Advanced Azide-Catalyzed Synthesis of Acylated 1,3-Dicarbonyl Compounds for Commercial Scale-Up
Advanced Azide-Catalyzed Synthesis of Acylated 1,3-Dicarbonyl Compounds for Commercial Scale-Up
The global demand for high-performance agrochemicals continues to drive innovation in the synthesis of key intermediates, particularly acylated 1,3-dicarbonyl compounds which serve as critical precursors for herbicides and plant growth regulators. A pivotal advancement in this domain is documented in Chinese Patent CN1185202C, which discloses a robust and environmentally superior process for preparing these valuable structures through the rearrangement of corresponding enol esters. This technology represents a significant leap forward from conventional methodologies by utilizing alkali metal azides as efficient catalysts or reagents, thereby overcoming the severe toxicity and waste disposal challenges associated with historical cyanide-based or Lewis acid-mediated routes. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, understanding the mechanistic elegance and commercial viability of this azide-catalyzed pathway is essential for securing a competitive supply chain. The core innovation lies in the ability to generate the target acylated cyclic 1,3-dicarbonyl compounds, such as substituted 1,3-cyclohexanediones, under mild conditions while avoiding the generation of hazardous byproducts.
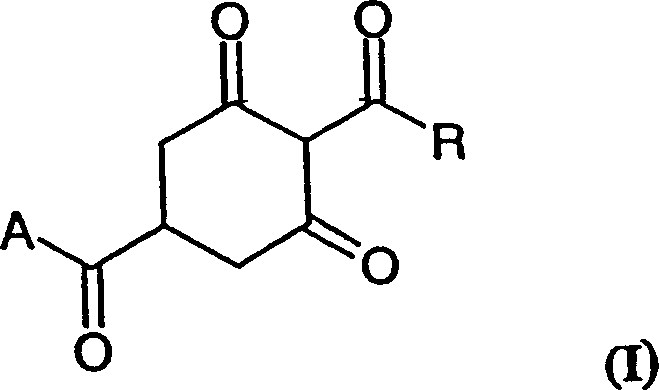
The structural versatility of the compounds produced via this method is illustrated by the general Formula I, where the R group can vary from simple alkyl chains to complex substituted phenyl rings, allowing for the tailored synthesis of specific herbicidal active ingredients. This flexibility ensures that manufacturers can adapt the process to produce a wide array of high-purity agrochemical intermediates required for next-generation crop protection solutions. By leveraging this patented technology, producers can achieve substantial cost reduction in agrochemical manufacturing through streamlined processing and reduced environmental compliance burdens.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of acylated 1,3-dicarbonyl compounds relied heavily on rearrangement reactions facilitated by harsh and problematic catalysts that posed significant operational and environmental risks. Prior art, including references such as US Patent 4,695,673 and EP 306996, disclosed the use of stoichiometric amounts of aluminum chloride, often requiring two moles of the Lewis acid per mole of substrate, which created severe corrosion issues in reactor vessels and generated massive quantities of acidic waste streams requiring complex neutralization protocols. Furthermore, alternative catalytic systems utilizing cyanide sources were plagued by the generation of hydrogen cyanide gas, a highly toxic volatile compound that necessitates elaborate scrubbing systems and poses acute safety hazards to personnel, thereby inflating the capital and operational expenditures of the facility. Other methods employing catalysts like 4-dimethylaminopyridine (DMAP) required rigorous recovery processes to prevent product contamination and economic loss, adding unnecessary unit operations to the manufacturing workflow. These conventional approaches not only suffered from low atom economy but also faced increasing regulatory scrutiny due to their heavy metal content and toxic effluent profiles, making them increasingly unsustainable for modern large-scale production.
