Advanced Aqueous Phase Transfer Catalysis for Commercial Bezafibrate Production
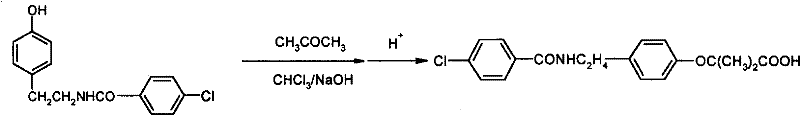
The pharmaceutical industry continuously seeks robust manufacturing pathways that balance high purity with operational efficiency, particularly for lipid-lowering agents like bezafibrate. Patent CN101353315B introduces a transformative preparation method that fundamentally shifts the paradigm from traditional anhydrous conditions to a safer, aqueous-based phase transfer catalysis (PTC) system. This innovation addresses critical bottlenecks in the synthesis of 2-[4-[2-(4-chlorobenzoylamino)ethyl]phenoxy]-2-methylpropionic acid by eliminating the need for rigorous solvent dehydration and hazardous solid base handling. By leveraging immiscible organic solvents alongside aqueous sodium hydroxide, the process achieves superior yields while significantly simplifying the downstream workup. For global procurement teams and R&D directors, this technology represents a strategic opportunity to optimize the supply chain for high-purity pharmaceutical intermediates, ensuring consistent quality and reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of bezafibrate has relied heavily on anhydrous reaction conditions, typically utilizing acetone as both a solvent and a reactant component. In these traditional protocols, solid sodium hydroxide must be mechanically pulverized to enhance its solubility in the organic medium, a step that introduces severe safety hazards due to the generation of corrosive alkaline dust. Furthermore, the requirement for strictly anhydrous environments necessitates energy-intensive distillation and rectification processes to dehydrate the acetone solvent before it can be recycled. These additional unit operations not only extend the overall production cycle time but also increase the capital expenditure required for specialized drying equipment. The cumulative effect of these inefficiencies results in higher operational costs and a greater potential for variability in product quality, posing significant challenges for supply chain reliability in large-scale manufacturing scenarios.
The Novel Approach
In stark contrast, the methodology disclosed in CN101353315B revolutionizes the process by embracing aqueous conditions through the strategic application of phase transfer catalysts. This novel approach allows for the direct use of concentrated sodium hydroxide solutions (30% to 70%), thereby completely bypassing the dangerous and labor-intensive step of solid base pulverization. The reaction system employs organic solvents that are immiscible with water, such as dichloromethane, toluene, or ethyl acetate, which facilitates a clean phase separation post-reaction. Instead of complex distillation trains, the solvent recovery is achieved through simple decantation and washing, allowing the organic phase to be recycled immediately with minimal processing. This shift not only drastically shortens the production timeline but also enhances the inherent safety profile of the plant, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Phase Transfer Catalyzed Condensation
The core chemical innovation lies in the efficient transport of hydroxide ions from the aqueous phase into the organic phase where the substrate resides. By utilizing quaternary ammonium salts, quaternary phosphonium salts, or polyethylene glycols as phase transfer catalysts, the system creates a lipophilic ion pair that shuttles the reactive hydroxide species across the interface. Once in the organic layer, the hydroxide ions deprotonate the phenolic hydroxyl group of the N-p-hydroxyphenethyl-4-chlorobenzamide precursor, generating a highly nucleophilic phenoxide anion. This activated species then undergoes condensation with the chloroform and acetone reagents to construct the critical ether linkage and the gem-dimethyl carboxylic acid moiety characteristic of the bezafibrate structure. The presence of the catalyst ensures that the reaction proceeds rapidly at moderate temperatures, typically between 40°C and 60°C, minimizing thermal degradation of sensitive functional groups.
Controlling the impurity profile is paramount for any reliable bezafibrate supplier, and this aqueous PTC method offers distinct advantages in this regard. The mild reaction conditions and the rapid consumption of reagents prevent the formation of common side products associated with prolonged exposure to harsh alkaline environments. Furthermore, the liquid-liquid extraction workup effectively partitions inorganic salts and water-soluble byproducts into the aqueous waste stream, leaving the organic phase enriched with the desired product. Subsequent acidification of the aqueous extract precipitates the crude bezafibrate, which can be easily purified via recrystallization from acetone to achieve purity levels exceeding 98%. This robust impurity control mechanism ensures that the final active pharmaceutical ingredient meets stringent regulatory specifications without requiring complex chromatographic purification steps.
