Optimizing Dabigatran Etexilate Intermediate Production via Early-Stage Cyclization and Catalytic Oxidation
Optimizing Dabigatran Etexilate Intermediate Production via Early-Stage Cyclization and Catalytic Oxidation
The pharmaceutical industry continuously seeks robust synthetic pathways for anticoagulant agents, particularly for direct thrombin inhibitors like dabigatran etexilate. A recent breakthrough detailed in patent CN116003384A introduces a highly efficient method for synthesizing a critical precursor, specifically ethyl 3-[[[2-[[(4-cyanophenyl)amino]methyl]-1-methyl-1H-benzimidazol-5-yl]carbonyl]pyridin-2-ylamino]propionate. This intermediate serves as a pivotal building block in the assembly of the final active pharmaceutical ingredient (API). The disclosed methodology addresses longstanding challenges in impurity control and yield optimization by fundamentally restructuring the order of synthetic operations. By shifting the benzimidazole ring formation to an earlier stage in the sequence, the process mitigates the generation of complex by-product profiles that often plague late-stage cyclizations. This strategic adjustment not only streamlines the purification workflow but also enhances the overall economic viability of the manufacturing process, making it a compelling candidate for industrial adoption by reliable pharmaceutical intermediate suppliers.
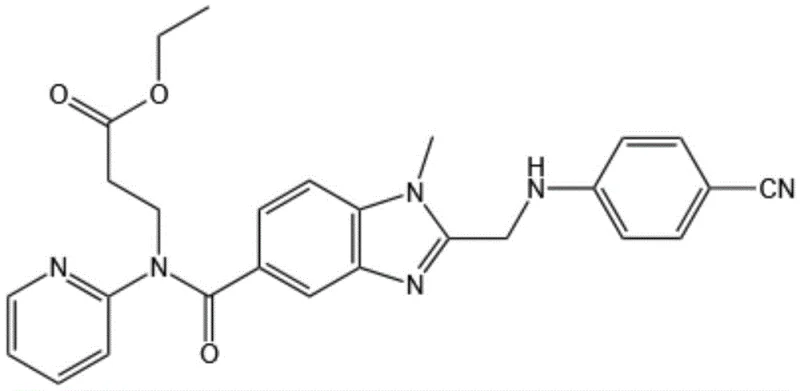
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzimidazole-based anticoagulant intermediates has relied on routes where the heterocyclic ring is constructed at a very late stage, often after the assembly of the bulky side chains. For instance, prior art such as patent WO9837075 describes a pathway where the condensation to form the benzimidazole ring occurs between a complex amine and an activated acid derivative. This approach suffers from significant drawbacks, primarily due to the steric hindrance and electronic deactivation present in the advanced intermediates. Consequently, the cyclization reaction often proceeds with poor selectivity, leading to a mixture of regioisomers and unreacted starting materials that are chemically similar to the desired product. Furthermore, traditional methods frequently necessitate the use of expensive coupling reagents like EDCI and HOBT in stoichiometric amounts for the ring-closing step, which generates substantial waste and complicates downstream processing. The resulting crude products typically require rigorous purification techniques, such as column chromatography, which are notoriously difficult to scale and represent a major bottleneck in cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the methodology presented in CN116003384A adopts a divergent strategy by executing the benzimidazole cyclization immediately after the initial substitution step, utilizing simple, low-molecular-weight precursors. This early-stage ring closure ensures that the heterocyclic core is fully established before the introduction of the more complex pyridine-containing side chain. By constructing the scaffold on a simpler substrate, the reaction kinetics are significantly improved, and the formation of unwanted isomers is effectively suppressed. The subsequent steps involve functionalizing this pre-formed core through highly selective transformations, such as the oxidation of a methyl group to a carboxylic acid, rather than struggling to close a ring on a congested molecule. This logical reordering of synthetic steps results in a much cleaner reaction profile, allowing for purification via simple recrystallization rather than chromatography. The overall synthetic route is depicted below, highlighting the streamlined progression from simple nitrotoluene derivatives to the complex target molecule.
