Advanced Aqueous Resolution Technology for High-Purity R-Tetrahydropapaverine Production
The pharmaceutical industry constantly seeks more efficient and environmentally benign pathways for synthesizing critical chiral intermediates. Patent CN110423219B introduces a groundbreaking resolution method for tetrahydroisoquinoline compounds, specifically targeting the production of (+/-)-1-(3,4-dimethoxybenzyl)-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline. This compound serves as a pivotal precursor for Cisatracurium besilate, a potent neuromuscular blocking agent used in anesthesia. The core innovation lies in the substitution of traditional organic solvents with water during the chiral resolution process. By utilizing N-acetyl-L-leucine as the resolving agent in an aqueous medium, this technology addresses significant pain points regarding solvent toxicity, waste generation, and operational complexity found in legacy manufacturing routes.
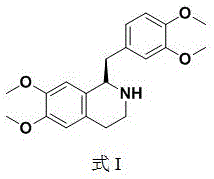
Traditional methods for resolving tetrahydropapaverine have long relied on volatile organic compounds such as toluene, methanol, ether, and acetone. These conventional approaches often necessitate massive solvent-to-substrate volume ratios, sometimes reaching as high as 50:1, which drastically inflates production costs and environmental burdens. Furthermore, existing techniques frequently require multiple rounds of refining or the use of expensive chiral metal catalysts that are difficult to recover. Some prior art even utilizes resolving agents derived from pesticides, which poses regulatory hurdles for pharmaceutical applications. In contrast, the novel approach detailed in the patent simplifies the workflow by leveraging the differential solubility of diastereomeric salts in water. This shift not only streamlines the isolation of the desired R-isomer but also inherently improves the safety profile of the manufacturing facility by minimizing exposure to hazardous vapors.
Mechanistic Insights into Aqueous Chiral Resolution
The success of this resolution strategy hinges on the precise formation of diastereomeric salts between the racemic tetrahydropapaverine and the chiral resolving agent, N-acetyl-L-leucine. In the aqueous phase, the S-isomer salt exhibits significantly lower solubility compared to its R-counterpart, allowing it to precipitate out of the solution upon cooling and stirring. This selective crystallization effectively removes the unwanted enantiomer from the system. The mother liquor, now enriched with the R-isomer, is subsequently extracted using a halogenated alkane such as dichloromethane. This liquid-liquid extraction step is crucial for transferring the target molecule into an organic phase where final crystallization can be induced. The addition of acetone to the concentrated organic extract acts as an anti-solvent, forcing the R-tetrahydropapaverine-N-acetyl-L-leucine salt to crystallize with high fidelity.
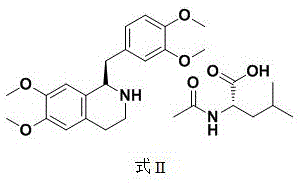
Impurity control is rigorously managed through a specialized purification protocol that further elevates the optical purity of the final product. If the initial crystallization yields a salt with optical purity between 70 percent and 99.9 percent, a secondary refinement step can be employed. This involves dissolving the crude salt in dichloromethane at controlled temperatures ranging from 35°C to 40°C and slowly adding acetone until turbidity appears. Upon cooling, the purified salt precipitates, leaving residual S-isomer impurities in the solution. This mechanism ensures that the final R-tetrahydropapaverine achieves an optical purity exceeding 99.95 percent, meeting the stringent requirements for downstream synthesis of neuromuscular blockers without the need for complex chromatographic separations.
How to Synthesize R-Tetrahydropapaverine Efficiently
The synthesis protocol outlined in the patent offers a robust framework for producing high-quality intermediates suitable for GMP manufacturing. The process begins with the neutralization of tetrahydropapaverine hydrochloride, followed by salt formation in water and sequential extraction and crystallization steps. This methodology is designed to maximize yield while minimizing the consumption of resources. For detailed operational parameters, including specific temperature ramps, stirring rates, and solvent ratios optimized for your specific reactor configuration, please refer to the standardized guide below.
- Neutralize tetrahydropapaverine hydrochloride in water to pH 8-9 and react with N-acetyl-L-leucine to form diastereomeric salts.
- Filter the precipitated S-isomer salt and extract the mother liquor containing the R-isomer with dichloromethane.
- Concentrate the organic phase and induce crystallization of the R-tetrahydropapaverine-N-acetyl-L-leucine salt using acetone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this aqueous resolution technology represents a strategic opportunity to optimize cost structures and mitigate supply risks. The elimination of bulk organic solvents for the primary resolution step translates directly into reduced raw material expenditure and lower costs associated with solvent recovery and disposal systems. Furthermore, the use of water as a medium simplifies the engineering controls required for fire safety and ventilation, potentially lowering capital investment for new production lines. The simplicity of the operation also reduces the likelihood of batch failures due to operator error, ensuring a more consistent supply of critical intermediates for downstream API synthesis.
- Cost Reduction in Manufacturing: The replacement of expensive organic solvents with water significantly lowers the variable costs associated with solvent purchase and recycling. Additionally, the process avoids the use of costly chiral metal catalysts, which are often difficult to remove and recover, thereby reducing the overall cost of goods sold. The simplified workflow requires fewer unit operations compared to multi-step refining processes found in older patents, leading to reduced labor and energy consumption per kilogram of product produced.
- Enhanced Supply Chain Reliability: The resolving agent, N-acetyl-L-leucine, is a commercially available amino acid derivative with a stable global supply chain, unlike specialized pesticide-derived acids or custom-synthesized catalysts. This availability ensures that production schedules are not disrupted by raw material shortages. Moreover, the robustness of the aqueous process allows for flexible scaling, enabling manufacturers to respond quickly to fluctuations in market demand for Cisatracurium besilate without extensive process re-validation.
- Scalability and Environmental Compliance: From an environmental perspective, the drastic reduction in organic solvent usage aligns with increasingly strict global regulations on volatile organic compound (VOC) emissions. The aqueous waste streams generated are easier to treat than complex organic mixtures, facilitating compliance with environmental protection standards. The process is inherently scalable from pilot batches to multi-ton commercial production, as the physical principles of crystallization and extraction remain consistent regardless of scale, ensuring reliable technology transfer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: What is the primary advantage of using water as a resolution solvent?
A: Using water eliminates the need for large volumes of hazardous organic solvents like toluene or methanol, significantly reducing environmental impact and solvent recovery costs while maintaining high optical purity.
Q: What optical purity can be achieved with this method?
A: The patented process allows for the production of R-tetrahydropapaverine-N-acetyl-L-leucine with an optical purity exceeding 99.95 percent after a simple purification step.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability due to its simple operation, mild reaction conditions, and the use of inexpensive, readily available reagents like N-acetyl-L-leucine.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable R-Tetrahydropapaverine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the synthesis of life-saving medications like Cisatracurium besilate. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative aqueous resolution method described in patent CN110423219B can be seamlessly integrated into your supply chain. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of R-tetrahydropapaverine meets the highest international standards for optical and chemical purity.
We invite you to collaborate with us to leverage this advanced technology for your pharmaceutical projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive efficiency and quality in your manufacturing operations.
