Advanced Chiral Resolution Technology for High-Purity Pharmaceutical Intermediates Manufacturing
Advanced Chiral Resolution Technology for High-Purity Pharmaceutical Intermediates Manufacturing
The landscape of chiral pharmaceutical manufacturing is constantly evolving, driven by the relentless demand for higher enantiomeric purity and more sustainable production methodologies. In this context, the technical disclosure found in patent CN102757367A presents a significant advancement in the splitting process of racemic ethyl benzene sulfonic acid, a critical intermediate in the synthesis of various bioactive compounds. This innovation addresses the longstanding challenges associated with traditional resolution methods, particularly the reliance on costly and scarce optical alkaline resolving agents. By leveraging a novel approach that utilizes L-p-hydroxyphenylglycine, the process offers a streamlined pathway to obtain D-ethyl benzene sulfonic acid with remarkable efficiency. For global procurement leaders and R&D directors, understanding the nuances of this technology is essential for optimizing supply chains and ensuring the consistent quality of fine chemical intermediates. The following analysis delves deep into the mechanistic and commercial implications of this patented methodology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the resolution of racemic ethyl benzene sulfonic acid has been hindered by significant economic and logistical bottlenecks inherent to conventional splitting technologies. Traditional processes predominantly rely on optically active alkaline resolving agents, which are characterized by their exorbitant market prices and limited availability in the global chemical supply chain. This dependency creates substantial vulnerability for manufacturers, as fluctuations in the supply of these specialized reagents can lead to production delays and unpredictable cost structures. Furthermore, the chemical handling of these traditional agents often requires stringent conditions to prevent racemization, adding layers of complexity to the operational workflow. The inefficiency of these older methods is not merely a financial burden but also a technical constraint that limits the scalability of production. Consequently, pharmaceutical companies seeking reliable pharmaceutical intermediates suppliers have long sought alternative routes that mitigate these risks without compromising on the stereochemical integrity of the final product.
The Novel Approach
In stark contrast to the legacy systems, the novel approach detailed in the patent introduces a robust and economically viable strategy centered around the use of L-p-hydroxyphenylglycine as the primary resolving agent. This method fundamentally shifts the paradigm by utilizing a material that is not only more accessible but also integrates seamlessly into existing synthetic workflows commonly found in the production of beta-lactam antibiotics like Amoxicillin. The process is characterized by its simplicity, involving a straightforward sequence of dissolution, reflux, cooling, and separation that minimizes the need for exotic equipment or hazardous conditions. By forming a complex salt that exhibits low water solubility, the technique ensures high separation efficiency, allowing for the effective isolation of the desired D-enantiomer. This transition represents a strategic leap forward in cost reduction in pharmaceutical intermediates manufacturing, offering a pathway that is both chemically elegant and commercially sound for large-scale operations.
Mechanistic Insights into L-HPG Catalyzed Resolution
To fully appreciate the technical merit of this process, one must examine the underlying chemical interactions that drive the chiral discrimination. The core of this mechanism lies in the formation of a diastereomeric salt between the racemic ethyl benzene sulfonic acid and the chiral L-p-hydroxyphenylglycine. When these components are combined in an aqueous acidic medium under reflux, the specific spatial arrangement of the L-HPG molecule preferentially binds with the D-enantiomer of the sulfonic acid. This selective interaction results in the precipitation of a complex salt, specifically the (-)-HPG-(+)-PES salt, which possesses distinct physical properties, notably its low solubility in water at reduced temperatures. The subsequent cooling to 20°C triggers the crystallization of this specific diastereomer, effectively pulling the desired isomer out of the solution while leaving the unwanted L-enantiomer in the mother liquor. This precise control over crystallization kinetics is what enables the high purity levels observed in the final product.
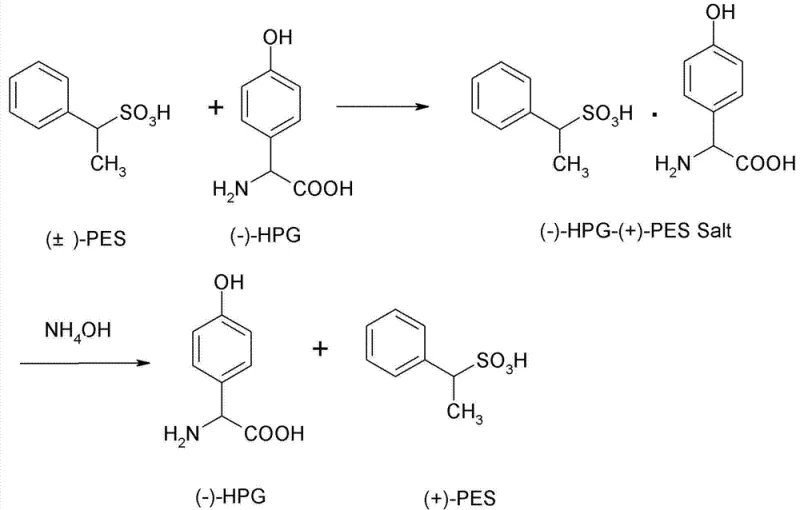
Following the initial separation, the process employs a neutralization step using an inorganic base, such as sodium hydroxide or ammonium hydroxide, to break the salt bond and release the free D-ethyl benzene sulfonic acid. The choice of base and the control of pH between 4 and 8 are critical parameters that influence the recovery rate and the optical purity of the isolated compound. The patent data indicates that this method can achieve a liquid phase purity of 98% with an optical activity of 21°, demonstrating the efficacy of the L-HPG system in maintaining stereochemical integrity. Furthermore, the mother liquor, which contains the unreacted L-enantiomer, is not discarded but is subjected to racemization under alkaline conditions. This recycling loop is a testament to the green chemistry principles embedded in the design, ensuring that raw materials are maximized and waste is minimized, a key consideration for modern commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize D-ethyl benzene sulfonic acid Efficiently
Implementing this resolution process in a production environment requires strict adherence to the optimized parameters outlined in the technical documentation to ensure reproducibility and safety. The procedure begins with the preparation of the resolving solution, where precise molar ratios of L-p-hydroxyphenylglycine and concentrated sulfuric acid are dissolved in water to create the acidic medium necessary for salt formation. Operators must maintain a reflux state during the addition of the racemic substrate to ensure complete interaction before initiating the cooling phase for crystallization. The detailed standardized synthesis steps see the guide below, which outlines the specific temperature controls and separation techniques required to maximize yield. Adhering to these protocols is essential for achieving the high-purity ethyl benzene sulfonic acid specifications demanded by downstream pharmaceutical applications.
