Revolutionizing Lansoprazole Intermediate Production Through Advanced One-Step Catalytic Technology
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing critical drug intermediates, and patent CN102225909B presents a groundbreaking advancement in the production of lansoprazole precursors. This specific intellectual property details a novel preparation method for 2-chloromethyl-3-methyl-4-trifluoroethoxy pyridine, a pivotal building block in the manufacture of the second-generation proton pump inhibitor lansoprazole. Unlike traditional multi-step sequences that suffer from cumulative yield losses and excessive waste generation, this innovation achieves the transformation in a single operational step. The core chemical structure of this valuable intermediate features a pyridine ring substituted with a trifluoroethoxy group and a chloromethyl moiety, which are essential for the subsequent biological activity of the final drug substance. 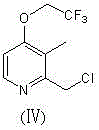 By streamlining the synthetic route, this technology addresses key pain points regarding resource utilization and environmental compliance, making it an attractive option for modern green chemistry initiatives in fine chemical manufacturing.
By streamlining the synthetic route, this technology addresses key pain points regarding resource utilization and environmental compliance, making it an attractive option for modern green chemistry initiatives in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 2-chloromethyl-3-methyl-4-trifluoroethoxy pyridine has relied on a cumbersome three-step sequence that imposes significant burdens on production efficiency and cost structures. The conventional pathway typically initiates with a rearrangement reaction using acetic anhydride to generate an acetoxy intermediate, followed by a hydrolysis step employing sodium hydroxide to yield a hydroxymethyl derivative, and finally concludes with a chlorination reaction using thionyl chloride. This fragmented approach not only extends the total processing time but also necessitates the handling of multiple hazardous reagents and the generation of substantial aqueous and organic waste streams at each stage. Furthermore, the isolation and purification required between each step lead to inevitable material losses, reducing the overall atom economy and increasing the cost of goods sold. 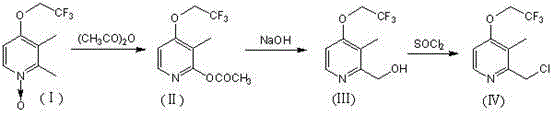 The complexity of managing three distinct reaction environments also increases the risk of operational errors and quality deviations, which can compromise the purity profile required for pharmaceutical applications.
The complexity of managing three distinct reaction environments also increases the risk of operational errors and quality deviations, which can compromise the purity profile required for pharmaceutical applications.
The Novel Approach
In stark contrast to the legacy methods, the technology disclosed in CN102225909B introduces a highly efficient one-step strategy that directly converts the N-oxide starting material into the target chloromethyl product. This innovative process utilizes phosphorus oxychloride as both the deoxygenating and chlorinating agent in the presence of triethylamine within a dichloromethane solvent system. By collapsing three discrete chemical transformations into a single reactor operation, this method drastically simplifies the workflow and eliminates the need for intermediate isolation and purification stages. The reaction proceeds under reflux conditions with nitrogen protection, ensuring that the sensitive phosphorus reagent remains active and unaffected by atmospheric moisture. 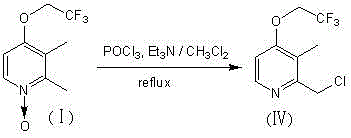 This consolidation of steps not only accelerates the production cycle but also significantly reduces the consumption of auxiliary materials and solvents, thereby aligning with the principles of sustainable manufacturing and offering a compelling value proposition for cost-conscious procurement teams.
This consolidation of steps not only accelerates the production cycle but also significantly reduces the consumption of auxiliary materials and solvents, thereby aligning with the principles of sustainable manufacturing and offering a compelling value proposition for cost-conscious procurement teams.
Mechanistic Insights into POCl3-Mediated Deoxygenation and Chlorination
The success of this one-step transformation lies in the unique dual functionality of phosphorus oxychloride when activated by a tertiary amine base under anhydrous conditions. Mechanistically, the reaction involves the simultaneous deoxygenation of the pyridine N-oxide at the 1-position and the chlorination of the methyl group at the 2-position, a feat that traditionally requires separate reagents and conditions. The triethylamine acts as a crucial acid scavenger, neutralizing the hydrogen chloride generated during the chlorination process and maintaining a weakly basic environment that facilitates the nucleophilic attack on the methyl group. This synergistic interaction allows the reaction to proceed smoothly to completion within a short timeframe, typically around one hour under reflux, achieving high conversion rates without the formation of complex by-products. The strict exclusion of water via nitrogen purging is paramount, as phosphorus oxychloride is highly susceptible to hydrolysis, which would otherwise deactivate the reagent and lower the overall yield of the desired intermediate.
From an impurity control perspective, this streamlined mechanism offers distinct advantages over the multi-step alternative by minimizing the opportunities for side reactions to occur. In traditional routes, each isolation step carries the risk of introducing contaminants or failing to remove specific impurities carried over from previous stages, leading to a complex impurity profile that is difficult to manage. The one-step process reduces the number of unit operations, thereby limiting the potential for thermal degradation or unwanted interactions with residual reagents. Moreover, the use of dichloromethane as a solvent provides excellent solubility for both the starting N-oxide and the final chloromethyl product, ensuring a homogeneous reaction mixture that promotes consistent kinetics throughout the batch. This high level of process control translates directly into a cleaner crude product, which simplifies downstream purification and ensures that the final intermediate meets the stringent purity specifications demanded by regulatory agencies for API synthesis.
