Scalable Synthesis of Mycophenolic Acid-13CD3 for Pharmacokinetic Studies
The pharmaceutical industry's relentless pursuit of precise pharmacokinetic data has elevated the demand for high-quality isotopically labeled standards, particularly for immunosuppressants like Mycophenolic Acid (MPA). Patent CN111153879B discloses a robust and economically viable synthesis method for Mycophenolic Acid-13CD3, a critical stable isotope-labeled analog used extensively in metabolic profiling and bioanalytical assay development. This technology represents a significant leap forward from traditional multi-step total syntheses, offering a streamlined pathway that leverages commercially available starting materials to achieve the target molecule with high fidelity. By utilizing a strategic combination of demethylation, esterification, and a pivotal Mitsunobu coupling, this process ensures that the expensive 13C label is incorporated efficiently at the latest possible stage, minimizing isotopic dilution and maximizing atom economy. For R&D directors and procurement specialists alike, this methodology offers a compelling value proposition: a reliable supply of high-purity reference standards without the prohibitive costs typically associated with custom isotopic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of isotopically labeled Mycophenolic Acid has been plagued by inefficient, linear routes that require the construction of the complex phthalide core from scratch using labeled precursors. These conventional approaches often involve upwards of ten to fifteen synthetic steps, each introducing potential yield losses and impurity profiles that complicate downstream purification. The reliance on early-stage introduction of the isotope means that any loss of material in subsequent steps results in a disproportionate loss of the expensive labeled reagent, driving up the final cost of goods significantly. Furthermore, the harsh conditions often required for core construction can lead to isotopic scrambling or loss of label integrity, compromising the utility of the final standard for quantitative mass spectrometry. From a supply chain perspective, these long routes are fragile; a bottleneck in any single step can halt the entire production campaign, leading to unpredictable lead times and supply discontinuity for critical research programs.
The Novel Approach
In stark contrast, the methodology outlined in CN111153879B adopts a convergent strategy that treats the Mycophenolic Acid scaffold as a modular building block rather than a target for total synthesis. By starting with a readily available Mycophenolic Acid derivative (Compound 1), the process bypasses the most complex and yield-limiting steps of ring formation. The innovation lies in the late-stage functionalization, where the isotopic label is introduced in Step 3 via a Mitsunobu reaction. This approach not only shortens the synthetic timeline drastically but also isolates the handling of the expensive Methanol-13CD3 to a single, high-yielding transformation. The use of mild reaction conditions, such as the LiI-mediated demethylation and ambient temperature hydrolysis, preserves the integrity of the sensitive olefinic side chain and the lactone ring. This strategic redesign transforms the synthesis from a high-risk, low-yield endeavor into a predictable, scalable operation suitable for commercial production of research-grade intermediates.
Mechanistic Insights into LiI-Mediated Demethylation and Mitsunobu Labeling
The success of this synthetic route hinges on two critical chemical transformations: the selective demethylation of the aryl methyl ether and the subsequent regioselective alkylation. The first step utilizes Lithium Iodide (LiI) in the presence of pyridine and 2,4,6-trimethylpyridine at elevated temperatures (135°C). Mechanistically, the iodide ion acts as a potent nucleophile, attacking the methyl group of the ether to generate methyl iodide and the corresponding phenolate salt, which is then protonated upon workup to yield the free phenol (Compound 2). The inclusion of sterically hindered bases like 2,4,6-trimethylpyridine is crucial; they serve to scavenge protons and prevent acid-catalyzed degradation of the sensitive lactone moiety while facilitating the nucleophilic substitution. This step achieves a remarkable yield of 95.6%, demonstrating the robustness of the halide-mediated cleavage under these specific buffered conditions.
Following esterification to protect the carboxylic acid, the core innovation is realized in the Mitsunobu reaction (Step 3), which couples the phenolic intermediate with Methanol-13CD3. 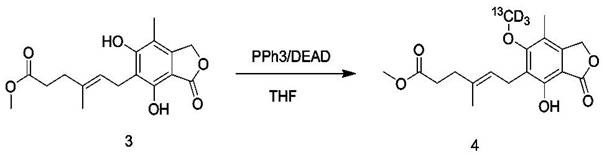 In this mechanism, triphenylphosphine (PPh3) reacts with diethyl azodicarboxylate (DEAD) to form a betaine intermediate, which activates the alcohol (Methanol-13CD3) by converting the hydroxyl group into a good leaving group. Simultaneously, the phenolic oxygen of Compound 3 attacks the activated methyl carbon, displacing the hydrazine byproduct and forming the new C-O bond with the 13C label. This reaction is highly advantageous because it proceeds with inversion of configuration at the alcohol carbon (irrelevant for methanol but critical for chiral alcohols) and tolerates a wide range of functional groups, including the ester and lactone present in the substrate. The reaction is conducted at 0°C to room temperature to minimize side reactions, yielding the labeled intermediate (Compound 4) which is subsequently hydrolyzed to the final acid.
In this mechanism, triphenylphosphine (PPh3) reacts with diethyl azodicarboxylate (DEAD) to form a betaine intermediate, which activates the alcohol (Methanol-13CD3) by converting the hydroxyl group into a good leaving group. Simultaneously, the phenolic oxygen of Compound 3 attacks the activated methyl carbon, displacing the hydrazine byproduct and forming the new C-O bond with the 13C label. This reaction is highly advantageous because it proceeds with inversion of configuration at the alcohol carbon (irrelevant for methanol but critical for chiral alcohols) and tolerates a wide range of functional groups, including the ester and lactone present in the substrate. The reaction is conducted at 0°C to room temperature to minimize side reactions, yielding the labeled intermediate (Compound 4) which is subsequently hydrolyzed to the final acid.