The Novel Approach
In stark contrast, the process detailed in CN1185202C introduces a paradigm shift by employing alkali metal azides, such as sodium azide or potassium azide, to facilitate the rearrangement of enol esters into the desired acylated products with remarkable efficiency and safety. This novel approach eliminates the need for stoichiometric Lewis acids and toxic cyanide reagents, replacing them with inexpensive and readily available inorganic salts that function effectively in catalytic amounts when paired with organic bases like triethylamine. A distinct advantage of this azide-based system is its benign decomposition profile; upon acidification during the workup phase, any residual azide species form hydrazoic acid which rapidly decomposes into harmless nitrogen gas, thereby simplifying waste treatment and eliminating the risk of toxic gas accumulation in the plant. The reaction proceeds smoothly in common organic solvents such as toluene, DMF, or acetonitrile at moderate temperatures ranging from 20°C to 75°C, ensuring energy efficiency and minimizing thermal degradation of sensitive functional groups. This methodological breakthrough allows for the direct isolation of high-purity products without the need for complex chromatographic purification, directly addressing the needs of procurement managers focused on cost reduction in agrochemical manufacturing.
Mechanistic Insights into Azide-Catalyzed Enol Ester Rearrangement
The chemical mechanism underpinning this transformation involves the nucleophilic activation of the enol ester substrate by the azide anion, which initiates a rearrangement cascade leading to the formation of the acylated 1,3-dicarbonyl skeleton. The presence of a base, typically a trialkylamine such as triethylamine used in molar excess relative to the enol ester, is crucial for deprotonating intermediate species and driving the equilibrium towards the product side. In certain embodiments, the addition of phase transfer catalysts like 18-crown-6 can further enhance reaction kinetics by solubilizing the alkali metal cations in non-polar solvent media, thereby increasing the nucleophilicity of the azide anion and accelerating the rate of rearrangement. The process is highly tolerant of various substituents on the aromatic ring or the acyl chain, allowing for the synthesis of diverse derivatives including those with nitro, halo, or alkoxy groups without compromising yield or selectivity. Crucially, the reaction must be conducted under substantially anhydrous conditions to prevent the saponification of the starting enol ester, a side reaction that would otherwise consume the raw material and reduce the overall process efficiency. By maintaining strict moisture control, manufacturers can ensure consistent batch-to-batch reproducibility and maximize the yield of the target cyclohexanedione derivatives.
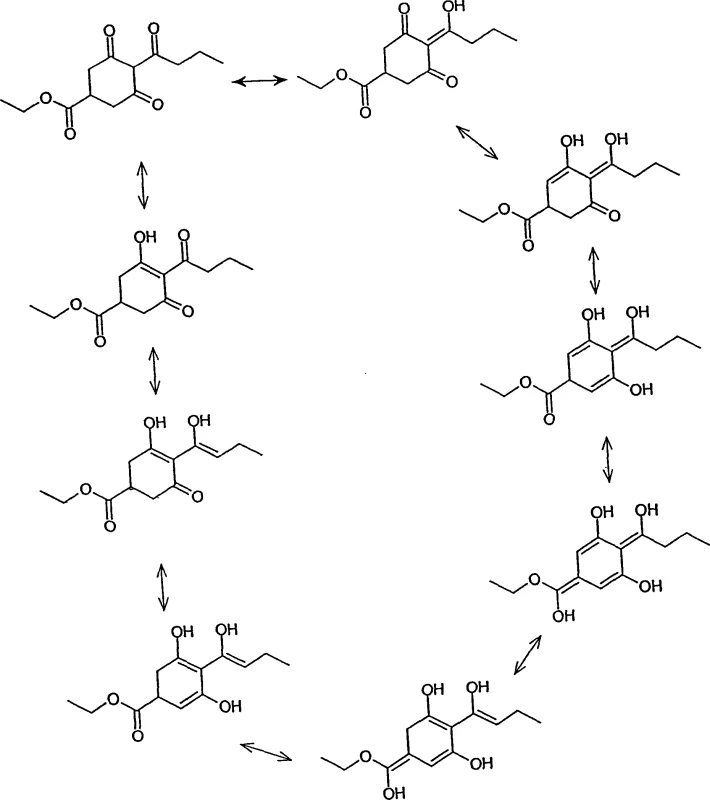
Furthermore, the products obtained via this route exist in dynamic equilibrium with their tautomeric forms, a phenomenon clearly depicted in the structural interconversions where hydrogen migration generates multiple stable isomers such as enols and ketones. Understanding this tautomerism is vital for R&D teams as it influences the physical properties, solubility, and reactivity of the final intermediate in downstream coupling reactions. The patent explicitly notes that references to acylated 1,3-dicarbonyl compounds include all such tautomeric forms, ensuring comprehensive intellectual property coverage and flexibility in product specification. This mechanistic clarity allows chemists to optimize crystallization and purification strategies to isolate the most thermodynamically stable or commercially desirable tautomer, ensuring high-purity agrochemical intermediates that meet stringent quality standards.
