Scalable Synthesis of p-Bromomethyl Isophenylpropionic Acid via Styrene Route for Loxoprofen Production
Introduction to Advanced Loxoprofen Intermediate Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways for non-steroidal anti-inflammatory drug (NSAID) intermediates, particularly for high-demand molecules like Loxoprofen Sodium. A pivotal advancement in this domain is detailed in patent CN111454145A, which discloses a novel preparation method for p-bromomethyl isophenylpropionic acid. This compound serves as a critical precursor in the synthesis of Loxoprofen Sodium, a potent analgesic and anti-inflammatory agent widely utilized in clinical settings across Japan, Korea, Europe, and America. The significance of this patent lies in its strategic departure from traditional, cost-prohibitive raw materials towards a more economically viable and industrially scalable approach. By leveraging styrene, a ubiquitous petrochemical feedstock, the disclosed methodology addresses longstanding supply chain bottlenecks associated with specialized starting materials. This report provides a deep technical and commercial analysis of this synthesis route, offering valuable insights for R&D directors seeking process optimization, procurement managers aiming for cost reduction, and supply chain leaders focused on continuity and scalability in the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing p-bromomethyl isophenylpropionic acid have historically relied on 4-methyl styrene as the primary starting material. While chemically feasible, this conventional approach presents severe logistical and economic challenges for large-scale manufacturing. The primary drawback is the scarcity and high cost of high-purity 4-methyl styrene, which is not as readily available in the global chemical market as basic styrene. Furthermore, the synthetic route originating from 4-methyl styrene often involves complex halogenation sequences that generate significant quantities of undesirable byproducts, specifically dibromo derivatives. As illustrated in the reaction scheme below, the conventional pathway frequently results in a mixture containing the target mono-brominated compound alongside a di-brominated impurity (Formula VI), necessitating an additional, yield-reducing debromination step to achieve pharmaceutical-grade purity. This elongated process flow not only increases operational complexity but also amplifies waste generation and solvent consumption, rendering it less attractive for modern green chemistry standards and cost-sensitive commercial production environments.
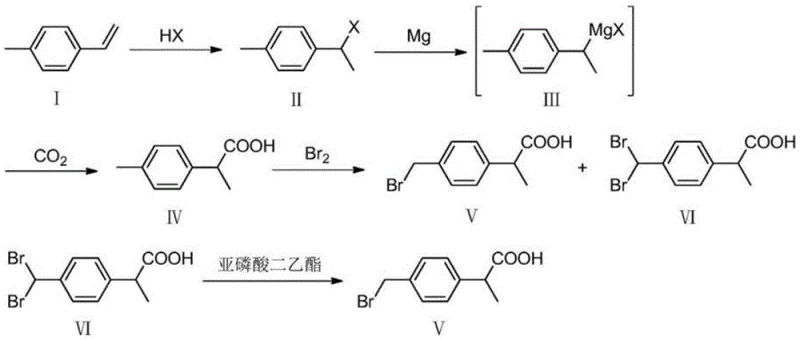
The Novel Approach
In stark contrast to the limitations of the prior art, the novel approach described in CN111454145A fundamentally re-engineers the synthesis backbone by utilizing styrene as the foundational raw material. This strategic shift capitalizes on the abundant availability and low cost of styrene, effectively decoupling the production of this key intermediate from the volatile supply dynamics of specialized vinyl aromatics. The new route streamlines the synthesis into three distinct, high-efficiency stages: hydrochlorination, Grignard carboxylation, and bromomethylation. By constructing the carbon skeleton first through a robust Grignard reaction and introducing the bromomethyl functionality in the final step via electrophilic aromatic substitution, the process achieves superior regioselectivity. This sequence eliminates the need for the problematic debromination step required in older methods, thereby shortening the overall production timeline and reducing the cumulative loss of material. The result is a streamlined, linear process that is inherently more amenable to continuous improvement and scale-up, offering a compelling value proposition for manufacturers aiming to optimize their production of Loxoprofen precursors.
