Advanced Organocatalytic Manufacturing of C-8 Lactam Lactone Intermediates for Renin Inhibitors
The pharmaceutical industry continuously seeks more efficient and safer pathways for synthesizing complex active pharmaceutical ingredients (APIs), particularly for cardiovascular treatments like renin inhibitors. Patent CN101679213A introduces a groundbreaking methodology for the preparation of C-8 lactam lactone compounds, which serve as critical intermediates in the synthesis of potent renin inhibitors such as Aliskiren. This invention represents a significant technological leap by utilizing organocatalytic Michael and Henry reactions to construct the core molecular framework with high stereochemical control. Unlike previous generations of synthesis that relied heavily on stoichiometric chiral auxiliaries and hazardous reagents, this new approach leverages catalytic amounts of chiral amines to drive asymmetry. The core target of this innovation is the compound of Formula (I), a 5-(5-oxo-tetrahydro-furan-2-yl)pyrrolidin-2-one derivative, which possesses the precise stereochemistry required for biological activity. 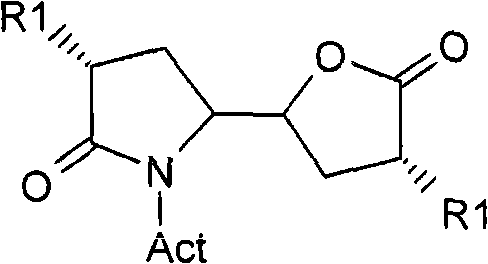 By establishing a robust protocol for generating these complex bicyclic structures, the patent addresses the growing demand for reliable pharmaceutical intermediates supplier capabilities that can deliver high-purity materials while adhering to stringent environmental and safety standards. The ability to synthesize these molecules through a streamlined sequence not only reduces the overall step count but also minimizes the generation of toxic byproducts, aligning with modern green chemistry principles essential for sustainable drug manufacturing.
By establishing a robust protocol for generating these complex bicyclic structures, the patent addresses the growing demand for reliable pharmaceutical intermediates supplier capabilities that can deliver high-purity materials while adhering to stringent environmental and safety standards. The ability to synthesize these molecules through a streamlined sequence not only reduces the overall step count but also minimizes the generation of toxic byproducts, aligning with modern green chemistry principles essential for sustainable drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of renin inhibitor intermediates like those described in WO2007/045420 involved multi-step sequences that were both economically and environmentally burdensome. Traditional routes frequently employed Evans auxiliaries to induce chirality, which necessitated the use of stoichiometric amounts of expensive chiral reagents that had to be attached and subsequently removed, adding unnecessary mass and cost to the process. Furthermore, the introduction of the nitrogen atom often relied on azide chemistry, which poses significant safety risks due to the explosive nature of azides and the toxicity of associated reagents. These conventional methods also struggled with scalability, as the accumulation of impurities during long synthetic sequences made purification difficult and yields unpredictable at larger scales. The reliance on heavy metal catalysts or harsh reaction conditions further complicated waste management, creating substantial hurdles for cost reduction in pharmaceutical intermediates manufacturing. Consequently, there was a pressing need for an alternative synthetic pathway that could bypass these inefficiencies while maintaining the rigorous stereochemical fidelity required for therapeutic efficacy.
The Novel Approach
The methodology disclosed in CN101679213A offers a transformative solution by replacing stoichiometric chiral induction with organocatalysis, specifically utilizing chiral amine catalysts to facilitate asymmetric Michael and Henry reactions. This novel approach allows for the construction of the C-8 lactam lactone skeleton in a more direct manner, avoiding the need for dangerous azide chemistry entirely. By using inexpensive starting materials such as isovaleraldehyde and nitroethylene precursors, the process significantly lowers the raw material costs associated with production. A key advantage of this new route is its compatibility with continuous flow processing, which enhances reaction control and safety while enabling easier scale-up from laboratory to commercial production volumes. The elimination of heavy metal catalysts simplifies the downstream purification process, reducing the burden on quality control laboratories to test for residual metals. This streamlined strategy not only accelerates the timeline for producing high-purity pharmaceutical intermediates but also provides a more sustainable manufacturing footprint, making it an ideal candidate for modern supply chains focused on efficiency and regulatory compliance.
Mechanistic Insights into Organocatalytic Nitro-Michael Addition
The heart of this innovative synthesis lies in the organocatalytic nitro-Michael addition, where a chiral amine catalyst activates an aldehyde substrate to form a reactive enamine intermediate. This enamine then undergoes a stereoselective conjugate addition with a nitroethylene species, which is often generated in situ from a precursor like 2-nitroethyl benzoate. The stereochemical outcome of this reaction is dictated by the specific geometry of the chiral catalyst, such as (S)-diphenyl-prolinol-O-TMS-ether, which creates a steric shield that directs the attack of the nucleophile to one specific face of the electrophile. 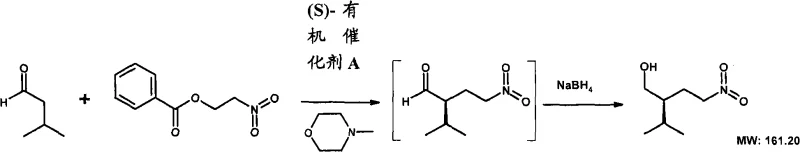 This mechanism ensures the formation of the desired enantiomer with high optical purity, typically achieving enantiomeric excess values that are sufficient for subsequent pharmaceutical applications without extensive recrystallization. The reaction conditions are mild, often proceeding at temperatures ranging from 0°C to room temperature in solvents like toluene or acetonitrile, which preserves the integrity of sensitive functional groups. Following the Michael addition, the resulting nitro-aldehyde is reduced to a nitro-alcohol, setting the stage for the subsequent carbon-carbon bond-forming events. This precise control over stereochemistry at the early stages of the synthesis is crucial, as it establishes the chiral centers that define the biological activity of the final renin inhibitor, thereby minimizing the risk of forming inactive or harmful diastereomers later in the sequence.