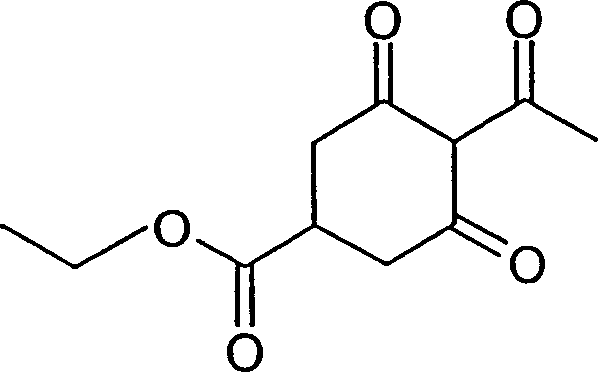
How to Synthesize Ethyl 2-Acetyl-1,3-Cyclohexanedione-5-Carboxylate Efficiently
To demonstrate the practical application of this technology, we examine the synthesis of ethyl 2-acetyl-1,3-cyclohexanedione-5-carboxylate, a representative intermediate that exemplifies the efficiency of the azide-catalyzed protocol. The procedure begins with the preparation of the O-acyl enol ester starting material, which is readily accessible via the acylation of ethyl 1,3-cyclohexanedione-5-carboxylate with acetyl chloride using standard techniques. The rearrangement step is performed by charging the enol ester into a reactor along with a catalytic quantity of sodium azide and a stoichiometric excess of triethylamine in a solvent mixture of toluene and DMF. The reaction mixture is then heated gently, typically initiating at 45°C where an exotherm may raise the temperature to 60°C, before being cooled and maintained at 30°C to complete the conversion without thermal stress. Following the reaction, the product is extracted into the organic phase, separated, and subjected to acidification with hydrochloric acid to precipitate the final diketone product, which is isolated as a viscous oil with excellent crude yields.
- Prepare the reaction mixture by combining the O-acyl enol ester starting material with a catalytic amount of alkali metal azide (e.g., sodium azide) and an organic base like triethylamine in a suitable solvent system such as toluene or DMF.
- Heat the reaction mixture to a temperature between 20°C and 75°C, preferably around 45°C to 55°C, and maintain stirring under substantially anhydrous conditions to prevent saponification of the enol ester.
- Upon completion, extract the rearrangement product as a salt, separate the phases, and acidify the aqueous layer with hydrochloric acid to isolate the final acylated 1,3-dicarbonyl compound.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this azide-catalyzed process offers transformative benefits that extend far beyond simple chemical yield improvements, fundamentally altering the cost structure and risk profile of producing acylated 1,3-dicarbonyl compounds. By transitioning away from legacy technologies that rely on hazardous cyanides or corrosive aluminum chloride, facilities can drastically simplify their environmental health and safety (EHS) protocols, leading to lower insurance premiums and reduced regulatory compliance costs. The elimination of expensive catalyst recovery units, which were mandatory for processes using DMAP or similar amines, results in a significantly simplified plant footprint and reduced capital expenditure for new production lines. Moreover, the use of commodity chemicals like sodium azide and triethylamine ensures a stable and resilient supply chain, mitigating the risk of raw material shortages that often plague specialty reagent markets. This robustness allows for continuous commercial scale-up of complex agrochemical intermediates without the bottlenecks associated with waste treatment or catalyst recycling.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the removal of costly unit operations associated with catalyst recovery and toxic waste neutralization. Traditional methods often incurred hidden costs related to the disposal of heavy metal sludge and the energy-intensive regeneration of Lewis acids, whereas the azide method generates nitrogen gas as a benign byproduct that requires no treatment. Additionally, the ability to run the reaction at mild temperatures (20°C to 75°C) reduces utility consumption for heating and cooling, contributing to a lower overall cost of goods sold (COGS). The high atom economy and minimal formation of side products further enhance the effective yield, ensuring that more of the expensive starting enol ester is converted into saleable product rather than waste.
