Advanced Copper-Catalyzed Synthesis of 2-Iodo-Biaryl Phosphonates for Scalable Pharmaceutical Manufacturing
Advanced Copper-Catalyzed Synthesis of 2'-Iodo-Biaryl Phosphonates for Scalable Pharmaceutical Manufacturing
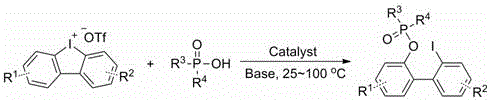
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more efficient, sustainable, and cost-effective synthetic routes for complex organic intermediates. A pivotal advancement in this domain is documented in Chinese Patent CN110437280B, which discloses a novel method for the high-efficiency and high-selectivity synthesis of 2'-iodo[1,1'-biaryl]-2-organophosphonate compounds. These specific organophosphorus structures serve as critical building blocks in the development of bioactive molecules, including kinase inhibitors and other therapeutic agents where phosphonate moieties mimic phosphate groups in biological systems. The patent introduces a transformative approach utilizing inexpensive transition metal copper salts as catalysts, reacting P(O)-OH containing compounds with diaryliodonium triflates in the presence of a base and organic solvent. This technological breakthrough addresses long-standing challenges in the industry, such as the reliance on expensive noble metal catalysts and the use of hazardous reagents, offering a pathway that is not only chemically robust but also commercially viable for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2'-halo[1,1'-biaryl]-2-organophosphonate compounds has been fraught with significant technical and economic hurdles that impede efficient commercialization. Traditional methodologies, such as the Atherton-Todd reaction, often necessitate the use of carbon tetrachloride, a reagent known for its severe environmental toxicity and regulatory restrictions, making it increasingly undesirable for modern green chemistry initiatives. Alternative oxidative dehydrogenation coupling reactions typically rely on air-sensitive P(O)-H compounds, which demand rigorous exclusion of oxygen and moisture, thereby complicating process engineering and increasing operational costs. Furthermore, conventional cross-coupling strategies frequently employ precious metal catalysts like palladium, silver, rhodium, or ruthenium, which are not only exorbitantly priced but also pose challenges regarding residual metal removal to meet stringent pharmaceutical purity standards. These legacy methods often suffer from poor reaction selectivity, cumbersome multi-step procedures, and low overall yields, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these archaic techniques, the methodology outlined in patent CN110437280B presents a streamlined, single-step cross-coupling reaction that leverages the unique reactivity of hypervalent iodine species. By employing diaryliodonium triflates as the electrophilic coupling partners and stable P(O)-OH compounds as the nucleophiles, this new route eliminates the need for air-sensitive reagents and toxic halogenating agents. The reaction proceeds efficiently under mild thermal conditions, typically ranging from 25°C to 100°C, and utilizes readily available copper salts which drastically reduce the raw material cost profile. The versatility of this system is evidenced by its compatibility with a broad spectrum of substrates, tolerating diverse functional groups such as halogens, alkyls, and alkoxy groups without significant loss in performance. This represents a paradigm shift towards atom-economical and environmentally benign synthesis, positioning it as a superior choice for the cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Copper-Catalyzed Cross-Coupling
The mechanistic underpinning of this transformation involves a sophisticated interplay between the copper catalyst and the hypervalent iodine oxidant, facilitating a selective C-P bond formation. It is hypothesized that the copper species, likely cycling between Cu(I) and Cu(III) oxidation states, activates the P(O)-OH substrate through coordination, enhancing its nucleophilicity towards the electron-deficient aryl ring of the diaryliodonium salt. The hypervalent iodine moiety acts as an excellent leaving group, driving the reductive elimination step that forge the critical carbon-phosphorus bond while releasing the iodo-substituted biaryl product. This catalytic cycle is highly efficient, minimizing side reactions such as homocoupling or dehalogenation, which are common pitfalls in transition metal catalysis. The use of a base, such as triethylamine or inorganic carbonates, plays a crucial role in neutralizing the triflic acid byproduct and regenerating the active catalytic species, ensuring the reaction proceeds to completion with high turnover numbers.
From an impurity control perspective, the high selectivity of this copper-catalyzed system is paramount for producing high-purity OLED material or pharmaceutical precursors. The reaction conditions are tuned to suppress the formation of regioisomers and over-phosphorylated byproducts, which simplifies the downstream purification process significantly. By avoiding the use of aggressive oxidants or harsh halogenating agents, the process minimizes the generation of inorganic salts and toxic waste streams, aligning with strict environmental compliance standards. The robustness of the catalyst system ensures consistent batch-to-batch reproducibility, a critical factor for maintaining stringent purity specifications required by global regulatory bodies. This level of control over the reaction trajectory allows manufacturers to deliver products with minimal impurity profiles, reducing the burden on quality control laboratories and accelerating time-to-market for new drug candidates.