How to Synthesize 2-chloromethyl-3-methyl-4-trifluoroethoxy pyridine Efficiently
Implementing this advanced synthesis route requires careful attention to reagent addition rates and temperature control to maximize safety and yield. The standardized protocol involves dissolving the N-oxide precursor in dichloromethane under a protective nitrogen atmosphere, followed by the controlled dropwise addition of the phosphorus oxychloride solution to manage exothermic potential. Once the initial mixing is complete, the system is heated to reflux, and triethylamine is introduced slowly to drive the reaction forward while scavenging acidic by-products. Detailed standard operating procedures regarding specific molar ratios, addition times, and workup protocols are essential for reproducible results in a GMP environment.
- Dissolve 2,3-dimethyl-4-trifluoroethoxypyridine-N-oxide in dichloromethane under nitrogen protection and add phosphorus oxychloride solution dropwise.
- Heat the reaction mixture to reflux and slowly introduce triethylamine to maintain weak base conditions while facilitating the reaction.
- After refluxing for one hour, wash the reaction liquid with saturated sodium bicarbonate, extract, dry, and concentrate to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this one-step synthesis technology represents a strategic opportunity to optimize cost structures and enhance supply reliability. By eliminating two entire reaction steps and their associated workups, the process inherently reduces the consumption of raw materials, solvents, and energy, leading to substantial cost savings in the manufacturing of pharmaceutical intermediates. The simplified workflow also means shorter production cycles, allowing manufacturers to respond more rapidly to market demand fluctuations and reduce inventory holding costs. Furthermore, the reduction in waste generation lowers the burden on environmental treatment facilities, mitigating regulatory risks and disposal expenses that often impact the bottom line in fine chemical production. These efficiencies collectively contribute to a more competitive pricing model without compromising on the quality or integrity of the supply chain.
- Cost Reduction in Manufacturing: The consolidation of three reaction steps into a single operation fundamentally alters the cost equation by removing the need for multiple sets of reagents, extensive solvent exchanges, and intermediate quality control testing. This reduction in process complexity directly translates to lower operational expenditures, as fewer man-hours are required for monitoring and handling, and less equipment time is consumed per batch. Additionally, the avoidance of expensive and hazardous reagents like thionyl chloride in a separate step further drives down material costs, while the improved yield minimizes the loss of valuable starting materials. These factors combine to create a robust economic advantage that can be passed down the supply chain, offering better value to downstream API manufacturers.
- Enhanced Supply Chain Reliability: A shorter and simpler synthetic route inherently reduces the number of potential failure points within the production process, thereby increasing the overall reliability of supply. With fewer unit operations, there is less chance of equipment downtime or batch failures due to procedural errors, ensuring a more consistent output of high-quality intermediate. The use of commercially available and stable reagents like phosphorus oxychloride and triethylamine also mitigates the risk of raw material shortages that can plague more specialized chemical supply chains. This stability is crucial for long-term planning and securing uninterrupted production schedules for critical pharmaceutical ingredients.
- Scalability and Environmental Compliance: The design of this one-step process is inherently scalable, utilizing standard reflux conditions and common solvents that are easily managed in large-scale reactors. The significant reduction in "three wastes" (waste water, waste gas, and solid waste) aligns perfectly with increasingly strict global environmental regulations, reducing the compliance burden on manufacturing sites. By minimizing the environmental footprint, companies can operate more sustainably and avoid potential fines or shutdowns related to pollution control, ensuring long-term viability and social responsibility in their operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this technology can be integrated into existing manufacturing frameworks. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: How does the new one-step method compare to traditional three-step processes?
A: The new method consolidates rearrangement, hydrolysis, and chlorination into a single reaction step using POCl3 and Et3N, significantly reducing reaction time, solvent consumption, and waste discharge compared to the conventional multi-step route involving acetic anhydride and thionyl chloride.
Q: What are the critical reaction conditions for maintaining high yield?
A: Maintaining a strict nitrogen atmosphere is crucial to prevent phosphorus oxychloride from reacting with moisture. Additionally, controlling the molar ratio of triethylamine to the starting oxide between 0.9:1 and 1.6:1 ensures optimal deoxygenation and chlorination efficiency.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes common solvents like dichloromethane and standard reflux conditions, making it highly scalable. The reduction in operational steps and simplified post-treatment workup greatly enhances its feasibility for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-chloromethyl-3-methyl-4-trifluoroethoxy pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and reliable intermediate supply chains for the global pharmaceutical industry. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of volume. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-chloromethyl-3-methyl-4-trifluoroethoxy pyridine meets the highest standards required for API synthesis. We are committed to leveraging advanced technologies like the one described in CN102225909B to deliver superior value and performance to our partners.
We invite you to collaborate with us to explore how this optimized synthesis route can benefit your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your decision-making process and help you secure a competitive edge in the market.
