Advanced Manufacturing of 3-Chloro-5-Aminobenzotrifluoride: A Strategic Route for Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways for complex fluorinated intermediates, particularly those serving as building blocks for novel therapeutics. Patent CN101157618A introduces a meticulously engineered preparation technology for 3-chloro-5-amino-benzotrifluoride, a critical intermediate in the synthesis of next-generation medicines. This patent outlines a rational five-step sequence starting from 4-chloro-2-trifluorotoluidine, employing a classic protection-deprotection strategy to achieve high regioselectivity. The process leverages standard chemical transformations including acetylation, nitration, hydrolysis, diazotization-deamination, and reduction. For R&D directors and procurement specialists, this methodology represents a significant advancement over less predictable direct substitution methods, offering a clear pathway to high-purity API intermediates with controlled impurity profiles. The total recovery rate reported in the patent reaches 41.18%, demonstrating the cumulative efficiency of the individual high-yield steps.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing meta-substituted anilines often suffer from poor regioselectivity and harsh reaction conditions that compromise yield and purity. Direct nitration of chlorobenzotrifluorides can lead to complex mixtures of isomers that are difficult and costly to separate, creating bottlenecks in the supply chain. Furthermore, older methods frequently rely on expensive catalysts or extreme temperatures that pose safety risks and increase energy consumption. In the context of cost reduction in fine chemical manufacturing, these inefficiencies translate to higher raw material waste and extended processing times. The inability to precisely control the position of substituents on the aromatic ring often results in batches that fail stringent quality specifications, leading to significant financial losses for manufacturers attempting to scale these processes.
The Novel Approach
The technology disclosed in CN101157618A circumvents these challenges by utilizing the amino group of the starting material as a temporary directing handle. By first acetylating the amine, the process protects the nitrogen from oxidation while activating the ring for specific electrophilic substitution. This strategic maneuver ensures that the nitro group is installed exactly where needed relative to the trifluoromethyl and chloro substituents. Subsequent removal of the original amino group via diazotization clears the way for the final reduction step, which installs the desired amino functionality at the target position. This logical progression transforms a potentially chaotic substitution pattern into a linear, predictable synthesis. For a reliable pharmaceutical intermediate supplier, this level of control is essential for guaranteeing batch-to-b consistency and meeting the rigorous demands of global regulatory bodies.
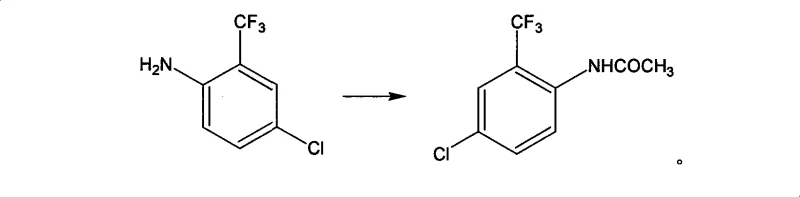
Mechanistic Insights into the Protection-Directed Synthesis
The core of this synthetic strategy lies in the electronic modulation of the aromatic ring through the acetylation step. As shown in the reaction scheme, the conversion of the free amine to the acetamide reduces the electron-donating power of the nitrogen, preventing poly-nitration and oxidative degradation during the subsequent acidic nitration step. The acetamide group acts as a moderate activator, directing the incoming nitronium ion to the position ortho to itself, which corresponds to the meta position relative to the trifluoromethyl group in the final numbering. This precise control is vital for avoiding the formation of regioisomers that would otherwise require chromatographic purification, a technique that is rarely feasible on a multi-ton scale. The use of acetic anhydride and acetic acid provides a mild yet effective medium for this transformation, ensuring that the sensitive trifluoromethyl group remains intact.
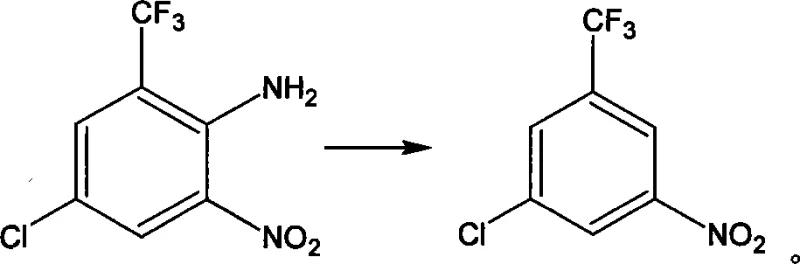
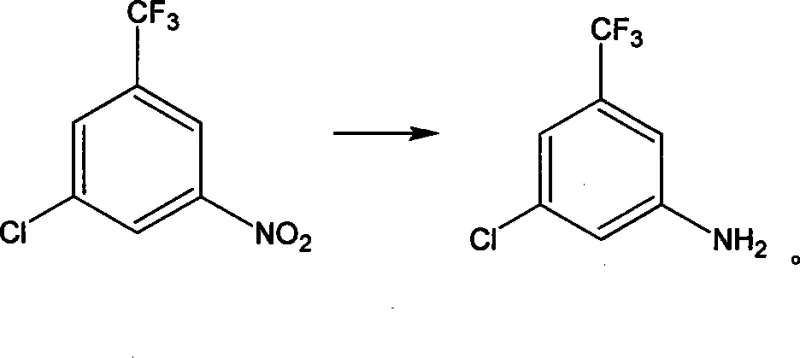
Following nitration and deacetylation, the mechanism shifts to the removal of the original nitrogen functionality. The deamination step involves the formation of a diazonium salt using sodium nitrite and sulfuric acid at low temperatures, followed by reductive elimination using hypophosphorous acid. This effectively replaces the amino group with a hydrogen atom, resetting the substitution pattern to the desired scaffold. Finally, the nitro group introduced earlier is reduced to an amine using iron powder in an acidic medium. This classical Bechamp reduction is highly selective for nitro groups and tolerates the presence of the chloro and trifluoromethyl substituents. The result is a clean conversion to 3-chloro-5-aminobenzotrifluoride, with the patent reporting contents exceeding 98% at various stages, highlighting the efficacy of this mechanistic design in minimizing side products.