- Dissolve 1mol of L-p-hydroxyphenylglycine and 1mol of concentrated sulfuric acid in 20mol of water under reflux conditions.
- Add aqueous solution containing racemic ethyl benzene sulfonic acid, maintain reflux for 1 hour, then cool to 20°C to separate the complex salt.
- Neutralize the complex salt with inorganic base to pH 4-8, crystallize, and remove salts from mother liquor to obtain D-ethyl benzene sulfonic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this resolution technology offers profound benefits that extend far beyond simple chemical conversion. The primary advantage lies in the drastic simplification of the supply chain for resolving agents; by shifting away from rare, expensive optical alkalines to L-p-hydroxyphenylglycine, manufacturers can leverage a commodity chemical that is produced in vast quantities for the antibiotic industry. This shift significantly reduces the risk of supply disruption and stabilizes raw material costs, providing a more predictable financial model for long-term production planning. Additionally, the simplified workflow reduces the operational overhead associated with handling hazardous or difficult-to-source reagents, thereby lowering the total cost of ownership for the manufacturing process. For supply chain heads, this translates to enhanced supply chain reliability and the ability to secure reducing lead time for high-purity pharmaceutical intermediates through more robust and resilient production lines.
- Cost Reduction in Manufacturing: The elimination of expensive, specialty resolving agents directly impacts the bottom line by lowering the variable cost per kilogram of the final product. Since L-p-hydroxyphenylglycine is a widely available intermediate, its market price is subject to stable competitive forces rather than the volatility of niche chemical markets. Furthermore, the ability to recycle the mother liquor through racemization means that the effective consumption of raw materials is optimized, leading to substantial cost savings over the lifecycle of the production campaign. This economic efficiency allows companies to offer more competitive pricing to their clients while maintaining healthy margins, a critical factor in the highly competitive fine chemicals sector.
- Enhanced Supply Chain Reliability: Dependence on single-source or scarce reagents is a major vulnerability in global chemical supply chains, often leading to bottlenecks that delay product launches. By utilizing a resolving agent that is integral to the mass production of common antibiotics, this process ensures a steady and reliable flow of materials even during periods of market tightness. The robustness of the process conditions, which do not require extreme pressures or temperatures, further enhances equipment uptime and reduces maintenance-related downtime. This reliability is paramount for meeting the just-in-time delivery expectations of major pharmaceutical partners who depend on consistent intermediate supplies for their own API manufacturing schedules.
- Scalability and Environmental Compliance: The simplicity of the aqueous-based system facilitates easy scale-up from pilot plant to full commercial production without the need for complex engineering modifications. The use of water as the primary solvent and the ability to recover materials through racemization aligns well with increasingly stringent environmental regulations regarding waste discharge and solvent usage. This environmental compatibility not only reduces the costs associated with waste treatment but also enhances the corporate sustainability profile of the manufacturer. Such attributes are increasingly important for qualifying as a preferred vendor for multinational corporations that prioritize green chemistry and sustainable sourcing in their supplier audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology, derived directly from the patent specifications and industry best practices. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios. The answers provided reflect the specific capabilities and limitations documented in the intellectual property, ensuring transparency and accuracy for decision-makers. We encourage stakeholders to review these insights to gain a comprehensive understanding of the operational requirements and potential benefits associated with this advanced splitting method.
Q: What is the primary advantage of using L-p-hydroxyphenylglycine for resolution?
A: Unlike traditional alkaline resolving agents which are expensive and difficult to source, L-p-hydroxyphenylglycine is a readily available intermediate often used in Amoxicillin synthesis, significantly reducing raw material costs and supply chain risks.
Q: What purity levels can be achieved with this splitting process?
A: The patented process demonstrates the capability to achieve liquid phase purity of approximately 98% with an optical activity of 21°, ensuring high-quality standards suitable for downstream pharmaceutical applications.
Q: Is the mother liquor recyclable in this resolution method?
A: Yes, the process includes a specific step for racemization of the mother liquor under alkaline conditions, allowing for the recovery and reuse of materials, which enhances overall atom economy and reduces waste disposal costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl Benzene Sulfonic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced chemical processes requires more than just laboratory success; it demands industrial expertise and unwavering commitment to quality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the L-HPG resolution process can be seamlessly transitioned to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Ethyl Benzene Sulfonic Acid meets the exacting standards required for pharmaceutical synthesis. We understand the critical nature of chiral purity and are dedicated to delivering products that support the development of life-saving medications globally.
We invite you to collaborate with us to explore how this technology can optimize your specific production needs and drive value across your organization. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can become a cornerstone of your supply chain strategy for high-performance chemical intermediates.
