Scalable Synthesis of Cyclobutene Intermediates for CXC Chemokine Inhibitors
The pharmaceutical industry continuously seeks robust synthetic routes for complex intermediates, particularly those targeting CXC chemokine-mediated diseases. Patent CN101932553A discloses a groundbreaking methodology for preparing 1,2-substituted 3,4-dioxo-1-cyclobutene compounds, which serve as critical scaffolds in modern drug discovery. This technology addresses long-standing challenges regarding the stability and safety of key intermediates, specifically by introducing in situ generation techniques and stable salt forms. For R&D directors and procurement specialists, understanding this patent is vital as it offers a pathway to more reliable manufacturing processes. The innovation lies not just in the final molecule but in the strategic manipulation of reaction conditions to bypass hazardous isolation steps. By leveraging these insights, manufacturers can achieve higher purity profiles while mitigating risks associated with handling sensitive chemical species. This report analyzes the technical merits and commercial implications of adopting this advanced synthesis strategy for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for cyclobutene derivatives often rely on the isolation of dialkyl squarylates, which present significant occupational health hazards due to their classification as potent skin sensitizers and irritants. Furthermore, conventional methods frequently utilize unstable free base intermediates, such as 3-amino-2-hydroxy-benzamide, which degrade rapidly during storage and transport, leading to inconsistent batch quality and yield losses. The necessity to purify these unstable intermediates before subsequent coupling reactions adds unnecessary complexity and cost to the manufacturing workflow. Additionally, prior art processes often require multiple solvent exchanges and harsh conditions that generate undue levels of impurities, complicating downstream purification and potentially compromising the safety profile of the final active pharmaceutical ingredient. These inefficiencies create bottlenecks in supply chains, making it difficult to guarantee consistent delivery schedules for large-scale production campaigns.
The Novel Approach
The novel approach detailed in the patent circumvents these issues by employing an in situ formation strategy for dialkyl squarylates, thereby eliminating the need to isolate these hazardous materials entirely. By reacting squaric acid directly with trialkyl orthoformates in the presence of an alcohol solvent, the reactive ester is generated and immediately consumed in the subsequent coupling step. This telescoped process significantly reduces exposure risks and simplifies the operational workflow. Moreover, the invention advocates for the use of stable salt forms of amino-hydroxybenzamide intermediates, which can be prepared remotely, stored for extended periods, and transported without degradation. This decoupling of intermediate preparation from final assembly allows for greater flexibility in manufacturing scheduling and inventory management. The result is a streamlined, safer, and more economically viable process that maintains high chemical integrity throughout the synthesis.
Mechanistic Insights into In Situ Squarylate Formation and Coupling
The core mechanistic advantage of this technology lies in the acid-catalyzed esterification of squaric acid using trialkyl orthoformates, which proceeds efficiently under reflux conditions to generate the dialkyl squarylate intermediate. This reactive species is then immediately engaged in a nucleophilic substitution reaction with an amino-hydroxybenzamide salt in the presence of a tertiary amine base. The use of a stable salt form for the benzamide component ensures that the nucleophile remains intact until the precise moment of reaction, preventing premature decomposition or side reactions that typically plague free base variants. Temperature control during this coupling phase is critical, with optimal results observed at low temperatures ranging from -5°C to +5°C to minimize impurity formation. The subsequent workup involves careful pH adjustment and crystallization protocols that leverage solubility differences to isolate the intermediate with high purity. This precise control over reaction kinetics and thermodynamics is what enables the consistent production of high-quality intermediates suitable for stringent pharmaceutical applications.
