Green Synthesis of 2-Amino-6-Chloroguanine: A Commercial Scale-Up Strategy for Antiviral Intermediates
Green Synthesis of 2-Amino-6-Chloroguanine: A Commercial Scale-Up Strategy for Antiviral Intermediates
The global demand for antiviral therapeutics, particularly nucleoside analogs like famciclovir, has necessitated a re-evaluation of supply chain resilience and manufacturing efficiency for key precursors. Patent CN113292507B introduces a transformative preparation method for 2-amino-6-chloroguanine, a critical intermediate in the synthesis of famciclovir and other purine-based antiviral agents. This technology represents a significant departure from legacy manufacturing protocols by replacing expensive guanine starting materials with more accessible pyrimidine derivatives and substituting hazardous phosphorus oxychloride with triphosgene. For R&D directors and procurement strategists, this patent offers a blueprint for a greener, more cost-effective production pathway that mitigates the severe environmental liabilities associated with high-phosphorus wastewater. The following analysis dissects the technical merits and commercial viability of this novel synthetic route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2-amino-6-chloroguanine has relied heavily on direct chlorination of guanine using phosphorus oxychloride (POCl3) or reactions involving phosphorus pentasulfide. These conventional pathways are plagued by substantial economic and environmental inefficiencies that burden modern supply chains. Firstly, guanine itself is a high-cost raw material, which inherently inflates the Bill of Materials (BOM) for the final active pharmaceutical ingredient (API). Secondly, the use of POCl3 generates massive quantities of acidic, high-phosphorus wastewater that requires complex and expensive neutralization and treatment processes before discharge. Furthermore, older methods often suffer from inconsistent yields, typically ranging between 30% and 60%, and involve unstable thio-intermediates that complicate process control. The reliance on phase transfer catalysts and extended reaction times exceeding 28 hours further diminishes the throughput capacity of manufacturing facilities, creating bottlenecks in the supply of essential antiviral intermediates.
The Novel Approach
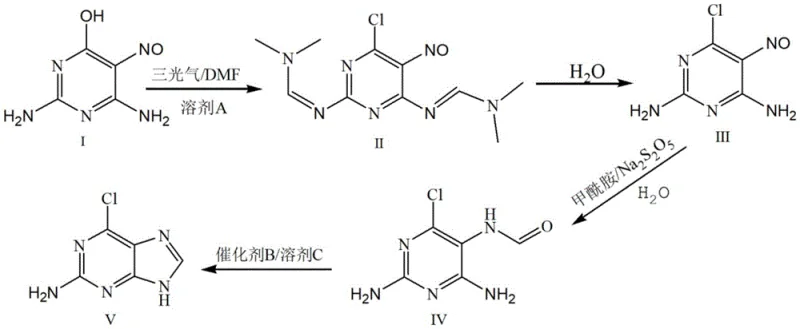
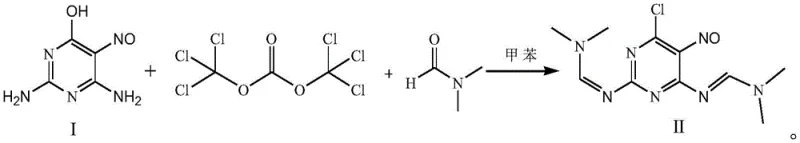
The methodology disclosed in CN113292507B fundamentally重构 s the synthetic logic by initiating the sequence with 2,4-diamino-5-nitroso-6-hydroxypyrimidine (Compound I) rather than guanine. This strategic shift eliminates the dependency on expensive purine starting materials and bypasses the generation of phosphorus-containing waste entirely. The core innovation lies in the initial chlorination step, where triphosgene is employed as the chlorinating agent in the presence of N,N-Dimethylformamide (DMF). As illustrated in the reaction scheme below, this approach facilitates a smooth transformation to the dichloro-intermediate (Compound II) under controlled thermal conditions. By avoiding phosphorus reagents, the process drastically simplifies wastewater treatment protocols, converting a major environmental liability into a manageable operational expense. Additionally, the overall molar yield of this new route is reported to exceed 90%, representing a substantial improvement in atom economy and resource utilization compared to traditional benchmarks.