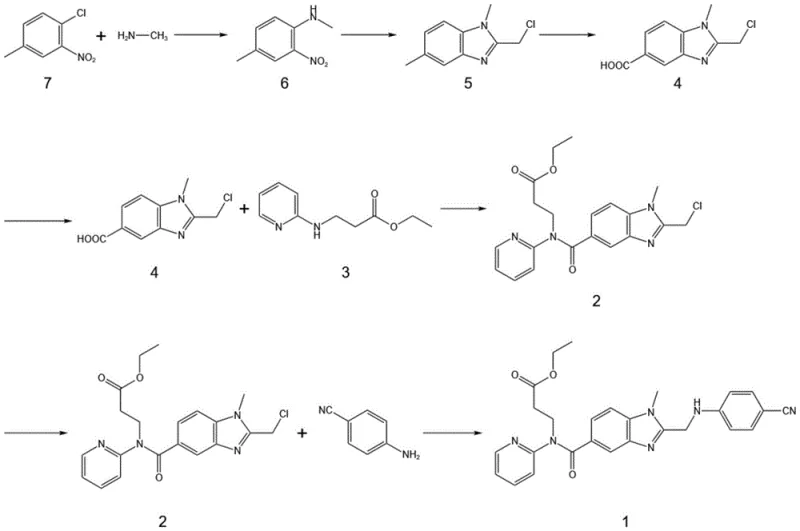
Mechanistic Insights into NHPI-Catalyzed Benzylic Oxidation
A cornerstone of this improved synthetic pathway is the utilization of an N-hydroxyphthalimide (NHPI) mediated oxidation system for converting the benzylic methyl group into a carboxylic acid functionality. This transformation, occurring in Step 3 of the sequence, replaces harsher traditional oxidants with a catalytic radical process that operates under controlled oxygen pressure. The mechanism involves the generation of phthalimide-N-oxyl (PINO) radicals by the metal catalyst, typically manganese dioxide or cobalt salts, which then abstract a hydrogen atom from the benzylic position of the benzimidazole intermediate. This creates a stable benzylic radical that reacts with molecular oxygen to form a hydroperoxide intermediate, which subsequently decomposes to the aldehyde and finally the carboxylic acid. The beauty of this system lies in its chemoselectivity; the mild radical conditions tolerate the sensitive nitrogen atoms within the benzimidazole and pyridine rings, preventing N-oxidation or ring degradation that might occur with stronger oxidizing agents like potassium permanganate or chromic acid.
Furthermore, the impurity control mechanism inherent in this catalytic cycle contributes significantly to the high purity specifications observed in the final product. Because the oxidation proceeds through a well-defined radical chain mechanism, side reactions such as over-oxidation of the aromatic ring or cleavage of the N-methyl group are minimized. The use of a heterogeneous metal catalyst like manganese dioxide also facilitates easy removal of the catalyst from the reaction mixture via simple filtration, eliminating the risk of heavy metal contamination in the final API. This level of control is crucial for meeting the stringent regulatory requirements for anticoagulant drugs, where trace impurities can have significant biological effects. The combination of high conversion rates, often exceeding 90% under optimized conditions at temperatures around 110°C, and the clean reaction profile makes this oxidation step a robust unit operation suitable for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Ethyl 3-[[[2-[[(4-cyanophenyl)amino]methyl]-1-methyl-1H-benzimidazol-5-yl]carbonyl]pyridin-2-ylamino]propionate Efficiently
The execution of this synthesis requires careful attention to reaction conditions, particularly during the cyclization and oxidation phases, to maximize yield and minimize by-product formation. The process begins with the nucleophilic substitution of 3-nitro-4-chlorotoluene with methylamine in a polar solvent like ethanol, followed by immediate cyclization with chloroacetic anhydride in the presence of a weak base such as potassium carbonate. Once the benzimidazole core is established, the critical oxidation step is performed in tetrahydrofuran under an oxygen atmosphere, utilizing NHPI and manganese dioxide at elevated temperatures. The resulting acid is then coupled with the pyridine-containing amine using standard peptide coupling protocols before the final condensation with p-aminobenzonitrile. Detailed operational parameters, including specific molar ratios, temperature gradients, and workup procedures for each stage, are essential for reproducing the high purity and yield reported in the patent literature.
- Perform nucleophilic substitution of 3-nitro-4-chlorotoluene (Compound 7) with methylamine to form N-methyl-(4-methyl-2-nitro)aniline (Compound 6).
- Execute ring closure reaction of Compound 6 with haloacetic anhydride under weak base catalysis to generate the benzimidazole core (Compound 5).
- Conduct selective oxidation of the methyl group in Compound 5 using NHPI and a metal catalyst (e.g., MnO2) under oxygen pressure to yield the carboxylic acid (Compound 4).
