Advanced Metal-Free Synthesis of Trifluoromethyl Aryl Sulfides for Commercial Scale-up
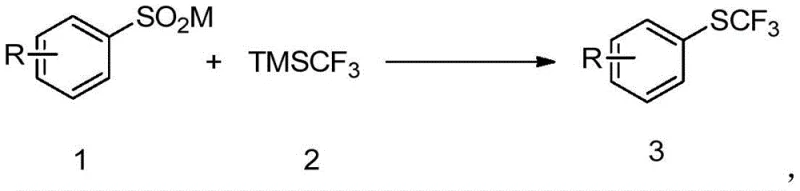
The introduction of the trifluoromethylthio group (SCF3) into organic molecules represents a pivotal strategy in modern medicinal chemistry and agrochemical design, primarily due to its exceptional ability to enhance lipophilicity and metabolic stability. Patent CN112375022B discloses a groundbreaking preparation method for trifluoromethyl aryl sulfides that addresses long-standing challenges in fluorine chemistry. Unlike traditional approaches that often rely on harsh conditions or toxic heavy metals, this invention utilizes aryl sulfinate salts and trifluoromethyl trimethylsilane (TMSCF3) as key substrates. The process operates under transition metal-free conditions, leveraging a synergistic system of additives, oxidants, and bases to achieve efficient coupling. This technological advancement is particularly significant for the pharmaceutical industry, where the demand for high-purity fluorinated intermediates is surging. By enabling reactions at mild temperatures ranging from room temperature to 120°C, this method offers a greener, more economical pathway to valuable SCF3-containing building blocks, positioning it as a critical asset for reliable pharma intermediate supplier networks seeking sustainable manufacturing solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the trifluoromethylthio arene motif has been fraught with significant synthetic hurdles that impede efficient commercial production. Conventional methodologies frequently necessitate the use of expensive and scarce transition metal catalysts, such as palladium or copper complexes, which not only drive up raw material costs but also introduce complex downstream purification requirements to meet stringent residual metal specifications. Furthermore, many established protocols involve the use of hazardous electrophilic trifluoromethylthiolating reagents that are unstable, difficult to handle, and pose safety risks during scale-up. Other reported methods, including radical trifluoromethylation of sulfur-containing substrates, often suffer from poor functional group tolerance and require extreme reaction conditions, such as high pressure or cryogenic temperatures, which are energy-intensive and operationally demanding. These limitations collectively result in lower overall yields, increased waste generation, and prolonged lead times, creating substantial bottlenecks for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach detailed in the patent utilizes a transition metal-free catalytic system that fundamentally simplifies the synthetic workflow. By employing readily available aryl sulfinates and TMSCF3, the method bypasses the need for precious metal catalysts entirely, relying instead on a robust organic redox system involving iodine and phosphine additives. This shift eliminates the risk of heavy metal contamination, thereby streamlining the purification process and ensuring the final product meets rigorous quality standards without extensive chelation or filtration steps. The reaction proceeds smoothly in polar aprotic solvents like DMF, demonstrating excellent compatibility with a wide range of substituents on the aromatic ring, including halogens, alkyl groups, and esters. This versatility allows for the rapid synthesis of diverse libraries of trifluoromethyl aryl sulfides, facilitating faster drug discovery cycles. Moreover, the ability to conduct the reaction at moderate temperatures significantly reduces energy consumption, aligning with modern green chemistry principles and enhancing the overall economic viability of the process for large-scale applications.
Mechanistic Insights into Transition Metal-Free Trifluoromethylthiolation
The mechanistic pathway of this transformation is a sophisticated interplay of reduction and oxidation steps that activate the sulfur center for nucleophilic attack. Initially, the aryl sulfinate substrate undergoes a reduction mediated by the phosphine additive, likely generating a diaryl disulfide intermediate in situ. This species is subsequently oxidized by the iodine oxidant to form a highly reactive aryl sulfenyl iodide (ArSI) species, which serves as the electrophilic partner in the coupling. Concurrently, the base, typically cesium fluoride, activates the TMSCF3 reagent by generating a nucleophilic trifluoromethyl anion equivalent. This nucleophile then attacks the sulfur atom of the ArSI intermediate, displacing the iodide and forming the desired carbon-sulfur bond to yield the trifluoromethyl aryl sulfide product. This elegant mechanism avoids the formation of stable metal-sulfur complexes that often plague transition metal-catalyzed reactions, ensuring high turnover and minimal catalyst deactivation.
From an impurity control perspective, this metal-free mechanism offers distinct advantages for producing high-purity OLED material or pharmaceutical intermediates. The absence of transition metals means there is no risk of metal-induced side reactions, such as homocoupling or beta-hydride elimination, which can generate difficult-to-remove impurities. The primary byproducts of the reaction are inorganic salts and phosphine oxides, which are generally water-soluble and easily removed during the aqueous workup phase described in the patent examples. This clean reaction profile simplifies the isolation process, often requiring only standard extraction and column chromatography to achieve high purity. For R&D teams focused on regulatory compliance, this translates to a more predictable impurity profile and reduced analytical burden. The robustness of the mechanism across different electronic environments on the aromatic ring further ensures consistent quality, making it a reliable choice for synthesizing complex fluorinated architectures.