This mechanism ensures the formation of the desired enantiomer with high optical purity, typically achieving enantiomeric excess values that are sufficient for subsequent pharmaceutical applications without extensive recrystallization. The reaction conditions are mild, often proceeding at temperatures ranging from 0°C to room temperature in solvents like toluene or acetonitrile, which preserves the integrity of sensitive functional groups. Following the Michael addition, the resulting nitro-aldehyde is reduced to a nitro-alcohol, setting the stage for the subsequent carbon-carbon bond-forming events. This precise control over stereochemistry at the early stages of the synthesis is crucial, as it establishes the chiral centers that define the biological activity of the final renin inhibitor, thereby minimizing the risk of forming inactive or harmful diastereomers later in the sequence.
Following the initial Michael addition, the synthesis proceeds through a nitro-aldol (Henry) reaction to extend the carbon chain and introduce additional functionality required for the final bicyclic structure. The nitro group serves as a versatile handle that can be transformed into an amine via hydrogenation, simultaneously triggering the cyclization to form the lactam ring. The final closure of the lactone ring is achieved through a selective oxidation of a primary alcohol in the presence of a secondary alcohol and the protected amine, often using catalytic systems like TPAP/NMO. 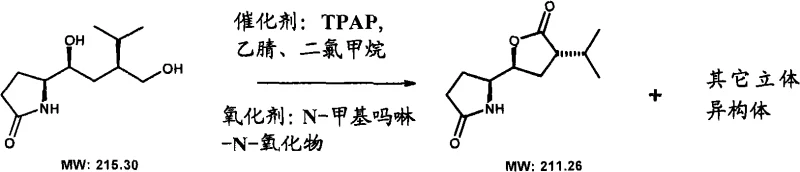 This oxidative cyclization is highly chemoselective, ensuring that the sensitive lactam moiety remains intact while the terminal alcohol is converted to the corresponding carbonyl, which spontaneously cyclizes with the neighboring hydroxyl group. The entire sequence is designed to minimize epimerization, a common issue in base-promoted reactions involving chiral centers alpha to carbonyls. By carefully selecting protecting groups like Boc or benzyl, the process maintains stability throughout the synthetic route, allowing for the isolation of intermediates with defined stereochemistry. This mechanistic robustness is essential for ensuring batch-to-batch consistency, a critical parameter for any reliable pharmaceutical intermediates supplier aiming to meet the strict specifications of global regulatory agencies.
This oxidative cyclization is highly chemoselective, ensuring that the sensitive lactam moiety remains intact while the terminal alcohol is converted to the corresponding carbonyl, which spontaneously cyclizes with the neighboring hydroxyl group. The entire sequence is designed to minimize epimerization, a common issue in base-promoted reactions involving chiral centers alpha to carbonyls. By carefully selecting protecting groups like Boc or benzyl, the process maintains stability throughout the synthetic route, allowing for the isolation of intermediates with defined stereochemistry. This mechanistic robustness is essential for ensuring batch-to-batch consistency, a critical parameter for any reliable pharmaceutical intermediates supplier aiming to meet the strict specifications of global regulatory agencies.
How to Synthesize C-8 Lactam Lactone Efficiently
The synthesis of these valuable intermediates involves a coordinated sequence of organocatalytic transformations that build molecular complexity from simple precursors. The process begins with the activation of an aldehyde using a chiral amine catalyst, followed by the addition of a nitroalkene equivalent to establish the first chiral center. Subsequent steps involve functional group manipulations such as reduction, protection, and chain extension via Henry reactions, culminating in a cascade of hydrogenation and oxidation steps that close both the lactam and lactone rings. The detailed standardized synthesis steps see the guide below, which outlines the specific reagents, conditions, and workup procedures necessary to achieve high yields and purity. This route is particularly advantageous because it avoids the use of cryogenic conditions or pyrophoric reagents, making it accessible for standard chemical manufacturing facilities equipped with常规 reaction vessels. The flexibility of the route also allows for the variation of R1 groups, enabling the production of a library of analogues for structure-activity relationship studies or different renin inhibitor variants.
- Perform an organocatalytic nitro-Michael addition between an aldehyde (e.g., isovaleraldehyde) and a nitroethylene precursor using a chiral amine catalyst to form a nitro-aldehyde intermediate.
