Advanced Rhodium-Catalyzed Synthesis of Silicon-Centered Chiral Aryl Silanes for Commercial Scale-Up
The pharmaceutical and advanced materials industries are constantly seeking robust methods to access novel chiral scaffolds, particularly those involving silicon centers which offer unique stereoelectronic properties. Patent CN114478608A introduces a groundbreaking approach to synthesizing silicon-centered chiral aryl silanes through an enantioselective intermolecular C-H silylation reaction. This technology represents a significant leap forward from previous limitations, utilizing a rhodium-catalyzed system to directly functionalize Si-H bonds with high precision. The general structure of these valuable compounds is depicted below, showcasing the versatility of the R1, R2, and Ar substituents which can be tailored for specific applications in organic electrophosphorescent devices or as key pharmaceutical intermediates.
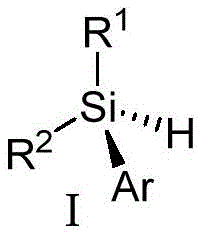
For procurement managers and supply chain heads, the ability to source reliable silicon-centered chiral aryl silane supplier products is critical for maintaining production continuity. This patent outlines a pathway that not only expands the chemical space available for R&D teams but also offers a streamlined manufacturing route. By avoiding the inefficiencies of traditional resolution methods, this process supports cost reduction in fine chemical intermediates manufacturing while ensuring high purity standards. The following analysis details the technical merits and commercial viability of adopting this catalytic strategy for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of chiral organosilanes has relied heavily on optical and kinetic resolution techniques involving the addition of stoichiometric chiral auxiliaries. These conventional methods suffer from inherent inefficiencies, primarily because the maximum theoretical yield is capped at 50%, leading to significant material waste and increased disposal costs. Furthermore, the separation of enantiomers often requires complex chromatographic processes that are difficult to scale economically. In the context of intermolecular reactions, prior art struggled with low reactivity and poor regioselectivity during C-H bond activation, making it exceptionally difficult to form the necessary acyclic Si-metal intermediates without generating substantial byproducts. The lack of effective methods to control stereoselectivity in flexible acyclic systems further hampered the development of diverse silicon-centered chiral libraries.
The Novel Approach
The methodology disclosed in CN114478608A overcomes these hurdles by employing a transition metal-catalyzed desymmetrization of dihydrosilanes. This strategy selectively functionalizes one Si-H bond of a dihydrosilane to generate a silicon-centered chiral monohydrosilane directly. The reaction utilizes a chiral rhodium catalyst system that facilitates an intermolecular Si-H/C-H cross-coupling with exceptional chemical and stereocontrol. As illustrated in the reaction scheme below, the process couples a dihydrosilane with an arene or heteroarene in the presence of a hydrogen acceptor. This approach eliminates the 50% yield ceiling associated with resolution and provides a direct, atom-economical route to high-value chiral silanes that were previously inaccessible or prohibitively expensive to produce.
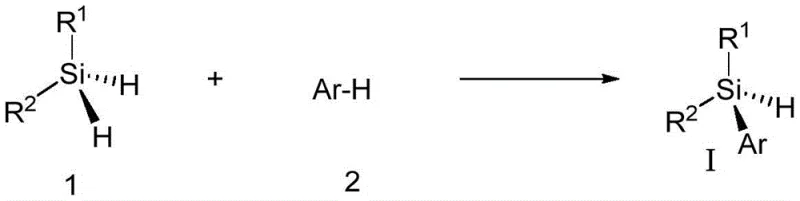
Mechanistic Insights into Rh-Catalyzed Enantioselective Silylation
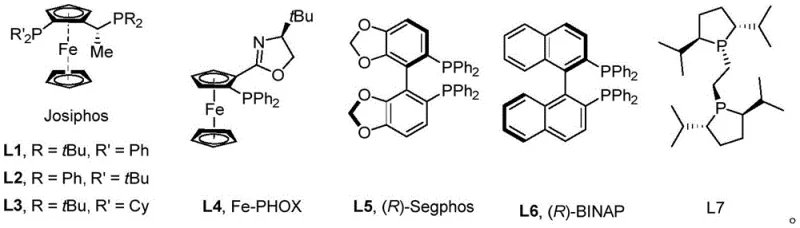
The success of this transformation hinges on the precise interaction between the rhodium catalyst and the chiral ligand environment. The inventors speculate that the introduction of sterically hindered groups, such as the tert-butyl group on the dihydrosilane substrate, plays a pivotal role in enhancing stereoselective control during the oxidative addition of the Si-H bond. This steric bulk helps to suppress competitive side reactions, such as the formation of Si-Si or Si-C byproducts, which often plague silane chemistry. Moreover, the bulky group stabilizes the newly formed chiral center, preventing racemization which is a common issue with flexible acyclic arylsilane products. The choice of ligand is equally critical, with Josiphos-type ligands demonstrating superior performance in inducing asymmetry compared to other bisphosphine systems.