How to Synthesize Mycophenolic Acid-13CD3 Efficiently
The synthesis of Mycophenolic Acid-13CD3 is executed through a logical four-step sequence that balances chemical efficiency with operational simplicity. The process begins with the thermal demethylation of the starting ether, followed by protection of the carboxylic acid as a methyl ester to prevent interference during the labeling step. The pivotal third step involves the Mitsunobu coupling with the isotopic methanol source, after which the protecting group is removed via basic hydrolysis to reveal the final active pharmaceutical ingredient analog. This sequence is designed to minimize purification burdens, with the majority of intermediates being solids that can be isolated via filtration or simple extraction, streamlining the workflow for process chemists.
- Demethylation of the starting phenolic ether using Lithium Iodide and pyridine derivatives at elevated temperatures to yield the free phenol intermediate.
- Esterification of the carboxylic acid side chain using thionyl chloride in methanol to protect the acid functionality prior to labeling.
- Execution of the Mitsunobu reaction using triphenylphosphine, DEAD, and Methanol-13CD3 to introduce the isotopic label selectively at the phenolic position.
- Final hydrolysis of the methyl ester using lithium hydroxide monohydrate to regenerate the carboxylic acid and afford the target Mycophenolic Acid-13CD3.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible benefits in terms of cost stability and supply security. By shifting from a total synthesis model to a semi-synthetic modification of a commercially available bulk chemical, the dependency on custom-made, exotic starting materials is eliminated. This structural change in the supply chain significantly de-risks the sourcing strategy, as the primary raw material (Compound 1) is derived from established fermentation or synthesis pipelines with high market liquidity. Furthermore, the reduction in step count directly correlates to a reduction in labor hours, solvent consumption, and waste disposal costs, all of which contribute to a more competitive pricing structure for the final labeled product without compromising on quality or purity specifications.
- Cost Reduction in Manufacturing: The most significant driver of cost efficiency in this process is the late-stage introduction of the 13C label. In traditional routes, expensive isotopic reagents are often subjected to multiple synthetic transformations, where yield losses compound exponentially. By reserving the Methanol-13CD3 for the third step, the process ensures that nearly 100% of the expensive isotope is retained in the final product, drastically improving the cost-per-gram metric. Additionally, the elimination of transition metal catalysts in favor of organic reagents like LiI and PPh3 removes the need for expensive metal scavenging resins and rigorous heavy metal testing, further lowering the operational expenditure associated with quality control and purification.
- Enhanced Supply Chain Reliability: The reliance on commodity reagents such as Lithium Iodide, Thionyl Chloride, and Triphenylphosphine ensures that the production of Mycophenolic Acid-13CD3 is not held hostage by the lead times of niche specialty chemical suppliers. These reagents are available in bulk quantities from multiple global vendors, providing a buffer against supply shocks. Moreover, the robustness of the reaction conditions—specifically the high tolerance of the Mitsunobu step to moisture and the stability of the intermediates—allows for flexible scheduling and batch sizing. This flexibility enables manufacturers to respond rapidly to fluctuating demand from the biopharmaceutical sector, ensuring consistent availability of this critical research tool.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this route is superior due to its simplified workup procedures. The use of standard extraction and crystallization techniques, rather than preparative HPLC or complex distillations, facilitates easy scale-up from gram to kilogram scales. The waste stream is primarily composed of organic solvents and phosphine oxide byproducts, which are well-understood and manageable within standard pharmaceutical waste treatment protocols. The absence of hazardous heavy metals or explosive reagents simplifies regulatory compliance and reduces the environmental footprint of the manufacturing process, aligning with the increasing industry emphasis on green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Mycophenolic Acid-13CD3. These insights are derived directly from the experimental data and process parameters detailed in the patent literature, providing a transparent view of the technology's capabilities and limitations for potential partners and end-users.
Q: What is the key advantage of this synthesis route for Mycophenolic Acid-13CD3?
A: The primary advantage is the use of a short, 4-step sequence starting from commercially available Mycophenolic Acid derivatives, avoiding the need for de novo construction of the complex phthalide core, which significantly reduces cost and lead time.
Q: How is the isotopic label introduced in this process?
A: The 13C label is introduced selectively via a Mitsunobu reaction in Step 3, using Methanol-13CD3 as the nucleophile to alkylate the phenolic hydroxyl group, ensuring high regioselectivity without affecting other functional groups.
Q: What are the critical purification challenges in this route?
A: The main challenge involves the removal of triphenylphosphine oxide (TPPO) generated during the Mitsunobu step, which requires careful column chromatography or crystallization strategies to meet high-purity specifications for research use.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mycophenolic Acid-13CD3 Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your pharmacokinetic data depends entirely on the quality of your internal standards. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that even complex isotopic labeling projects are executed with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying isotopic enrichment and chemical purity to the highest industry standards, guaranteeing that every batch of Mycophenolic Acid-13CD3 meets your stringent purity specifications. We understand the critical nature of immunosuppressant research and are committed to delivering materials that enable accurate, reproducible scientific outcomes.
We invite you to collaborate with us to optimize your supply chain for labeled intermediates. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our streamlined synthesis route can reduce your overall project costs. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive quotation that reflects our commitment to quality and value.