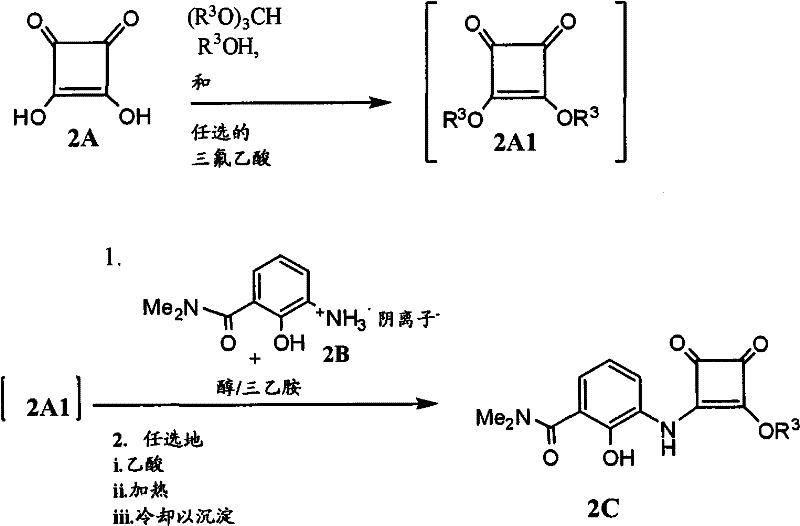
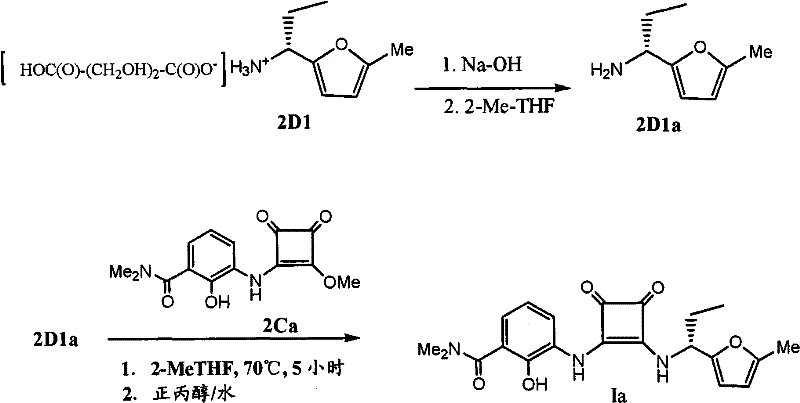
Following the formation of the squarylate intermediate, the process advances to the coupling with an aminofuran component, which is generated in situ from its corresponding salt form. The patent highlights the unexpected benefit of using 2-methyl-tetrahydrofuran (2-MeTHF) as a solvent for this coupling step, which significantly improves the impurity profile compared to traditional solvents. The aminofuran salt is treated with an aqueous base to release the free base, which partitions into the organic layer, ready for immediate reaction with the squarylate intermediate. This phase separation technique allows for the removal of inorganic byproducts before the coupling occurs, further enhancing the purity of the final product. The reaction is driven to completion by heating, followed by a sophisticated crystallization process involving solvent swaps and temperature cycling to control crystal size and morphology. Such attention to physical form is crucial for downstream processing, ensuring that the material flows well and dissolves predictably in subsequent formulation steps.
How to Synthesize 1,2-Substituted 3,4-Dioxo-1-Cyclobutene Compounds Efficiently
Implementing this synthesis requires strict adherence to the patented sequence of in situ esterification followed by controlled coupling and crystallization. The process begins with the generation of the dialkyl squarylate in an alcohol solvent, followed by the addition of the amino-hydroxybenzamide salt and a tertiary amine base at low temperatures. After isolating the intermediate, it is coupled with the aminofuran free base in 2-MeTHF under heated conditions. The detailed standardized synthesis steps见下方的指南 ensure reproducibility and compliance with quality standards. Operators must monitor reaction temperatures closely and utilize the specified solvent exchange protocols to maximize yield and purity. This methodology represents a significant upgrade over legacy processes, offering a robust framework for manufacturing these complex pharmaceutical intermediates at scale.
- Prepare dialkyl squarylate in situ by reacting squaric acid with trialkyl orthoformate in an alcohol solvent under reflux conditions.
- React the in situ formed dialkyl squarylate with an amino-hydroxybenzamide salt in the presence of a tertiary amine base at controlled low temperatures.