Mechanistic Insights into Grignard Carboxylation and Bromomethylation
The core of this synthetic innovation lies in the precise execution of the Grignard carboxylation and the subsequent bromomethylation steps. In the second stage, α-methyl benzyl chloride is converted into its corresponding Grignard reagent using magnesium metal in 2-methyltetrahydrofuran (2-MeTHF). The choice of 2-MeTHF is critical; it offers excellent solvating power for organomagnesium species while providing a higher boiling point and better stability compared to traditional diethyl ether, facilitating safer operation at the required reaction temperatures of 0-30°C. Upon formation of the Grignard reagent, carbon dioxide gas is introduced to effect carboxylation. This step builds the propionic acid side chain with high fidelity. The subsequent hydrolysis using dilute hydrochloric acid cleanly liberates the free acid, 2-phenylpropionic acid, which is then isolated via high-vacuum rectification to ensure exceptional purity before entering the final stage. This meticulous control over the organometallic step prevents the formation of homocoupling byproducts, ensuring a clean substrate for the final functionalization.
The final transformation involves the bromomethylation of 2-phenylpropionic acid using paraformaldehyde and hydrobromic acid, catalyzed by concentrated sulfuric acid. The mechanism proceeds via the in situ generation of a reactive bromomethyl cation equivalent, which attacks the electron-rich aromatic ring. Crucially, the patent specifies a controlled addition of sulfuric acid at temperatures between 20-50°C, followed by heating to 60-120°C. This thermal profile is essential for driving the reaction to completion while minimizing poly-bromination or ring degradation. The propionic acid side chain acts as a moderate activating group, directing the incoming electrophile predominantly to the para-position due to steric hindrance at the ortho-positions. The reaction scheme below highlights this final critical step, demonstrating how the carboxylic acid moiety remains intact while the bromomethyl group is installed with high precision, yielding the target p-bromomethyl isophenylpropionic acid ready for downstream coupling reactions.
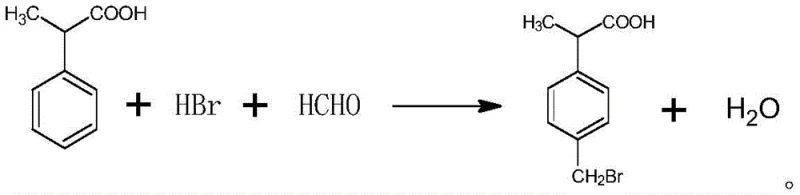
How to Synthesize p-Bromomethyl Isophenylpropionic Acid Efficiently
The synthesis of p-bromomethyl isophenylpropionic acid via this novel styrene-based route offers a clear pathway for process chemists to establish a robust manufacturing protocol. The procedure begins with the hydrochlorination of styrene in dichloromethane, followed by the conversion to the Grignard reagent and subsequent carboxylation to form the acid backbone. The final step involves the careful bromomethylation using paraformaldehyde and hydrobromic acid under acidic conditions. Each stage is optimized for high yield and purity, utilizing standard laboratory and plant equipment. For R&D teams looking to implement this technology, the following guide outlines the standardized operational sequence derived from the patent examples, ensuring reproducibility and safety during scale-up trials.
- Perform hydrochlorination of styrene in dichloromethane at low temperature (-5 to 20°C) to generate α-methyl benzyl chloride with high regioselectivity.
- Convert α-methyl benzyl chloride into a Grignard reagent using magnesium in 2-methyltetrahydrofuran, followed by carboxylation with CO2 and hydrolysis to yield 2-phenylpropionic acid.