- Couple Compound 4 with ethyl 3-[(pyridin-2-yl)amino]propionate (Compound 3) using EDCI to form the amide linkage (Compound 2).
- Finalize the synthesis by condensing Compound 2 with p-aminobenzonitrile under acidic conditions to obtain the target intermediate (Compound 1).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this novel synthetic route offers substantial strategic benefits that directly impact the bottom line and operational resilience. The primary advantage lies in the drastic simplification of the purification process. By avoiding the need for column chromatography, which is a batch-intensive and solvent-heavy operation, manufacturers can significantly reduce production cycle times and solvent consumption. This elimination of complex purification steps translates directly into cost reduction in pharmaceutical intermediate manufacturing, as it lowers both the direct material costs associated with silica gel and solvents and the indirect costs related to equipment occupancy and labor. Additionally, the reliance on commodity chemicals such as 3-nitro-4-chlorotoluene and methylamine ensures a stable and diversified supply base, reducing the risk of raw material shortages that can disrupt production schedules.
- Cost Reduction in Manufacturing: The economic efficiency of this process is driven by the high atom economy of the early-stage cyclization and the use of catalytic rather than stoichiometric oxidants. Traditional routes often require expensive coupling reagents in large excess to drive difficult late-stage cyclizations, generating significant amounts of urea by-products that must be disposed of. In contrast, the new method utilizes cheaper reagents like chloroacetic anhydride and catalytic amounts of manganese dioxide, which are inexpensive and readily available. The ability to purify intermediates via recrystallization rather than chromatography further reduces the cost of goods sold (COGS) by minimizing solvent recovery loads and waste disposal fees, creating a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: The robustness of the synthetic steps contributes to a more predictable and reliable supply chain. The oxidation step, which uses oxygen gas and a solid catalyst, is inherently scalable and less prone to the variability often seen in reactions requiring sensitive organometallic reagents. This stability ensures consistent batch-to-batch quality, reducing the incidence of out-of-specification results that can lead to production delays. Furthermore, the use of common organic solvents like ethyl acetate and ethanol, which are widely produced and stocked globally, mitigates the risk of logistics bottlenecks. This reliability is critical for maintaining continuous supply to downstream API manufacturers who operate on tight just-in-time inventory schedules.
- Scalability and Environmental Compliance: The process design aligns well with green chemistry principles, facilitating easier regulatory approval and environmental compliance. The reduction in solvent usage and the elimination of chromatographic silica waste lower the environmental footprint of the manufacturing process. The oxidation step generates water as a primary by-product, which is far easier to treat than the heavy metal waste or complex organic residues associated with traditional oxidants. This environmental compatibility simplifies the permitting process for new production lines and supports corporate sustainability goals. The straightforward nature of the unit operations, such as filtration and crystallization, allows for seamless technology transfer from pilot plant to multi-ton commercial production scales without significant re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications of adopting this route for large-scale production.
Q: How does the new synthesis route improve product purity compared to prior art?
A: The novel route performs the benzimidazole cyclization at an early stage (Step 2) rather than late-stage. This prevents the formation of difficult-to-remove regioisomers and by-products that typically occur when cyclizing complex amides, thereby significantly enhancing crude purity and reducing the need for column chromatography.
Q: What are the specific advantages of the NHPI/metal catalytic oxidation step?
A: The use of N-hydroxyphthalimide (NHPI) combined with a metal catalyst like manganese dioxide allows for highly selective oxidation of the benzylic methyl group to a carboxylic acid under mild oxygen pressure. This method offers higher conversion rates and selectivity compared to traditional strong oxidants, minimizing over-oxidation and side reactions.
Q: Is this synthetic method suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. It utilizes common organic solvents like ethanol and ethyl acetate, avoids expensive palladium catalysts in the main backbone construction, and achieves high yields (often exceeding 88%) with simple workup procedures like recrystallization instead of complex chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dabigatran Etexilate Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving anticoagulant therapies. Our technical team has thoroughly analyzed the synthetic pathway described in CN116003384A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative route to market. We are equipped with state-of-the-art reactors capable of handling high-pressure oxidation reactions and stringent purity specifications to ensure that every batch meets the rigorous demands of global regulatory agencies. Our rigorous QC labs employ advanced analytical techniques to monitor impurity profiles at every stage, guaranteeing the consistency and safety of the materials we supply to our partners.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this cost-effective and robust synthetic technology. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your supply chain economics. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments for this key dabigatran intermediate, and let us support your journey towards more efficient and sustainable drug manufacturing.
