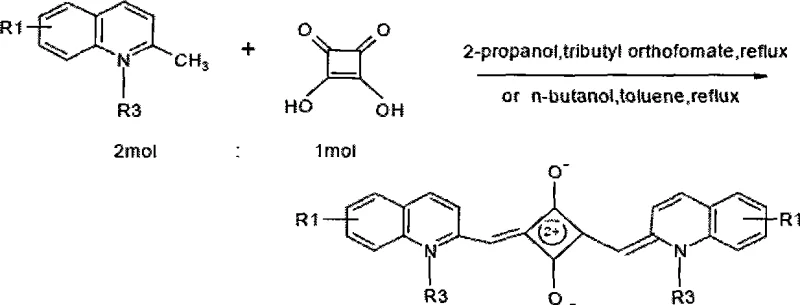
This novel synthetic route allows for the precise tuning of optical properties by varying substituents on the quinoline ring, such as nitro, amino, or sulfonic groups, without compromising the core stability of the dye molecule. The result is a versatile platform of dyes that offer high luminescence quantum yields and exceptional light stability, making them superior candidates for next-generation bio-analytical reagents.
Mechanistic Insights into Quinoline Quaternization and Condensation
The synthesis begins with the strategic functionalization of the quinoline backbone, specifically through the quaternization of the nitrogen atom in 2-methylquinoline derivatives. This step is critical as it activates the methyl group for subsequent condensation while simultaneously introducing solubilizing chains, such as carboxylic acid groups, via reaction with iodoacids. The mechanism relies on nucleophilic substitution where the lone pair on the quinoline nitrogen attacks the alkyl halide, forming a stable quaternary ammonium salt that serves as the active methylene component in the final dye assembly.
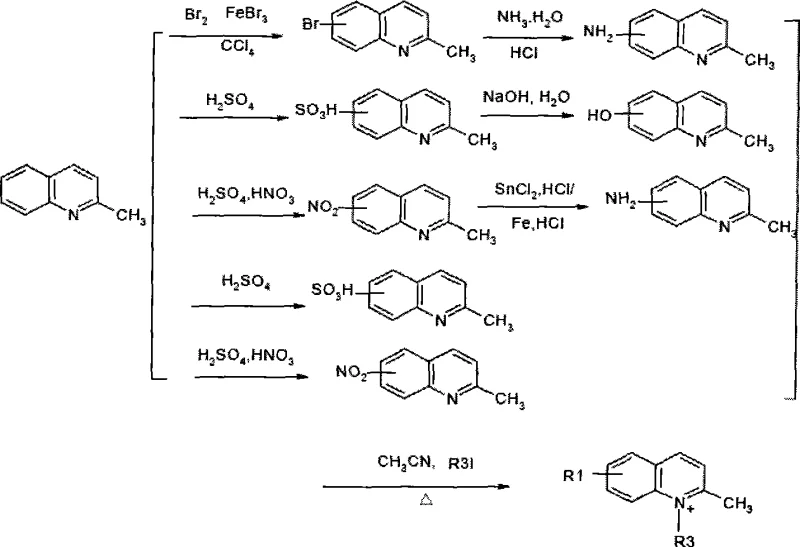
Following the formation of the quaternary salt, the core squaraine structure is assembled through a acid-catalyzed condensation with squaric acid. The reaction proceeds via the formation of a semisquaraine intermediate, which then couples with a second equivalent of the quinoline salt. The presence of electron-withdrawing or electron-donating groups at the 5 or 6 positions of the quinoline ring modulates the electron density of the entire pi-system, directly influencing the HOMO-LUMO gap and thus the emission wavelength. This mechanistic control allows for the creation of both symmetrical and asymmetrical dyes, providing chemists with the flexibility to optimize spectral properties for specific detection instruments.
Impurity control is inherently managed by the choice of reagents and the crystallization steps. The use of azeotropic distillation effectively drives the equilibrium towards the product by removing water, minimizing the formation of hydrolyzed byproducts. Furthermore, the rigidity introduced by the intramolecular hydrogen bonds between the quinoline nitrogen substituents and the squaric acid oxygen atoms restricts non-radiative decay pathways, thereby enhancing the fluorescence intensity and reducing the likelihood of photodegradation during storage or usage.
