Advanced Biocatalytic Resolution for High-Purity D-Alpha-Hydroxybutyric Acid Production
The pharmaceutical and fine chemical industries are constantly seeking more efficient routes to chiral building blocks, and the production of D-alpha-hydroxybutyric acid stands as a prime example of this technological evolution. Patent CN101974603B introduces a groundbreaking biocatalytic method that leverages the unique properties of NAD-independent hydroxyacid dehydrogenases found in Pseudomonas species. Unlike traditional chemical synthesis or cofactor-dependent enzymatic routes, this innovation utilizes a clever thermal inactivation strategy to achieve high enantiomeric excess without the burden of expensive electron acceptors. For R&D directors and procurement specialists alike, this represents a paradigm shift towards more sustainable and cost-effective manufacturing of critical intermediates used in antibiotic synthesis and biodegradable polymers. The method not only simplifies the upstream fermentation requirements but also streamlines the downstream purification process, making it an ideal candidate for large-scale industrial adoption by a reliable pharmaceutical intermediate supplier.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of high-purity D-alpha-hydroxybutyric acid has been plagued by significant economic and technical hurdles that hindered its widespread application. Conventional enzymatic reduction methods typically rely on D-lactate dehydrogenase, which necessitates the continuous addition of expensive cofactors like NADH to drive the reaction forward. This dependency creates a massive cost bottleneck, as regenerating these cofactors requires complex auxiliary enzyme systems or stoichiometric amounts of reducing agents, rendering the process economically unviable for bulk manufacturing. Furthermore, alternative resolution strategies using lipases often suffer from low substrate tolerance, typically operating at concentrations around 1 mmol/L, which results in poor volumetric productivity and massive reactor volumes. These inefficiencies translate directly into higher operational expenditures and extended lead times, creating a fragile supply chain that struggles to meet the rigorous demands of modern API manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN101974603B circumvents these limitations by exploiting the differential thermal stability of isozymes within Pseudomonas whole cells. By subjecting the crude biocatalyst to a controlled heat treatment between 45°C and 65°C, the process selectively inactivates the unwanted D-hydroxyacid dehydrogenase while preserving the activity of the L-specific enzyme. This thermal switch allows the L-enzyme to exclusively oxidize the L-isomer of the racemic substrate into alpha-ketobutyric acid, leaving the desired D-alpha-hydroxybutyric acid untouched in the reaction mixture. This approach eliminates the need for external cofactors entirely, as the enzymes are NAD-independent, and supports significantly higher substrate concentrations up to 800 mmol/L. The result is a robust, high-yield process that drastically reduces raw material costs and simplifies the overall process flow, offering a compelling value proposition for cost reduction in API manufacturing.
Mechanistic Insights into Thermal Selective Inactivation
The core of this technological breakthrough lies in the precise manipulation of enzyme kinetics and protein stability to achieve kinetic resolution without genetic engineering. The Pseudomonas strains utilized, such as Pseudomonas putida or Pseudomonas stutzeri, naturally express both L- and D-hydroxyacid dehydrogenases. Under normal physiological conditions, these enzymes would act on both enantiomers, leading to a racemic mixture. However, the patent reveals that the D-isozyme possesses a lower thermal stability threshold compared to its L-counterpart. By incubating the whole cell suspension or crude enzyme extract at temperatures ranging from 50°C to 60°C for 10 to 30 minutes, the tertiary structure of the D-enzyme is irreversibly denatured. This leaves a biocatalyst that is functionally mono-specific for the L-enantiomer. When introduced to racemic alpha-hydroxybutyrate, the surviving L-enzyme catalyzes the oxidation of the L-form to alpha-ketobutyrate, effectively removing it from the equilibrium and driving the resolution towards the D-product.
Quality control in this process is paramount, and the enantiomeric excess (ee) serves as the critical metric for success. The patent specifies rigorous HPLC analysis using chiral columns to quantify the ratio of D- to L-isomers, ensuring the final product meets the stringent purity requirements for pharmaceutical applications. The calculation of enantiomeric excess is fundamental to validating the efficacy of the thermal treatment step.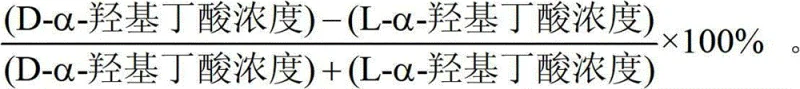 As demonstrated in the experimental data, optimizing the heat treatment parameters can push the ee value above 99%, indicating near-perfect stereochemical control. This level of purity is essential for downstream applications, particularly in the synthesis of azinothricin family antibiotics, where impurities can compromise biological activity. The mechanism ensures that the byproduct, alpha-ketobutyric acid, is also generated in high yield, providing a secondary revenue stream and enhancing the overall atom economy of the process.
