Advanced One-Step Synthesis of 4-Aminoindole Derivatives for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust methodologies for constructing complex heterocyclic scaffolds, particularly indole derivatives which serve as critical pharmacophores in numerous bioactive molecules. A significant breakthrough in this domain is detailed in patent CN110156657B, which discloses a novel synthetic method for 4-aminoindole derivatives. This technology addresses a longstanding challenge in medicinal chemistry: the direct functionalization of the indole ring at the 4-position. Traditionally, accessing this specific substitution pattern has been fraught with difficulties, often requiring convoluted multi-step sequences that suffer from poor regioselectivity and low overall yields. The disclosed invention leverages a sophisticated palladium-catalyzed cascade reaction involving aniline derivatives, amine derivatives, and norbornene mediators to achieve this transformation in a single operational step. For R&D directors and process chemists, this represents a paradigm shift, offering a streamlined route to high-value intermediates used in the synthesis of G-protein coupled receptor (GPCR) modulators and GOT1 inhibitors for pancreatic cancer treatment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-aminoindole has relied heavily on classical aromatic substitution strategies that are inherently inefficient and environmentally burdensome. The conventional pathway typically initiates with o-nitrotoluene, which must undergo further nitration to produce 2-methyl-1,3-dinitrobenzene. This initial step is notoriously problematic, often resulting in yields of less than 10%, creating a massive bottleneck in material throughput right at the beginning of the synthesis. Subsequent steps involve nucleophilic addition with N,N-dimethylformamide dimethyl acetal (DMFDMA) followed by transition metal-catalyzed reduction to finally close the indole ring. These processes demand harsh reaction conditions, including strong acids and high temperatures, which not only pose safety risks but also generate significant amounts of hazardous waste. Furthermore, the lack of selectivity in these traditional routes makes it exceedingly difficult to introduce diverse functional groups at the 4-position without protecting group strategies, thereby inflating the step count and reducing the overall economic viability of the process for commercial API manufacturing.
The Novel Approach
In stark contrast, the methodology described in CN110156657B utilizes a modern organometallic strategy known as the Catellani reaction, which elegantly merges ortho-C-H activation with ipso-substitution. By employing aniline derivatives bearing a leaving group (such as I, Br, or Cl) at the ortho-position, the reaction proceeds through a palladium/norbornene cooperative catalytic cycle. This allows for the simultaneous installation of the amine functionality and the construction of the pyrrole ring in one pot. The reaction conditions are remarkably mild, typically operating between 60-150°C in common solvents like toluene, which significantly reduces energy consumption compared to traditional nitration routes. As illustrated in the general reaction scheme below, this approach tolerates a wide variety of substituents on the aniline ring, enabling the rapid generation of diverse chemical libraries for drug discovery programs.
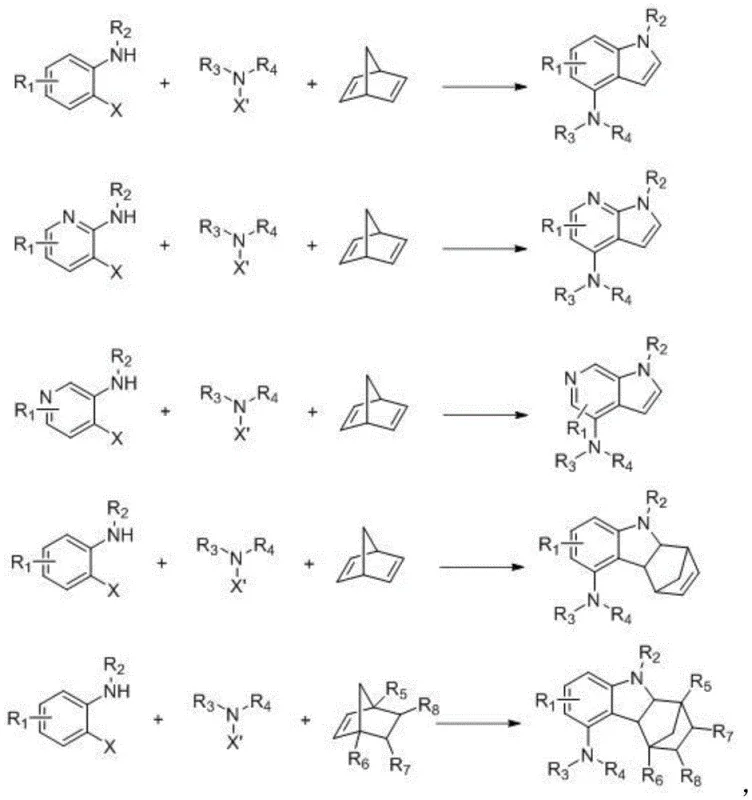
This novel approach not only simplifies the synthetic route but also drastically improves the atom economy and step efficiency. For procurement managers, this translates to a reduction in the number of raw materials required and a decrease in the inventory complexity associated with managing intermediates for multi-step syntheses. The ability to directly access 4-aminoindoles from commercially available anilines means that supply chains can be shortened, reducing lead times and mitigating the risk of supply disruptions associated with specialized nitro-compounds. Moreover, the high yields reported in the patent examples, such as the 82.0% yield obtained in Example 1, demonstrate the practical viability of this method for producing high-purity pharmaceutical intermediates on a meaningful scale.