- Reduce the resulting nitro-aldehyde to a nitro-alcohol using a reducing agent like sodium borohydride, maintaining stereochemical integrity.
- Execute a nitro-aldol (Henry) reaction followed by hydrogenation and selective oxidation to induce bicyclic cyclization, forming the final lactam-lactone structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this organocatalytic technology offers substantial strategic benefits beyond mere technical feasibility. The shift from stoichiometric chiral auxiliaries to catalytic systems drastically reduces the material cost per kilogram of the intermediate, as expensive chiral reagents are used in minute quantities and can potentially be recovered. This inherent cost efficiency translates directly into a more competitive pricing structure for the final API, allowing pharmaceutical companies to better manage their COGS (Cost of Goods Sold). Furthermore, the elimination of azide chemistry removes a major safety bottleneck, reducing insurance premiums and the need for specialized blast-proof infrastructure, which contributes to significant overhead savings. The simplified purification profile, resulting from the absence of heavy metals and fewer side reactions, shortens the production cycle time and increases the throughput of existing manufacturing assets. These factors combined create a resilient supply chain capable of responding quickly to market demands without compromising on quality or compliance.
- Cost Reduction in Manufacturing: The transition to organocatalysis eliminates the need for purchasing and disposing of large quantities of stoichiometric chiral auxiliaries, which are often costly and generate significant waste. By using catalytic amounts of readily available amines, the process achieves a leaner material balance, directly lowering the variable costs associated with raw material consumption. Additionally, the avoidance of azide reagents reduces the expenses related to hazardous waste disposal and specialized safety protocols. The streamlined synthesis also means fewer unit operations, which decreases energy consumption and labor hours per batch. This holistic reduction in operational complexity ensures that the manufacturing process remains economically viable even when scaling up to multi-ton quantities, providing a clear path for cost reduction in pharmaceutical intermediates manufacturing.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as isovaleraldehyde and simple nitro compounds, are commodity chemicals that are widely available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the organocatalytic reactions means that the process is less sensitive to minor fluctuations in reaction conditions, leading to higher success rates and fewer failed batches that could disrupt supply continuity. The potential for continuous flow processing further enhances reliability by enabling steady-state production that is less prone to the variability seen in batch processing. This stability ensures a consistent flow of high-purity intermediates to downstream API manufacturers, mitigating the risk of production delays caused by raw material shortages or process upsets. Such reliability is crucial for maintaining the uninterrupted supply of life-saving cardiovascular medications to patients worldwide.
- Scalability and Environmental Compliance: This synthetic route is inherently designed for scalability, with reaction conditions that can be easily translated from laboratory glassware to industrial reactors without significant re-engineering. The absence of heavy metal catalysts simplifies the environmental compliance landscape, as there is no need for rigorous testing and removal of trace metals to meet ICH Q3D guidelines. The reduced use of hazardous reagents like azides aligns with increasingly strict environmental regulations regarding worker safety and emissions. Moreover, the ability to run reactions in greener solvents or under solvent-free conditions in certain steps further minimizes the environmental footprint of the manufacturing process. This alignment with green chemistry principles not only facilitates regulatory approval but also enhances the corporate social responsibility profile of the manufacturing entity, making it a preferred partner for sustainability-conscious pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route for C-8 lactam lactones. These answers are derived directly from the technical disclosures within the patent documentation, providing clarity on the practical aspects of the technology. Understanding these details is essential for R&D teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of the supply source. The information covers critical aspects such as stereocontrol, safety improvements, and scalability potential, ensuring that all stakeholders have a comprehensive understanding of the value proposition offered by this advanced manufacturing method.
Q: How does this organocatalytic process improve safety compared to traditional methods?
A: Traditional methods often rely on hazardous azide chemistry to introduce nitrogen atoms. This novel process eliminates the need for dangerous azide reagents, significantly enhancing operational safety and reducing waste treatment complexity.
Q: What level of stereochemical purity can be achieved with this method?
A: The use of chiral organic catalysts, such as diphenylprolinol silyl ethers, allows for high enantiomeric and diastereomeric excess without the need for stoichiometric chiral auxiliaries, ensuring high-purity intermediates suitable for API synthesis.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the patent explicitly mentions that parts of the synthetic route can be adapted for continuous flow processing. This capability, combined with the use of inexpensive starting materials, makes the process highly attractive for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C-8 Lactam Lactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development and commercialization of renin inhibitors. As a dedicated CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the enantiomeric excess required for clinical success. We understand the complexities involved in organocatalytic processes and have the technical expertise to optimize reaction parameters for maximum yield and minimal impurity formation. Our commitment to quality and reliability makes us the ideal partner for bringing your cardiovascular drug candidates from the bench to the market.
We invite you to contact our technical procurement team to discuss how we can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain insights into how implementing this organocatalytic route can optimize your budget without sacrificing quality. We encourage you to reach out for specific COA data and route feasibility assessments tailored to your timeline and volume needs. Let us collaborate to secure a stable and cost-effective supply chain for your next-generation renin inhibitors, ensuring that you can focus on what matters most: delivering effective therapies to patients.