How to Synthesize Bezafibrate Efficiently
Implementing this synthesis route requires precise control over stoichiometry and temperature to maximize the benefits of the phase transfer system. The process begins by charging the reactor with the amide precursor, the chosen immiscible organic solvent, acetone, and the catalyst, followed by the addition of the aqueous base. Chloroform is then introduced dropwise while maintaining the temperature within the optimal window to manage the exotherm and ensure complete conversion. Detailed standard operating procedures regarding specific molar ratios, agitation speeds, and crystallization parameters are essential for reproducible commercial success. The following guide outlines the standardized synthesis steps derived from the patent examples to assist technical teams in process validation.
- Charge the reactor with N-p-hydroxyphenethyl-4-chlorobenzamide, an immiscible organic solvent (e.g., dichloromethane or toluene), acetone, and a phase transfer catalyst.
- Add aqueous sodium hydroxide solution (30-70% concentration) and heat the mixture to 20-40°C before dropwise addition of chloroform.
- Maintain reaction temperature at 40-60°C for 1-3 hours, then separate layers, acidify the aqueous phase to precipitate the crude product, and recrystallize.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this aqueous PTC technology translates into tangible operational improvements that directly impact the bottom line. By eliminating the need for solvent rectification and solid base handling, the process significantly reduces utility consumption and labor costs associated with hazardous material management. The simplified workflow allows for faster batch turnover, enhancing the overall throughput of the manufacturing facility without requiring additional capital investment in new reactors. Moreover, the use of common, readily available solvents and reagents mitigates the risk of raw material shortages, ensuring a stable and continuous supply of high-purity intermediates for downstream API production.
- Cost Reduction in Manufacturing: The elimination of energy-intensive distillation columns for solvent dehydration represents a major saving in utility costs, as the immiscible solvent system allows for direct recycling via phase separation. Additionally, removing the solid sodium hydroxide crushing step reduces equipment maintenance costs and lowers the insurance premiums associated with handling corrosive powders. The overall simplification of the process flow leads to substantial cost savings in both fixed and variable operational expenditures, making the final product more competitive in the global market.
- Enhanced Supply Chain Reliability: Operating under aqueous conditions removes the sensitivity to ambient humidity that plagues anhydrous processes, thereby reducing the rate of batch failures due to moisture ingress. The robustness of the reaction allows for flexible scheduling and easier scale-up, ensuring that delivery timelines are met consistently even during periods of high demand. This reliability is crucial for maintaining the continuity of supply for critical lipid-lowering medications, minimizing the risk of stockouts for pharmaceutical partners.
- Scalability and Environmental Compliance: The process generates less hazardous waste compared to traditional methods, as the aqueous waste streams are easier to treat and the organic solvents are efficiently recovered. The absence of complex dehydration steps simplifies the plant design, facilitating easier scale-up from pilot batches to multi-ton commercial production. This alignment with green chemistry principles not only reduces the environmental footprint but also ensures compliance with increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented bezafibrate synthesis method. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the technology's capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: How does the aqueous PTC method improve safety compared to traditional anhydrous synthesis?
A: Traditional methods require pulverizing solid sodium hydroxide, creating corrosive dust hazards. This patented method uses aqueous NaOH solutions, eliminating powder handling risks and allowing operation under safer, non-anhydrous conditions.
Q: What are the solvent recovery advantages of this specific bezafibrate process?
A: Unlike conventional routes requiring energy-intensive distillation and rectification to remove water, this process uses water-immiscible solvents. The organic layer is simply separated, washed, and reused directly, drastically cutting energy consumption.
Q: Can this synthesis route be scaled for multi-ton commercial production?
A: Yes, the process is explicitly designed for industrialization. It avoids complex dehydration steps and utilizes standard liquid-liquid extraction techniques, making it highly scalable from pilot plants to 100 MT annual capacity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bezafibrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the pharmaceutical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the aqueous PTC method for bezafibrate are translated into reality with precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the highest international standards, providing our partners with the confidence needed for successful regulatory filings.
We invite forward-thinking organizations to collaborate with us to leverage these technological advancements for their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can drive value and efficiency for your organization.