Mechanistic Insights into Pd/Norbornene Cooperative Catalysis
The success of this synthesis hinges on the intricate interplay between the palladium catalyst and the norbornene mediator, a mechanism that warrants deep technical analysis for process optimization. The catalytic cycle begins with the oxidative addition of the palladium(0) species into the carbon-halogen bond of the ortho-substituted aniline derivative. This is followed by the insertion of norbornene into the Pd-C bond, which acts as a transient mediator to direct the palladium center to the ortho-C-H bond of the aniline ring. This C-H activation step is crucial as it forms a palladacycle intermediate that effectively blocks the typical ipso-substitution pathway, forcing the reaction to proceed through the desired ortho-functionalization. Subsequently, the amine derivative, activated by the base, undergoes nucleophilic attack or transmetallation, leading to the formation of the new C-N bond. Finally, reductive elimination releases the 4-aminoindole product and regenerates the active palladium catalyst, while the norbornene mediator is extruded from the cycle.
Understanding this mechanism is vital for controlling the impurity profile of the final product. One of the key advantages of this catalytic system is its high regioselectivity; the norbornene mediator ensures that functionalization occurs exclusively at the 4-position (ortho to the amino group in the starting aniline), minimizing the formation of regioisomers that are difficult to separate. In traditional electrophilic aromatic substitution, mixtures of 4-, 5-, 6-, and 7-substituted indoles are common, requiring expensive and wasteful chromatographic separations. In this Pd-catalyzed route, the rigid geometry of the palladacycle intermediate dictates the outcome, resulting in a much cleaner crude reaction mixture. This inherent selectivity simplifies downstream processing, as evidenced by the straightforward workup procedures involving simple filtration and column chromatography described in the patent examples. For quality control teams, this means more consistent batch-to-batch purity and reduced analytical burden.
How to Synthesize 4-Aminoindole Efficiently
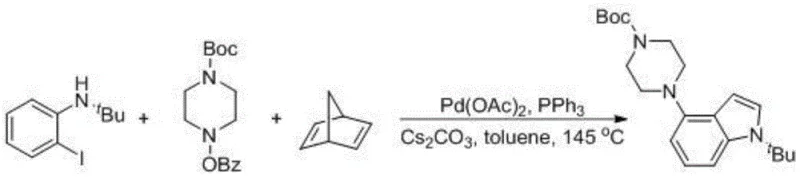
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and reproducibility. The patent provides detailed embodiments that serve as a robust starting point for process development. The standard protocol involves charging a reaction vessel with the aniline derivative, the amine coupling partner, a palladium source such as palladium acetate, a phosphine ligand like triphenylphosphine, and an inorganic base such as cesium carbonate. The choice of base and ligand is critical; for instance, the use of Cs2CO3 and PPh3 in toluene at 145°C has been shown to deliver excellent results. The reaction is typically run under an inert atmosphere to prevent catalyst oxidation, and the norbornene derivative is added as the key mediator. Detailed standardized synthesis steps for this transformation are provided in the guide below.