- Enhanced Supply Chain Reliability: From a sourcing perspective, the reagents required for this synthesis are globally available bulk chemicals, reducing dependency on single-source suppliers of exotic catalysts. Sodium azide and triethylamine are produced at massive scales for various industries, ensuring consistent availability and price stability even during market fluctuations. This reliability is critical for maintaining uninterrupted production schedules for high-purity agrochemical intermediates, preventing costly downtime that can ripple through the entire value chain. The simplicity of the workup procedure also means that production cycles are shorter, allowing for faster turnaround times and improved responsiveness to customer demand spikes.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on homogeneous catalysis and standard solvent systems that behave predictably in large reactors. The absence of highly exothermic runaway risks associated with cyanide chemistry makes the scale-up from pilot plant to multi-ton production significantly safer and more manageable. Furthermore, the generation of nitrogen gas instead of toxic effluents aligns perfectly with modern green chemistry principles and stringent environmental regulations, facilitating easier permitting for new facilities. This environmental compatibility not only future-proofs the manufacturing asset but also enhances the brand reputation of the supplier as a sustainable partner in the agrochemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this azide-catalyzed rearrangement technology, providing clarity for stakeholders evaluating its adoption. These insights are derived directly from the experimental data and claims presented in the patent literature, ensuring accuracy and relevance for industrial application. Understanding these nuances is key to leveraging the full potential of the process for optimizing production workflows and achieving strategic sourcing goals.
Q: What are the primary safety advantages of using azide catalysts over cyanide sources in this synthesis?
A: Unlike cyanide catalysts which generate highly toxic hydrogen cyanide gas that contaminates process streams, the azide catalyst system described in CN1185202C forms hydrazoic acid upon acidification, which safely decomposes into nitrogen gas, significantly reducing toxicity risks and waste treatment complexity.
Q: How does this process improve cost efficiency compared to traditional Lewis acid methods?
A: Traditional methods often require stoichiometric amounts of expensive catalysts like aluminum chloride or dimethylaminopyridine that must be recovered. The novel azide process operates with catalytic amounts of inexpensive alkali metal azides and eliminates the need for complex catalyst recovery systems, thereby lowering raw material and processing costs.
Q: Can this rearrangement reaction be scaled for industrial production of herbicide intermediates?
A: Yes, the process utilizes mild reaction temperatures (20°C to 75°C) and common solvents like toluene and DMF. The use of readily available reagents such as sodium azide and triethylamine, combined with simple workup procedures involving phase separation and acidification, makes it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acylated 1,3-Dicarbonyl Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in securing a competitive edge in the global agrochemical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity agrochemical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By integrating the azide-catalyzed rearrangement process into our manufacturing portfolio, we offer our partners a supply solution that balances cost-effectiveness with uncompromising quality and safety standards.
We invite you to engage with our technical procurement team to discuss how this technology can be tailored to your specific project requirements. Contact us today to request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener, more efficient synthesis route. Our experts are ready to provide specific COA data and route feasibility assessments to support your R&D and supply chain planning, ensuring you have the reliable acylated 1,3-dicarbonyl compounds supplier partnership needed to drive your business forward.
