Advanced Synthesis of 1,7-Naphthyridine Derivatives for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust and scalable pathways for constructing nitrogen-containing heterocycles, particularly the 1,7-naphthyridine scaffold, which serves as a critical backbone for kinase inhibitors and immunomodulators. Patent CN110885329B discloses a groundbreaking synthetic method for 1,7-naphthyridine derivatives that fundamentally shifts the paradigm from complex transition-metal catalysis to a more efficient Lewis acid-mediated approach. This innovation addresses the longstanding challenges of toxicity and cost associated with traditional routes, offering a streamlined three-step sequence starting from the readily available 2-chloro-3-amino-pyridine. By replacing hazardous reagents and expensive catalysts with safer, commercially viable alternatives, this technology provides a compelling value proposition for manufacturers aiming to optimize their supply chains for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,7-naphthyridine ring system has relied heavily on multi-step sequences involving palladium-catalyzed cross-coupling reactions and the use of highly toxic cyanide sources. As illustrated in the prior art reaction schemes, earlier methods often necessitated the use of 5-bromo-3-methylpyridine-2-carboxylic acid ethyl ester or 2-cyano-3-bromo-5-chloropyridine as starting materials, requiring intricate urethane exchanges and imidization steps. These conventional pathways are fraught with significant operational drawbacks, including the necessity for pressurized reaction vessels and the handling of hypertoxic substances like zinc cyanide, which pose severe environmental and safety risks. Furthermore, the reliance on palladium catalysts introduces substantial cost burdens, not only due to the price of the metal itself but also because of the rigorous downstream processing required to scavenge residual heavy metals to meet stringent pharmaceutical purity standards.
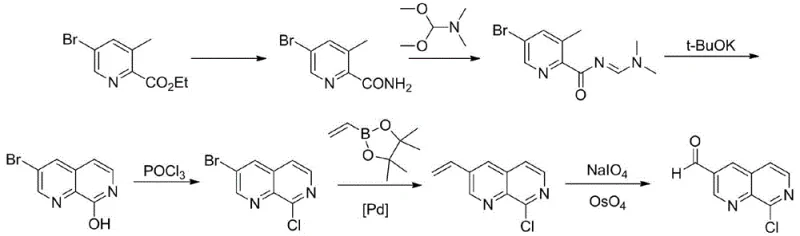
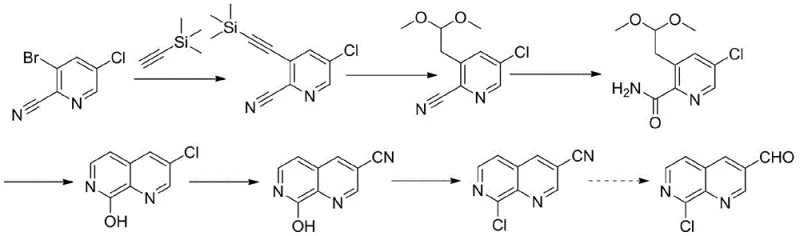
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology described in CN110885329B introduces a remarkably concise and safe alternative that bypasses the need for transition metals entirely. The new strategy leverages a direct ortho-lithiation followed by formylation and a subsequent Lewis acid-catalyzed cyclization to construct the bicyclic core efficiently. This approach utilizes 2-chloro-3-amino-pyridine, a commodity chemical, as the foundational building block, thereby simplifying the raw material sourcing landscape. By eliminating the requirement for specialized high-pressure equipment and toxic cyanide reagents, this novel route drastically reduces the barrier to entry for industrial production while simultaneously enhancing the overall safety profile of the manufacturing facility. The transition from a metal-heavy protocol to a lithium-mediated cyclization represents a significant technological leap towards greener and more sustainable chemical manufacturing.
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization
The core innovation of this synthesis lies in the strategic application of lithium tetrafluoroborate as a Lewis acid catalyst to drive the cyclization of the formylated intermediate with an acrylate derivative. The mechanism begins with the protection of the amino group on the pyridine ring, which is crucial for directing the subsequent lithiation to the correct position and preventing unwanted side reactions at the nitrogen center. Following protection, the use of strong bases like n-butyllithium in the presence of TMEDA facilitates a highly regioselective deprotonation at the position ortho to the protected amine. This generates a reactive organolithium species that undergoes nucleophilic attack on a formylating agent such as N-formylmorpholine, installing the essential aldehyde functionality required for ring closure. The final cyclization step is driven by the activation of the acrylate double bond by the Lewis acid, promoting a Michael addition followed by intramolecular condensation to forge the 1,7-naphthyridine skeleton with high fidelity.