How to Synthesize 2'-Iodo[1,1'-biaryl]-2-organophosphonate Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and purity. The process begins with the precise weighing of the P(O)-OH compound and the diaryliodonium triflate substrate, typically in a molar ratio favoring the iodine salt to drive the equilibrium forward. A suitable copper salt, such as cuprous iodide or cuprous bromide, is selected based on optimization data, along with an appropriate organic solvent like tetrahydrofuran or toluene that ensures good solubility of all components. The reaction is conducted under an inert nitrogen atmosphere to prevent any potential oxidation of the catalyst or sensitive intermediates, although the reagents themselves are relatively stable. Following the reaction period, standard workup procedures involving aqueous quenching and organic extraction are employed, followed by purification via column chromatography to isolate the target molecule.
- Mix P(O)-OH compound, diaryliodonium triflate, base, and copper salt in an organic solvent under nitrogen protection.
- Stir the reaction mixture at temperatures between 25°C and 100°C for a duration of 6 to 12 hours.
- Purify the resulting crude product using column chromatography to isolate the target 2'-iodo[1,1'-biaryl]-2-organophosphonate derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed technology offers substantial strategic benefits that extend beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the raw material portfolio, replacing expensive and volatile noble metals with abundant and stable copper salts. This shift not only lowers the direct cost of goods sold but also mitigates the risk associated with the price volatility of precious metals, leading to more predictable budgeting and financial planning. Additionally, the use of stable P(O)-OH precursors eliminates the need for specialized storage infrastructure required for air-sensitive reagents, thereby reducing facility overheads and enhancing operational safety. The streamlined nature of the process, requiring fewer unit operations and milder conditions, translates directly into reduced energy consumption and shorter production cycles, enhancing the overall agility of the supply chain.
- Cost Reduction in Manufacturing: The substitution of palladium or rhodium catalysts with inexpensive copper salts results in a significant decrease in catalyst loading costs, which is particularly impactful when scaling to multi-ton production volumes. Furthermore, the high selectivity of the reaction minimizes the loss of valuable starting materials to byproducts, improving the overall mass balance and atom economy of the process. The elimination of toxic reagents like carbon tetrachloride also reduces the costs associated with hazardous waste disposal and environmental remediation, contributing to a leaner and more sustainable cost structure. These cumulative savings allow for a more competitive pricing strategy in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: Sourcing stable and non-hazardous raw materials ensures a more resilient supply chain that is less susceptible to disruptions caused by regulatory changes or transportation restrictions on dangerous goods. The robustness of the reaction conditions means that the process can be easily transferred between different manufacturing sites without extensive re-validation, providing flexibility in production planning. By reducing the dependency on scarce noble metals, manufacturers can secure long-term supply agreements with greater confidence, ensuring continuity of supply for critical pharmaceutical projects. This reliability is essential for maintaining trust with downstream clients who depend on timely delivery of high-quality intermediates for their own drug development timelines.
- Scalability and Environmental Compliance: The mild reaction temperatures and atmospheric pressure conditions facilitate easy scale-up from kilogram to multi-ton scales without the need for specialized high-pressure reactors or cryogenic cooling systems. The process generates fewer hazardous byproducts, simplifying waste treatment protocols and ensuring compliance with increasingly stringent environmental regulations across different jurisdictions. The use of common organic solvents allows for efficient recovery and recycling, further minimizing the environmental footprint of the manufacturing operation. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate social responsibility profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method, derived directly from the detailed specifications and examples provided in the patent literature. Understanding these nuances is critical for R&D teams evaluating the feasibility of adopting this route for their specific pipeline projects. The answers reflect the empirical data and theoretical advantages demonstrated in the patent, offering a clear picture of the technology's capabilities and limitations in a real-world industrial context.
Q: Why is copper catalysis preferred over palladium for this synthesis?
A: Copper salts are significantly more abundant and cost-effective than noble metals like palladium or rhodium. Furthermore, the copper-catalyzed method described in CN110437280B operates under milder conditions and avoids the need for expensive ligands, reducing overall production costs.
Q: What are the safety advantages of using P(O)-OH compounds over P(O)-H compounds?
A: P(O)-OH compounds are generally stable towards air and moisture, whereas P(O)-H compounds are often air-sensitive and require stringent handling protocols. Using stable P(O)-OH precursors enhances operational safety and simplifies storage requirements in a manufacturing setting.
Q: Does this method support a wide range of functional groups?
A: Yes, the patent demonstrates high substrate tolerance, successfully accommodating various substituents such as methyl, methoxy, fluoro, chloro, and trifluoromethyl groups on the biaryl scaffold without compromising yield or selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2'-Iodo[1,1'-biaryl]-2-organophosphonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN110437280B to revolutionize the production of complex organic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-purity materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2'-iodo[1,1'-biaryl]-2-organophosphonate delivered meets the highest industry standards. We are committed to leveraging our technical expertise to optimize these copper-catalyzed processes, delivering cost-effective solutions that accelerate our partners' drug development programs.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this copper-catalyzed method for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as your trusted partner in bringing high-value pharmaceutical intermediates to market efficiently and sustainably.