As demonstrated in the experimental data, optimizing the heat treatment parameters can push the ee value above 99%, indicating near-perfect stereochemical control. This level of purity is essential for downstream applications, particularly in the synthesis of azinothricin family antibiotics, where impurities can compromise biological activity. The mechanism ensures that the byproduct, alpha-ketobutyric acid, is also generated in high yield, providing a secondary revenue stream and enhancing the overall atom economy of the process.
How to Synthesize D-Alpha-Hydroxybutyric Acid Efficiently
Implementing this synthesis route requires careful attention to the preparation of the biocatalyst and the subsequent resolution conditions to maximize yield and purity. The process begins with the fermentation of selected Pseudomonas strains to accumulate the necessary dehydrogenase activity, followed by the critical thermal inactivation step. Once the selective biocatalyst is prepared, it is mixed with the racemic substrate under controlled pH and temperature conditions to facilitate the resolution. The detailed standardized synthesis steps see the guide below.
- Cultivate Pseudomonas strains to express NAD-independent hydroxyacid dehydrogenase and harvest whole cells.
- Apply controlled heat denaturation (45-65°C) to selectively inactivate D-dehydrogenase while preserving L-dehydrogenase activity.
- Resolve racemic substrate using the treated biocatalyst, followed by ion exchange separation and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this NAD-independent enzymatic resolution offers substantial strategic benefits beyond mere technical feasibility. The elimination of expensive cofactors and the ability to use cheaper racemic substrates instead of pure precursors fundamentally alters the cost structure of production. This shift allows for significant margin improvements and protects against volatility in the pricing of specialized reagents. Furthermore, the robustness of the whole-cell system means that the process is less sensitive to minor fluctuations in operating conditions, ensuring consistent batch-to-batch quality and reliability. This stability is crucial for maintaining long-term supply contracts and meeting the just-in-time delivery requirements of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the removal of NADH dependency, which traditionally accounts for a significant portion of enzymatic process costs. By utilizing NAD-independent enzymes, the process avoids the capital and operational expenses associated with cofactor regeneration systems. Additionally, the ability to operate at high substrate concentrations reduces the volume of water and solvents required for downstream processing, leading to lower utility costs and waste treatment fees. The dual production of alpha-ketobutyric acid further amortizes the production costs across two valuable products, enhancing the overall economic efficiency of the facility.
- Enhanced Supply Chain Reliability: The use of commercially available Pseudomonas strains and standard fermentation media ensures that the raw material supply is secure and not subject to the bottlenecks often seen with proprietary genetically modified organisms. The simplicity of the downstream processing, which relies on standard ion exchange resins rather than complex chromatography, means that the technology can be easily transferred to multiple manufacturing sites. This flexibility reduces the risk of supply disruption and allows for rapid scaling in response to market demand, ensuring that customers receive their high-purity intermediates without delay.
- Scalability and Environmental Compliance: From an environmental perspective, this biocatalytic route is inherently greener than chemical alternatives, operating under mild aqueous conditions without the need for hazardous organic solvents or heavy metal catalysts. The high conversion rates minimize the amount of unreacted starting material that needs to be recycled or disposed of, aligning with strict environmental regulations. The process is designed for scalability, with parameters optimized for large-volume reactors, making it suitable for commercial scale-up of complex intermediates. This alignment with green chemistry principles not only reduces regulatory risk but also enhances the corporate sustainability profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic resolution technology. These answers are derived directly from the patent specifications and are intended to clarify the operational advantages and quality standards associated with this method. Understanding these details is essential for partners evaluating the feasibility of integrating this intermediate into their own synthesis pipelines.
Q: Why is the NAD-independent feature critical for cost reduction?
A: Traditional methods often require expensive cofactors like NADH. This patent utilizes NAD-independent enzymes, eliminating the need for costly cofactor regeneration systems and significantly lowering raw material costs.
Q: How does the thermal treatment improve enantiomeric purity?
A: By heating the crude enzyme or whole cells to 45-65°C, the unstable D-hydroxyacid dehydrogenase is selectively denatured. This leaves only the L-enzyme active to consume the L-isomer, ensuring the remaining product is highly enriched D-alpha-hydroxybutyric acid.
Q: What are the scalability advantages of this whole-cell method?
A: The process uses robust Pseudomonas strains that tolerate high substrate concentrations (up to 800 mM). Combined with simple downstream processing like ion exchange, it allows for efficient commercial scale-up without complex extraction steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-Alpha-Hydroxybutyric Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics and advanced materials. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical equipment to verify every batch. Our dedication to technical excellence ensures that we can support your most demanding projects with reliability and precision.
We invite you to collaborate with us to explore how this advanced enzymatic resolution technology can optimize your supply chain and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our D-alpha-hydroxybutyric acid can serve as a cornerstone for your successful product development.