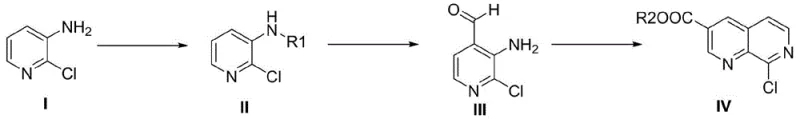
From an impurity control perspective, this mechanistic pathway offers superior manageability compared to radical-based or metal-catalyzed alternatives. The use of specific Lewis acids like lithium tetrafluoroborate ensures that the cyclization proceeds under mild thermal conditions, typically between 50°C and 80°C, which minimizes the formation of thermal degradation byproducts. Moreover, the absence of palladium eliminates the risk of metal-induced side reactions such as homocoupling or dehalogenation, which are common pitfalls in cross-coupling chemistry. The reaction conditions are tuned to favor the formation of the desired ester product, which can be easily purified through standard crystallization or extraction techniques. This high level of selectivity translates directly into a cleaner crude product profile, reducing the load on downstream purification units and ensuring that the final intermediate meets the rigorous quality specifications demanded by global regulatory bodies.
How to Synthesize 8-Chloro-1,7-naphthyridine-3-carboxylate Efficiently
The synthesis of this valuable intermediate is achieved through a logical three-step sequence that balances reactivity with operational simplicity. The process begins with the protection of the starting amine, followed by a low-temperature lithiation-formylation sequence, and concludes with a thermally driven cyclization. Each step has been optimized to maximize yield while minimizing the generation of waste, making it an ideal candidate for transfer to commercial manufacturing sites. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and temperature profiles, are outlined in the guide below to ensure reproducibility and safety during scale-up operations.
- Protect the amino group of 2-chloro-3-amino-pyridine using di-tert-butyl dicarbonate or chloroformates to form Compound II.
- Perform ortho-lithiation on Compound II using n-butyllithium and TMEDA at low temperature, followed by formylation with DMF or N-formylmorpholine to yield Compound III.
- Execute a Lewis acid-catalyzed cyclization reaction between Compound III and an acrylate compound using lithium tetrafluoroborate to generate the target 1,7-naphthyridine ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers transformative benefits that extend far beyond simple yield improvements. The most significant advantage is the complete elimination of palladium catalysts, which are subject to volatile market pricing and complex supply dynamics. By removing this dependency, manufacturers can achieve substantial cost savings not only on raw material acquisition but also on the extensive purification processes traditionally required to reduce metal residues to ppm levels. Additionally, the removal of toxic reagents like zinc cyanide simplifies regulatory compliance and waste disposal protocols, leading to lower operational expenditures related to environmental health and safety management. This streamlined approach ensures a more resilient supply chain that is less vulnerable to disruptions caused by the scarcity of specialized reagents or catalysts.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with inexpensive lithium salts results in a drastic reduction in direct material costs. Furthermore, the simplified workup procedures eliminate the need for costly metal scavenger resins and the associated filtration steps, thereby reducing both consumable expenses and processing time. The overall process efficiency is enhanced by the high yields observed in each step, which maximizes the throughput of the existing reactor infrastructure without requiring capital-intensive upgrades.
- Enhanced Supply Chain Reliability: Sourcing strategies are significantly de-risked by the use of commodity chemicals such as 2-chloro-3-amino-pyridine and common acrylates, which are widely available from multiple global suppliers. This diversification of the supply base prevents bottlenecks that often occur when relying on single-source specialty reagents or custom-synthesized starting materials. The robustness of the reaction conditions also means that production can be maintained consistently across different manufacturing sites, ensuring continuity of supply for downstream API synthesis.
- Scalability and Environmental Compliance: The process operates under atmospheric pressure and moderate temperatures, making it inherently safer and easier to scale from pilot plant to full commercial production. The absence of hypertoxic cyanide sources aligns perfectly with modern green chemistry initiatives, reducing the environmental footprint of the manufacturing process. This compliance advantage facilitates faster regulatory approvals and reduces the administrative burden associated with handling hazardous materials, allowing for a smoother and faster time-to-market for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy technologies in terms of safety, cost, and scalability. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this new synthesis route improve safety compared to traditional methods?
A: Traditional methods often rely on highly toxic reagents like zinc cyanide and require pressurized equipment for coupling reactions. This novel route eliminates the need for cyanide sources and palladium catalysts, significantly reducing EHS risks and operational hazards.
Q: What are the cost advantages of avoiding palladium catalysts in this process?
A: By replacing expensive palladium-catalyzed coupling steps with a lithium tetrafluoroborate mediated cyclization, the process removes the high cost of precious metal catalysts and the subsequent expensive purification steps required to remove residual heavy metals from the final API intermediate.
Q: Is this synthetic method suitable for large-scale industrial production?
A: Yes, the method utilizes commercially available raw materials like 2-chloro-3-amino-pyridine and operates under standard atmospheric pressure without complex special operations. The simplified workflow and high yields make it highly adaptable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,7-Naphthyridine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN110885329B and is fully prepared to execute this route with precision and efficiency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory concept to industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,7-naphthyridine intermediate delivered meets the highest international standards.
We invite you to collaborate with us to leverage this cost-effective and safe synthesis technology for your next development program. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your budget. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a reliable supply of high-quality intermediates for your vital therapeutic projects.