How to Synthesize Quinoline Squaraine Dye Efficiently
The synthesis protocol outlined in the patent offers a reproducible method for generating high-purity dyes suitable for research and commercial use. The process is divided into two main stages: the preparation of the functionalized quinoline quaternary salt and the subsequent condensation with squaric acid. Operators must carefully control reaction temperatures and stoichiometry to maximize yield and minimize side reactions. For detailed operational parameters, safety guidelines, and specific molar ratios required for different substituents, please refer to the standardized synthesis guide below.
- Prepare quinoline quaternary ammonium salts by reacting substituted 2-methylquinoline derivatives with iodoacids in acetonitrile under reflux.
- Condense the quaternary ammonium salts with squaric acid in a benzene and n-butanol mixture, removing water via azeotropic distillation.
- Purify the resulting dark grey to black solid dye through silica gel column chromatography and recrystallization with ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology presents a compelling value proposition by simplifying the manufacturing landscape for high-performance fluorescent dyes. The reliance on readily available starting materials like 2-methylquinoline and squaric acid eliminates the dependency on scarce or geopolitically sensitive precursors. Additionally, the absence of expensive transition metal catalysts, which are often required in cross-coupling reactions for similar fluorophores, drastically reduces the raw material costs and removes the need for complex heavy metal removal steps downstream. This streamlined process translates directly into lower production costs and a more resilient supply chain capable of meeting fluctuating market demands.
- Cost Reduction in Manufacturing: The synthetic route utilizes common organic solvents such as acetonitrile, n-butanol, and ethanol, which are inexpensive and easily sourced in bulk quantities globally. By avoiding proprietary catalysts and complex multi-step protection-deprotection sequences, the overall process mass intensity is significantly improved. This efficiency leads to substantial cost savings in terms of both material consumption and waste disposal, allowing for competitive pricing strategies in the high-value bio-reagent market without sacrificing product quality.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which primarily involve standard reflux and distillation techniques, ensures high batch-to-batch consistency. This reliability is crucial for long-term supply contracts with pharmaceutical companies that require strict adherence to quality specifications. The scalability of the process from gram-scale laboratory synthesis to multi-kilogram production is seamless, as it does not require specialized high-pressure equipment or cryogenic conditions, thereby reducing the risk of production delays and ensuring continuous availability of critical intermediates.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to traditional dye synthesis methods that often involve heavy metals or chlorinated solvents in excessive amounts. The ability to recover and recycle solvents like benzene and n-butanol through distillation further aligns with modern green chemistry principles and environmental regulations. This compliance reduces the regulatory burden on manufacturing facilities and facilitates easier permitting for capacity expansion, ensuring that supply can scale up rapidly to meet the growing demand for near-infrared imaging agents in clinical diagnostics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and application of this quinoline squaraine dye technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on performance metrics and operational feasibility. Understanding these details is essential for R&D teams evaluating the dye for specific assay development or for procurement officers assessing vendor capabilities.
Q: What is the primary advantage of quinoline-based squaraine dyes over traditional pyridine analogues?
A: Quinoline-based dyes offer extended conjugation systems that shift fluorescence emission into the near-infrared region (800-1160 nm), providing superior tissue penetrability and reduced background absorption compared to visible-light pyridine dyes.
Q: How does the introduction of carboxyl groups affect the dye's performance?
A: Introducing active carboxyl groups on the nitrogen atom enhances water solubility and allows for intramolecular hydrogen bonding with the squaric acid ring, which restricts rotation and significantly improves light stability and quantum yield.
Q: Is the synthesis process suitable for large-scale industrial production?
A: Yes, the method utilizes standard reflux conditions, common organic solvents like acetonitrile and n-butanol, and avoids expensive transition metal catalysts, making it highly scalable and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Squaraine Dye Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the synthesis of complex quinoline derivatives and squaraine dyes with stringent purity specifications, ensuring that every batch meets the rigorous demands of the pharmaceutical and biotechnology industries. With our rigorous QC labs and commitment to process optimization, we guarantee the delivery of high-purity near-infrared dyes that perform consistently in sensitive biological applications.
We invite you to collaborate with us to leverage this advanced technology for your next generation of diagnostic tools. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing expertise can accelerate your project timelines and reduce your overall cost of goods sold.