- Execute bromomethylation of 2-phenylpropionic acid using paraformaldehyde and hydrobromic acid with controlled sulfuric acid addition to obtain the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this styrene-based synthesis route translates into tangible strategic advantages regarding cost stability and supply security. The shift from niche raw materials to commodity chemicals fundamentally alters the cost structure of the intermediate. By eliminating the dependency on 4-methyl styrene, manufacturers can insulate their production costs from the price volatility associated with specialized fine chemical feedstocks. Furthermore, the simplification of the process flow—specifically the removal of the debromination step—reduces the number of unit operations required. Fewer processing steps mean lower consumption of utilities, solvents, and labor hours, which cumulatively drives down the cost of goods sold (COGS). This efficiency gain allows for more competitive pricing models in the global API market, enabling suppliers to offer better value to downstream pharmaceutical partners without compromising on margin.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the substitution of expensive starting materials with low-cost styrene and the elimination of yield-lossing purification steps. In the conventional route, the formation of dibromo byproducts necessitates a corrective debromination reaction, which consumes additional reagents and reduces the overall mass balance of the final product. By avoiding this inefficiency, the new route maximizes atom economy and throughput. Additionally, the use of recyclable solvents like dichloromethane and 2-methyltetrahydrofuran further enhances the economic profile, as solvent recovery systems can be easily integrated into the plant design to minimize waste disposal costs and raw material procurement expenses.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of styrene, a high-volume petrochemical produced globally in massive quantities. Unlike 4-methyl styrene, which may have limited suppliers and long lead times, styrene is readily accessible from multiple geographic regions, reducing the risk of supply disruptions. This abundance ensures that production schedules can be maintained consistently, even during periods of market tightness for specialty chemicals. Moreover, the robustness of the Grignard and bromomethylation reactions means that the process is less sensitive to minor variations in raw material quality, providing a buffer against supply chain fluctuations and ensuring a steady flow of high-quality intermediate to API manufacturers.
- Scalability and Environmental Compliance: From an operational perspective, the process is designed for seamless scale-up from pilot plant to commercial tonnage. The reactions utilize standard exothermic control measures and conventional separation techniques such as distillation and crystallization, which are well-understood by plant operators. This familiarity reduces the technical risk associated with technology transfer. Environmentally, the shorter synthetic route generates less chemical waste per kilogram of product. The avoidance of heavy metal catalysts or exotic reagents simplifies effluent treatment, helping facilities meet increasingly stringent environmental regulations. The ability to produce high-purity material with a smaller environmental footprint aligns with the sustainability goals of modern pharmaceutical supply chains, enhancing the corporate social responsibility profile of the manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a reliable foundation for decision-making. Understanding these details is crucial for evaluating the feasibility of adopting this route for your specific production needs.
Q: Why is the styrene-based route superior to the 4-methyl styrene route for this intermediate?
A: The styrene-based route utilizes a widely available commodity chemical, whereas 4-methyl styrene is expensive and difficult to source in high purity. Additionally, the new route avoids the formation of dibromo byproducts that require complex debromination steps, significantly simplifying purification and improving overall yield.
Q: What are the critical control points in the bromomethylation step to ensure purity?
A: Critical control involves the dropwise addition of concentrated sulfuric acid at a controlled temperature (20-50°C) followed by a specific heat preservation phase (60-120°C). This precise thermal management minimizes side reactions and ensures high regioselectivity for the para-position bromomethylation.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for industrial scalability. It employs standard unit operations such as distillation, phase separation, and crystallization, and uses common solvents like dichloromethane and 2-methyltetrahydrofuran, making it highly adaptable for multi-ton production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Bromomethyl Isophenylpropionic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development and commercialization of life-saving medications like Loxoprofen Sodium. Our technical team has extensively analyzed advanced synthetic routes, including the styrene-based methodology discussed herein, to ensure we offer the most efficient and cost-effective solutions to our global partners. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of p-bromomethyl isophenylpropionic acid meets the exacting standards required for pharmaceutical synthesis.
We invite you to collaborate with us to optimize your supply chain for NSAID intermediates. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By leveraging our technical expertise and manufacturing capabilities, we can help you reduce lead times and secure a stable supply of this vital building block. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value for your organization.
