Streamlining Lansoprazole Intermediate Production via One-Step Deoxygenative Chlorination
Introduction to Patent CN102225909B: A Paradigm Shift in Proton Pump Inhibitor Synthesis
The global demand for high-quality proton pump inhibitors (PPIs) continues to drive innovation in the synthesis of their critical precursors. Patent CN102225909B introduces a groundbreaking methodology for the preparation of 2-chloromethyl-3-methyl-4-trifluoroethoxypyridine, a pivotal intermediate in the manufacturing of Lansoprazole. This specific chemical entity serves as the foundational scaffold upon which the therapeutic efficacy of the final drug depends. Historically, the synthesis of this pyridine derivative has been plagued by inefficiencies, requiring multiple discrete reaction vessels and extensive purification protocols. The technical disclosure within this patent represents a significant leap forward, transitioning from a cumbersome multi-step sequence to a highly efficient one-pot transformation. By leveraging the dual reactivity of phosphorus oxychloride in the presence of a tertiary amine base, the inventors have achieved simultaneous deoxygenation of the pyridine N-oxide and chlorination of the adjacent methyl group. This consolidation not only simplifies the operational workflow but also aligns with modern green chemistry principles by substantially reducing the generation of hazardous waste streams. For R&D directors and process chemists, this patent offers a robust alternative to legacy methods, promising enhanced throughput and superior cost-efficiency in the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovation described in CN102225909B, the industrial standard for synthesizing 2-chloromethyl-3-methyl-4-trifluoroethoxypyridine relied heavily on a linear three-step sequence, as detailed in earlier literature such as CN101137371A. This traditional pathway begins with a rearrangement reaction involving acetic anhydride to generate an acetate intermediate, followed by a hydrolysis step using sodium hydroxide to yield the corresponding alcohol. The final transformation requires chlorination using thionyl chloride to install the crucial chloromethyl functionality. Each of these stages necessitates separate reaction setups, distinct work-up procedures, and often, the isolation and drying of intermediate solids. This fragmented approach inherently accumulates material losses at every transfer point, leading to diminished overall yields and increased consumption of raw materials. Furthermore, the reliance on acetic anhydride and thionyl chloride introduces significant safety and environmental challenges, including the management of corrosive byproducts and acidic off-gases. The cumulative effect of these operational burdens results in prolonged production cycles and elevated manufacturing costs, creating a bottleneck for suppliers aiming to meet the rigorous demands of the global API market.
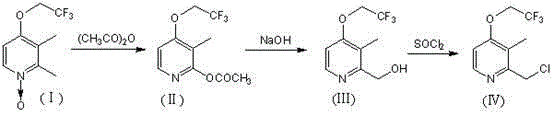
The Novel Approach
In stark contrast to the legacy protocols, the novel approach disclosed in this patent streamlines the entire synthetic trajectory into a single, cohesive reaction step. By utilizing 2,3-dimethyl-4-trifluoroethoxypyridine-N-oxide as the starting material, the process bypasses the need for intermediate isolation entirely. The key to this efficiency lies in the strategic selection of phosphorus oxychloride (POCl3) as the chlorinating and deoxygenating agent, facilitated by triethylamine as an acid scavenger. This reagent combination allows for the direct conversion of the N-oxide to the desired chloromethyl pyridine without traversing the unstable alcohol or acetate states. The reaction proceeds smoothly in dichloromethane under reflux conditions, demonstrating remarkable tolerance and selectivity. This consolidation of chemical transformations eliminates the logistical complexity of multi-stage processing, thereby reducing the footprint of the manufacturing facility and minimizing the potential for human error during scale-up. For procurement managers, this translates to a more reliable supply chain with reduced dependency on a wide array of specialized reagents, ultimately driving down the cost of goods sold through operational simplification.
Mechanistic Insights into POCl3-Mediated Deoxygenative Chlorination
The core chemical innovation of this patent rests on the unique ability of phosphorus oxychloride to act as both a deoxygenating agent for the pyridine N-oxide moiety and a chlorinating source for the benzylic-like methyl position. Mechanistically, the reaction initiates with the activation of the N-oxide oxygen by the electrophilic phosphorus center, forming a reactive phosphate intermediate. This activation weakens the N-O bond, facilitating its cleavage. Concurrently, the chlorine atoms on the phosphorus species participate in a nucleophilic substitution or radical-type mechanism at the 2-position methyl group, replacing a hydrogen atom with a chlorine atom. The presence of triethylamine is critical in this cycle; it serves to neutralize the hydrochloric acid generated during the chlorination event, preventing the protonation of the pyridine nitrogen which could otherwise deactivate the ring towards further reaction or lead to polymerization. The molar ratio of triethylamine to the starting N-oxide is optimized between 0.9:1 and 1.6:1, with a preferred stoichiometry of 1.1:1 ensuring complete acid scavenging without excessive reagent waste. This precise balancing of reagents ensures that the reaction environment remains sufficiently basic to drive the equilibrium forward while avoiding the degradation of sensitive functional groups.
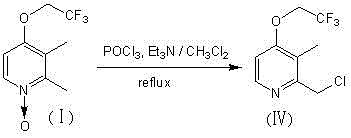
Controlling the impurity profile in this reaction is paramount for meeting the stringent quality standards required for pharmaceutical intermediates. The one-step nature of the process inherently limits the formation of byproducts that typically arise from the handling and storage of reactive intermediates in multi-step syntheses. For instance, the avoidance of the free alcohol intermediate prevents potential dehydration or etherification side reactions that could complicate downstream purification. Additionally, the use of nitrogen protection throughout the reaction is a critical control parameter, as phosphorus oxychloride is highly susceptible to hydrolysis by atmospheric moisture. By rigorously excluding water, the process maintains the integrity of the chlorinating agent, ensuring high conversion rates and minimizing the formation of phosphoric acid derivatives that could contaminate the final product. The experimental data supports this mechanistic understanding, showing that under optimized conditions, the reaction achieves yields as high as 90%, indicating a highly selective transformation with minimal diversion of starting material into waste streams.