How to Synthesize Trifluoromethyl Aryl Sulfide Efficiently
The practical execution of this synthesis involves a straightforward one-pot procedure that is amenable to both laboratory optimization and industrial manufacturing. The process begins by charging a reaction vessel with the aryl sulfinate substrate, TMSCF3, iodine, triphenylphosphine, and cesium fluoride in a solvent such as N,N-dimethylformamide. The mixture is then stirred under a nitrogen atmosphere at temperatures between 25°C and 120°C for a duration of 8 to 24 hours, allowing the redox cycle to proceed to completion. Following the reaction, the crude product is isolated through a standard workup involving dilution with ethyl acetate, washing with water and brine to remove inorganic salts, and drying over anhydrous magnesium sulfate. The final purification is achieved via column chromatography, yielding the target trifluoromethyl aryl sulfide with high purity. For detailed operational parameters and specific stoichiometric ratios optimized for different substrates, please refer to the standardized guide below.
- Mix aryl sulfinate, TMSCF3, iodine oxidant, triphenylphosphine additive, and cesium fluoride base in DMF solvent.
- Stir the reaction mixture at temperatures between 25°C and 120°C for 8 to 24 hours under nitrogen atmosphere.
- Purify the crude product via filtration, washing, and column chromatography to obtain high-purity trifluoromethyl aryl sulfide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis route presents a compelling value proposition centered on cost efficiency and supply security. The elimination of expensive transition metal catalysts directly reduces the bill of materials, while the use of commodity chemicals like aryl sulfinates and iodine ensures a stable and diversified supply base that is less susceptible to geopolitical fluctuations affecting rare earth or precious metal markets. Furthermore, the simplified purification workflow reduces the consumption of specialized scavenging resins and solvents, leading to substantial cost savings in downstream processing. The mild reaction conditions also lower energy overheads and extend the lifespan of reactor equipment by avoiding corrosive or extreme environments. These factors collectively contribute to a more resilient and cost-effective supply chain for fluorinated intermediates.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the complete removal of transition metal catalysts from the process. In traditional fluorination methods, the cost of palladium or copper catalysts, combined with the expensive ligands required to stabilize them, can constitute a major portion of the production cost. Additionally, the regulatory requirement to reduce residual metals to parts-per-million levels often necessitates additional purification steps, such as activated carbon treatment or specialized filtration, which add time and expense. By utilizing a metal-free organic redox system, this method inherently avoids these costs, allowing for a leaner manufacturing process. The reagents used, such as TMSCF3 and aryl sulfinates, are increasingly available from multiple global suppliers, fostering price competition and further driving down input costs for large-scale production runs.
- Enhanced Supply Chain Reliability: Supply chain continuity is critical for maintaining production schedules in the pharmaceutical and agrochemical sectors. This synthesis method relies on raw materials that are chemically stable and widely produced, reducing the risk of shortages associated with specialized or hazardous reagents. For instance, unlike some electrophilic trifluoromethylthiolating agents that require cold chain logistics or have short shelf lives, the substrates in this patent are robust and easy to store. The flexibility in reaction temperature, which can range from ambient to moderately elevated levels, also provides operational resilience, allowing manufacturers to adapt to varying utility availability without compromising yield. This adaptability ensures that production timelines remain consistent, reducing lead time for high-purity pharmaceutical intermediates and preventing costly delays in drug development pipelines.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and waste management, but this method is designed with scalability in mind. The reaction exotherm is manageable, and the use of common solvents like DMF facilitates easy integration into existing infrastructure. From an environmental standpoint, the absence of heavy metals significantly reduces the toxicity of the waste stream, simplifying effluent treatment and lowering disposal costs. This aligns with increasingly strict environmental regulations globally, minimizing the risk of compliance-related shutdowns. The green nature of the process, characterized by atom economy and reduced hazard, enhances the corporate sustainability profile of manufacturers, making it an attractive option for companies aiming to reduce their environmental footprint while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethylthiolation technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented process (CN112375022B) specifically operates without transition metal catalysts, utilizing an organic additive and oxidant system instead, which significantly reduces downstream purification costs.
Q: What are the optimal reaction conditions for maximum yield?
A: The patent indicates that using N,N-dimethylformamide (DMF) as the solvent with cesium fluoride as the base at temperatures around 100°C provides superior yields compared to other solvents or bases.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the mild reaction conditions (25-120°C) and the use of commercially available, stable reagents like aryl sulfinates and TMSCF3 make this process highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Aryl Sulfide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology for the next generation of fluorinated drugs and materials. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our facilities are equipped with state-of-the-art reactors capable of handling the specific solvent and temperature requirements of this process, while our stringent purity specifications and rigorous QC labs guarantee that every batch meets the highest international standards. We understand that consistency is key in the supply of complex fine chemical intermediates, and our dedicated technical team is prepared to optimize this route for your specific substrate needs.
We invite you to collaborate with us to leverage this innovative chemistry for your upcoming projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this metal-free protocol for your specific molecule. We encourage you to contact our technical procurement team today to request specific COA data for similar fluorinated compounds and to discuss route feasibility assessments tailored to your production goals. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