- Couple the resulting intermediate with a free base aminofuran derived from its salt form using aqueous base extraction and subsequent heating in 2-MeTHF.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible operational efficiencies and risk mitigation strategies. By eliminating the need to handle hazardous dialkyl squarylates as isolated materials, facilities can reduce safety compliance costs and insurance premiums associated with handling potent sensitizers. The ability to produce and store stable salt intermediates allows for decoupled manufacturing schedules, meaning key building blocks can be stockpiled during off-peak times to buffer against supply disruptions. This flexibility enhances overall supply chain resilience, ensuring that production lines remain active even if upstream raw material deliveries face delays. Furthermore, the simplified workflow reduces the number of unit operations required, which lowers energy consumption and waste generation, aligning with broader sustainability goals. These qualitative improvements collectively contribute to a more stable and cost-effective supply environment for critical pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The in situ generation of reactive intermediates eliminates entire isolation and purification steps, which drastically reduces solvent usage and waste disposal costs associated with traditional batch processing. By avoiding the handling of hazardous materials, facilities also save on specialized containment equipment and personal protective gear, leading to substantial operational expenditure savings. The higher purity of the crude product reduces the burden on downstream purification, minimizing yield losses during recrystallization and chromatography. Consequently, the overall cost of goods sold is optimized through a leaner, more efficient chemical process that maximizes resource utilization.
- Enhanced Supply Chain Reliability: The stability of the salt form intermediates allows for long-term storage without significant degradation, enabling manufacturers to maintain strategic reserves of key materials. This capability mitigates the risk of production stoppages caused by just-in-time delivery failures or raw material shortages. Additionally, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output even when sourcing from different suppliers. This reliability is paramount for maintaining continuous supply to downstream drug product manufacturers who depend on uninterrupted availability of high-quality intermediates.
- Scalability and Environmental Compliance: The process is designed with commercial scale-up in mind, utilizing common solvents and reagents that are readily available in large quantities. The reduction in hazardous waste generation and the elimination of skin sensitizers from the workflow simplify environmental permitting and regulatory compliance. Safer chemistry principles are inherently built into the design, reducing the potential for workplace accidents and exposure incidents. This alignment with green chemistry initiatives not only protects the workforce but also enhances the corporate reputation of manufacturers as responsible stewards of environmental and social governance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. They are derived from the specific pain points identified in the background art and the beneficial effects demonstrated in the patent examples. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the objective technical data provided in the patent documentation, ensuring accuracy and reliability for decision-making purposes. Stakeholders are encouraged to review these points when assessing the potential for technology transfer or licensing agreements.
Q: How does the in situ formation of dialkyl squarylate improve safety?
A: By generating the dialkyl squarylate intermediate directly within the reaction mixture, the process eliminates the need to isolate and handle pure dialkyl squarylates, which are known potent skin sensitizers and irritants, thereby significantly enhancing operational safety.
Q: Why are salt forms of intermediates preferred over free bases?
A: Salt forms of intermediates like the amino-hydroxybenzamide exhibit superior stability compared to their free base counterparts, allowing for easier storage, transportation, and handling without degradation, which is critical for maintaining supply chain continuity.
Q: What are the scalability benefits of this synthetic route?
A: The process utilizes commercially available starting materials and avoids unstable intermediates, enabling robust scale-up from laboratory to industrial production while maintaining high purity and consistent yield through controlled crystallization steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Substituted 3,4-Dioxo-1-Cyclobutene Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at adapting complex patented routes like the one described in CN101932553A to fit large-scale reactor configurations while maintaining stringent purity specifications. We understand the critical nature of pharmaceutical intermediates and operate rigorous QC labs to ensure every batch meets the highest standards of quality and consistency. Our infrastructure supports the safe handling of sensitive chemistries, ensuring that safety and efficiency are never compromised during scale-up. Partnering with us means gaining access to a supply chain that is both resilient and technically sophisticated.
We invite you to contact our technical procurement team to discuss your specific requirements for cyclobutene intermediates. Request a Customized Cost-Saving Analysis to understand how implementing this novel synthesis route can optimize your budget. We are ready to provide specific COA data and route feasibility assessments tailored to your project timelines. Let us help you secure a reliable supply of high-quality intermediates that drive your drug development programs forward with confidence and speed.