To achieve the high enantiomeric excess (ee) values reported, ranging from 80% to 90% for many substrates, the catalyst system must effectively differentiate between the two enantiotopic Si-H bonds. The reaction mechanism likely proceeds through a rhodium-silyl intermediate that undergoes selective C-H activation of the aromatic substrate. The presence of a hydrogen acceptor, such as norbornene derivatives (NBE-OMe), is essential to drive the equilibrium forward by removing hydrogen from the system. The structural diversity of the ligands tested, including Fe-PHOX and BINAP derivatives as shown below, highlights the sensitivity of the reaction to the electronic and steric properties of the phosphine environment, allowing for fine-tuning based on specific substrate requirements.
How to Synthesize Silicon-Centered Chiral Aryl Silanes Efficiently
The synthesis protocol described in the patent provides a robust framework for producing these complex molecules under relatively mild conditions. The process typically involves charging a reaction vessel with the dihydrosilane substrate, the heterocyclic or aromatic coupling partner, the rhodium catalyst precursor, and the optimized chiral ligand in an anhydrous solvent like toluene. A crucial operational detail is the addition of a hydrogen acceptor to facilitate the dehydrogenative coupling. The reaction is conducted under an inert atmosphere to prevent catalyst deactivation and is heated to moderate temperatures, typically around 40°C, for a period of 24 hours. Following the reaction, standard workup procedures involving solvent evaporation and silica gel column chromatography yield the pure chiral product. For detailed standardized synthesis steps, please refer to the guide below.
- Prepare the reaction mixture by combining dihydrosilane substrate, heterocyclic substrate, Rhodium catalyst, and chiral ligand in anhydrous toluene under argon.
- Add a hydrogen acceptor such as NBE-OMe to the system to drive the dehydrogenative coupling forward efficiently.
- Stir the mixture at 40°C for 24 hours, then purify the resulting chiral monohydrosilane via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this catalytic technology offers substantial strategic benefits for supply chain management and cost optimization. By shifting from resolution-based methods to direct asymmetric synthesis, manufacturers can theoretically double their output from the same amount of starting material, significantly improving material efficiency. This reduction in waste generation aligns with modern environmental compliance standards and reduces the burden on waste treatment facilities. Furthermore, the use of commercially available rhodium catalysts and ligands ensures that the supply chain remains resilient, as these reagents are sourced from established global suppliers. The mild reaction conditions also translate to lower energy consumption compared to high-temperature or high-pressure alternatives, contributing to overall operational cost savings.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chiral auxiliaries and the avoidance of 50% yield loss inherent in resolution processes lead to drastic improvements in cost efficiency. By maximizing atom economy, the cost per kilogram of the final chiral silane is significantly lowered, making high-purity silicon-centered chiral aryl silanes more accessible for large-scale applications. Additionally, the simplified purification process reduces solvent usage and processing time, further driving down manufacturing expenses without compromising on quality.
- Enhanced Supply Chain Reliability: The substrates required for this synthesis, such as dihydrosilanes and various heteroarenes, are readily available from multiple chemical vendors, reducing the risk of single-source bottlenecks. The robustness of the catalytic system against varying electronic properties of substrates means that supply chain disruptions for specific precursors can be mitigated by switching to alternative substituted analogs without re-optimizing the entire process. This flexibility ensures consistent production schedules and reliable delivery timelines for downstream customers.
- Scalability and Environmental Compliance: The reaction operates at moderate temperatures and atmospheric pressure, which simplifies the engineering requirements for scaling up from laboratory to commercial production. The use of toluene as a solvent is well-established in industrial settings with existing recovery infrastructure. Moreover, the high selectivity of the reaction minimizes the formation of hazardous byproducts, easing the regulatory burden associated with waste disposal and ensuring that the manufacturing process meets stringent environmental safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy for R&D and technical procurement teams evaluating this technology for their specific projects.
Q: What is the primary advantage of this Rh-catalyzed method over traditional resolution?
A: Unlike traditional optical resolution which limits theoretical yield to 50%, this catalytic asymmetric desymmetrization allows for direct construction of the silicon center with high atom economy and superior enantioselectivity.
Q: What types of substrates are compatible with this synthesis protocol?
A: The method supports a broad scope including furans, thiophenes, benzofurans, and various substituted benzenes, tolerating electron-withdrawing and electron-donating groups effectively.
Q: How is stereocontrol achieved in this intermolecular reaction?
A: Stereocontrol is managed through the use of bulky groups like tert-butyl on the dihydrosilane and specific chiral ligands such as Josiphos, which prevent racemization and guide selective Si-H bond activation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Silicon-Centered Chiral Aryl Silane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this rhodium-catalyzed technology in advancing the field of chiral silicon materials. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market-ready product is seamless. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications and enantiomeric excess values, guaranteeing that every batch meets the high standards required for pharmaceutical and electronic material applications.
We invite you to collaborate with us to leverage this innovative synthesis route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the supply of high-purity silicon-centered chiral aryl silanes.