How to Synthesize 2-Chloromethyl-3-methyl-4-trifluoroethoxypyridine Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the specified operational parameters to maximize yield and safety. The process begins with the dissolution of the N-oxide starting material in anhydrous dichloromethane under an inert nitrogen atmosphere. Phosphorus oxychloride is introduced as a solution in the same solvent to ensure controlled mixing and heat dissipation. Following the initial addition, the mixture is heated to reflux, and triethylamine is added slowly to manage the exotherm associated with acid neutralization. The reaction is maintained at reflux for approximately one hour to ensure complete conversion. Post-reaction work-up involves a careful quench with saturated sodium bicarbonate solution to neutralize residual acids, followed by extraction and drying. The crude product is then concentrated under reduced pressure to yield the target oil. For detailed standard operating procedures and specific equipment requirements, please refer to the technical guide below.
- Dissolve 2,3-dimethyl-4-trifluoroethoxypyridine-N-oxide in dichloromethane under nitrogen protection.
- Add phosphorus oxychloride solution dropwise at room temperature, then heat to reflux.
- Slowly introduce triethylamine, maintain reflux for 1 hour, then work up with bicarbonate wash and distillation.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this one-step synthetic route offers profound commercial benefits that extend far beyond the laboratory bench, directly impacting the bottom line for pharmaceutical manufacturers and their supply chain partners. By collapsing a three-step sequence into a single operation, the process dramatically reduces the total processing time, allowing for faster turnover of reactor vessels and increased annual production capacity without the need for capital investment in new infrastructure. This efficiency gain is particularly valuable in the context of tight supply markets for PPI intermediates, where speed to market can determine competitive advantage. Furthermore, the reduction in unit operations significantly lowers the consumption of utilities such as steam for heating and cooling water for condensation, contributing to a smaller carbon footprint and lower energy costs per kilogram of product. The simplified workflow also reduces the labor hours required for monitoring and handling, freeing up skilled personnel for other critical tasks within the facility.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps removes the need for filtration, drying, and re-dissolution processes, which are often the most resource-intensive parts of batch manufacturing. This leads to substantial savings in solvent usage, as the same batch of dichloromethane can potentially be recovered and recycled more efficiently in a continuous or semi-continuous loop. Additionally, the high yield of 90% reported in the patent examples means that less raw material is required to produce the same amount of final product, directly lowering the variable cost of production. The removal of expensive and hazardous reagents like thionyl chloride from the process further reduces procurement costs and the expenses associated with hazardous waste disposal and regulatory compliance.
- Enhanced Supply Chain Reliability: A shorter synthetic route inherently reduces the number of potential failure points in the manufacturing process. In a traditional three-step synthesis, a yield loss or quality issue at any stage can compromise the entire batch, leading to delays and supply shortages. The robustness of the one-step POCl3 method mitigates this risk, providing a more predictable and stable output. Moreover, the reagents used—phosphorus oxychloride and triethylamine—are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages compared to more specialized reagents. This reliability ensures consistent delivery schedules for downstream API manufacturers, fostering stronger long-term partnerships and trust.
- Scalability and Environmental Compliance: The simplicity of the reaction conditions makes this process highly amenable to scale-up from kilogram to multi-ton production. The use of standard glass-lined or stainless steel reactors compatible with dichloromethane and mild bases means that existing infrastructure can be utilized without major modifications. From an environmental perspective, the reduction in 'three wastes' (waste water, waste gas, and solid waste) is a significant advantage. By avoiding the generation of acetate salts and sulfur dioxide byproducts associated with the old method, the facility can operate with a lighter environmental burden, simplifying permitting and reducing the costs of effluent treatment. This alignment with green chemistry principles enhances the corporate social responsibility profile of the manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and technical specifications outlined in CN102225909B, providing clarity for stakeholders evaluating the feasibility of this route for their specific applications. Understanding these nuances is essential for making informed decisions about process adoption and supplier selection.
Q: What is the primary advantage of the POCl3 method over the traditional acetic anhydride route?
A: The POCl3 method consolidates three distinct reaction steps (rearrangement, hydrolysis, chlorination) into a single operational unit, drastically reducing processing time and solvent consumption.
Q: How does the new process impact impurity profiles?
A: By eliminating intermediate isolation steps and avoiding harsh hydrolysis conditions, the one-step approach minimizes the formation of side-products associated with multi-step handling, leading to a cleaner crude profile.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of common solvents like dichloromethane and standard reagents like triethylamine, combined with high yields up to 90%, makes it highly scalable for commercial API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloromethyl-3-methyl-4-trifluoroethoxypyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic methodologies like the one described in CN102225909B requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO and supplier in the fine chemical sector, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art reaction systems capable of handling corrosive reagents like phosphorus oxychloride safely and efficiently, ensuring that the theoretical benefits of this one-step process are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-chloromethyl-3-methyl-4-trifluoroethoxypyridine meets the exacting standards required for GMP-grade API synthesis. Our commitment to quality assurance ensures that your downstream processes remain uninterrupted and compliant with global regulatory requirements.
We invite you to collaborate with us to leverage this innovative technology for your Lansoprazole supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this optimized route can improve your margins. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you secure not just a chemical supplier, but a strategic ally dedicated to driving efficiency and excellence in pharmaceutical manufacturing.