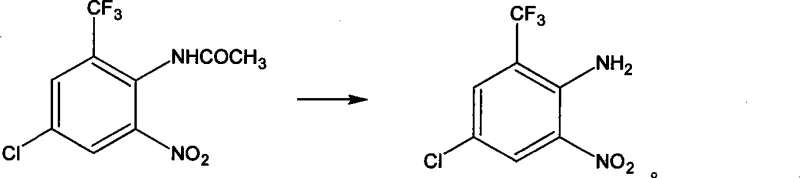
How to Synthesize 3-Chloro-5-Aminobenzotrifluoride Efficiently
The synthesis protocol described in the patent provides a detailed roadmap for executing this transformation with high fidelity. The process begins with the careful control of temperature during acetylation to prevent exotherms, followed by a controlled nitration where the addition rate of nitric acid is critical to maintaining selectivity. The subsequent hydrolysis requires reflux conditions to ensure complete removal of the acetyl group, while the deamination step demands strict temperature control to stabilize the diazonium intermediate. The final reduction utilizes inexpensive iron powder, making the process economically attractive while maintaining high chemical purity. Operators must adhere to the specified stoichiometric ratios, particularly the excess of acetic anhydride and the precise amount of reducing agents, to maximize the overall yield of 41.18%.
- Acetylation of 4-chloro-2-trifluorotoluidine using acetic anhydride and acetic acid to protect the amino group.
- Nitration of the protected intermediate followed by hydrolytic deacetylation to reveal the nitro-aniline structure.
- Diazotization and deamination to remove the original amino group, followed by catalytic reduction of the nitro group to the final amine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial benefits for supply chain stability and cost management. The reliance on commodity chemicals such as acetic acid, sulfuric acid, and iron powder means that the process is not vulnerable to the price volatility associated with precious metal catalysts or specialized reagents. This accessibility of raw materials significantly de-risks the procurement process, ensuring that production schedules are not disrupted by supply shortages. For procurement managers, this translates to more predictable budgeting and the ability to secure long-term contracts for commercial scale-up of complex fluorinated compounds without fear of sudden cost spikes. The robustness of the chemistry also implies lower failure rates in production, further enhancing the reliability of supply.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of standard unit operations drastically lowers the operational expenditure. By avoiding complex purification steps like column chromatography and relying instead on crystallization and filtration, the process reduces solvent consumption and waste disposal costs. The high yields reported in the individual steps (e.g., 98% for acetylation) minimize raw material waste, directly contributing to a lower cost of goods sold (COGS). This efficiency allows manufacturers to offer competitive pricing for high-purity API intermediates while maintaining healthy margins.
- Enhanced Supply Chain Reliability: The use of stable intermediates and common reagents ensures that the supply chain is resilient against external shocks. The process does not depend on single-source suppliers for exotic catalysts, allowing for flexible sourcing strategies. Furthermore, the scalability of the reaction conditions—ranging from simple stirring and heating to standard reflux—means that production can be easily ramped up to meet surging demand. This flexibility is crucial for reducing lead time for high-purity intermediates in a fast-paced pharmaceutical market where time-to-market is a critical competitive advantage.
- Scalability and Environmental Compliance: The process generates waste streams that are relatively straightforward to treat, primarily consisting of aqueous acidic waste and iron sludge, which are well-understood in industrial wastewater treatment. The absence of heavy metals simplifies the environmental compliance burden, reducing the costs associated with hazardous waste disposal. The ability to run these reactions in standard glass-lined or stainless steel reactors makes the technology readily transferable to existing manufacturing facilities, accelerating the timeline from pilot plant to full commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 3-chloro-5-aminobenzotrifluoride based on the patented methodology. Understanding these details is crucial for partners evaluating the feasibility of integrating this intermediate into their own supply chains. These insights reflect the practical realities of scaling fine chemical processes and the specific advantages offered by this protection-deprotection strategy.
Q: What is the primary advantage of this synthetic route for 3-chloro-5-aminobenzotrifluoride?
A: The primary advantage is the strategic use of the starting aniline as a temporary directing and blocking group. This allows for precise regioselective nitration at the desired position, which is subsequently removed via deamination, ensuring high positional purity of the final product.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states that each step utilizes conventional unit operations such as standard reflux, filtration, and washing. The use of common reagents like acetic anhydride, sulfuric acid, and iron powder facilitates easy scale-up from laboratory to commercial tonnage without requiring exotic catalysts.
Q: How does this method address impurity control?
A: The multi-step protection and deprotection strategy minimizes side reactions. Specifically, acetylation prevents over-oxidation of the amine during nitration, while the final reduction step using iron powder is a robust method known for generating clean amino products with minimal byproduct formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Chloro-5-Aminobenzotrifluoride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. Our expertise in fluorinated chemistry positions us as a strategic partner capable of navigating the complexities of multi-step syntheses like the one described in CN101157618A.
We invite you to collaborate with us to optimize your supply chain and accelerate your drug development timelines. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term goals. Let us be your trusted partner in bringing innovative therapies to market efficiently and reliably.