- Combine aniline derivatives, amine derivatives, palladium catalyst (e.g., Pd(OAc)2), ligand (e.g., PPh3), and base (e.g., Cs2CO3) in a reaction vessel under inert atmosphere.
- Add solvent (e.g., toluene) and norbornene derivative, seal the vessel, and heat the mixture to 60-150°C (optimally 145°C) for approximately 36 hours.
- Upon completion, filter the mixture, wash with ethyl acetate, concentrate the filtrate, and purify the residue via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic methodology offers substantial strategic advantages for organizations looking to optimize their supply chain for indole-based APIs. The shift from a multi-step nitration/reduction sequence to a single-step catalytic coupling fundamentally alters the cost structure of the intermediate. By eliminating the need for hazardous nitration steps and the associated waste treatment costs, manufacturers can achieve significant cost reductions in pharmaceutical intermediate manufacturing. The simplified workflow also reduces the labor hours and equipment occupancy time required per batch, allowing for higher throughput in existing facilities without the need for major capital expenditure on new reactors or safety infrastructure.
- Cost Reduction in Manufacturing: The elimination of multiple isolation and purification steps inherent in the traditional route leads to a drastic simplification of the production process. Traditional methods require handling unstable nitro-intermediates and performing multiple reductions, each adding cost in terms of reagents, solvents, and energy. In contrast, this one-pot method consolidates these operations, significantly lowering the variable cost per kilogram of the final product. Furthermore, the use of commodity chemicals like anilines and norbornene, which are available in bulk quantities, ensures stable pricing and reduces exposure to the volatility of specialized fine chemical markets.
- Enhanced Supply Chain Reliability: Relying on a shorter synthetic route inherently reduces supply chain risk. With fewer steps, there are fewer points of failure where a reaction might stall or an intermediate might fail quality specifications. The starting materials, such as ortho-haloanilines and cyclic amines, are widely produced by multiple global suppliers, ensuring a competitive sourcing landscape. This diversity of supply sources mitigates the risk of single-supplier dependency, a critical factor for supply chain heads managing the continuity of API production. Additionally, the robustness of the reaction conditions allows for flexibility in manufacturing locations, enabling regional production strategies that can further shorten logistics lead times.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents like toluene which are well-understood in large-scale chemical engineering. The workup procedure, involving simple filtration and concentration, avoids complex aqueous extractions that generate large volumes of wastewater. This aligns with increasingly stringent environmental regulations, reducing the burden on effluent treatment plants. The high atom efficiency of the catalytic cycle means less waste is generated per unit of product, supporting sustainability goals. The ability to scale this reaction from gram to kilogram scales without fundamental changes to the chemistry ensures a smooth technology transfer from R&D to commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 4-aminoindole synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a reliable basis for decision-making. Understanding these nuances helps stakeholders evaluate the fit of this technology within their specific project pipelines and manufacturing capabilities.
Q: What is the primary advantage of this 4-aminoindole synthesis method?
A: The primary advantage is the ability to construct the 4-aminoindole skeleton in a single step from readily available aniline derivatives, bypassing the harsh multi-step nitration and reduction sequences required by conventional methods.
Q: Can this method be applied to heterocyclic substrates?
A: Yes, the patent demonstrates successful application on pyridine derivatives, such as N-(tert-butyl)-3-iodopyridin-2-amine, yielding the corresponding azaindole structures with good efficiency.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes standard solvents like toluene and common reagents, with a simple workup procedure involving filtration and chromatography, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Aminoindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN110156657B for accelerating drug development timelines. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly translated into reliable commercial supply. Our state-of-the-art facilities are equipped to handle sensitive palladium-catalyzed reactions with strict adherence to safety and quality protocols. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4-aminoindole intermediate meets the exacting standards required for pharmaceutical applications, minimizing the risk of downstream processing issues.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this efficient synthetic route for their indole-based projects. Whether you require custom synthesis of specific derivatives or large-scale manufacturing of the core scaffold, our technical team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how partnering with us can optimize your supply chain and reduce overall project costs.