Mechanistic Insights into Triphosgene-Mediated Chlorination and Cyclization
A deep dive into the reaction mechanism reveals the sophisticated role of DMF in this synthesis, acting as more than just a solvent. In the initial stage, DMF reacts with the hydroxyl group of the substrate (Compound I) to form a protected intermediate species. This transient protection significantly enhances the solubility of the substrate in organic media such as toluene, overcoming the inherent insolubility that hampers direct chlorination attempts. Once solubilized, the protected intermediate undergoes efficient chlorination with triphosgene at temperatures between 105°C and 115°C. The use of triphosgene, a solid phosgene equivalent, allows for precise dosing and safer handling compared to gaseous reagents, ensuring consistent reaction kinetics. Following the formation of Compound II, the process proceeds through a carefully controlled hydrolysis step at pH 4-6. This specific acidity range is critical; it is sufficient to cleave the protecting groups and hydrolyze the imine functionalities to reveal the amino groups in Compound III without degrading the sensitive nitroso or chloro substituents on the pyrimidine ring.
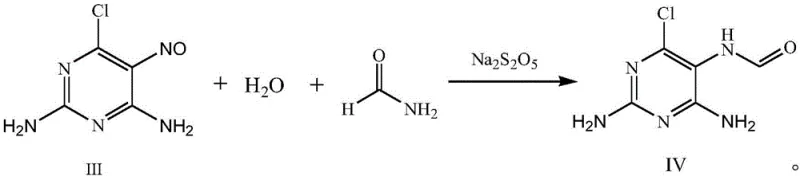
The subsequent transformation of Compound III to Compound IV involves a reduction-acylation sequence utilizing sodium metabisulfite and formamide. This step is pivotal for constructing the imidazole ring precursor required for the final guanine structure. The reduction potential of sodium metabisulfite effectively manages the nitroso group, while formamide serves as the one-carbon source for ring closure. Finally, the cyclization of Compound IV to the target 2-amino-6-chloroguanine is achieved through acid-catalyzed dehydration. The mechanistic elegance of this route lies in its modularity; each step generates intermediates that can be isolated or carried forward in solution, offering flexibility for process optimization. The avoidance of heavy metal catalysts and the use of recyclable solvents like toluene further underscore the green chemistry principles embedded in this design, ensuring that the impurity profile remains clean and easy to manage during downstream purification.
How to Synthesize 2-Amino-6-Chloroguanine Efficiently
Implementing this synthesis route requires strict adherence to the thermal and stoichiometric parameters defined in the patent to ensure maximum yield and purity. The process is designed to be scalable, moving seamlessly from laboratory verification to pilot plant operations. The initial chlorination must be conducted with slow addition of the triphosgene solution to prevent exothermic runaway, maintaining the temperature within the narrow window of 105-115°C. Subsequent hydrolysis and reduction steps rely on precise pH control and temperature ramping to drive the reactions to completion while minimizing side products. For detailed operational protocols, including specific solvent volumes, stirring rates, and workup procedures, please refer to the standardized synthesis guide below which outlines the critical process parameters for each stage of the transformation.
- Dissolve 2,4-diamino-5-nitroso-6-hydroxypyrimidine in solvent A and DMF, then react with triphosgene at 105-115°C to form intermediate Compound II.
- Hydrolyze the aqueous solution of Compound II under weakly acidic conditions (pH 4-6) at 70-80°C to precipitate intermediate Compound III.
- Perform reduction-acylation on Compound III using formamide and sodium metabisulfite at elevated temperatures to generate intermediate Compound IV.
- Cyclize Compound IV using a catalyst and solvent C under reflux with dehydration to obtain the final 2-amino-6-chloroguanine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers compelling strategic advantages that extend beyond simple unit cost calculations. The primary value driver is the radical reduction in raw material expenditure achieved by substituting high-cost guanine with commodity-grade pyrimidine derivatives. This shift insulates the supply chain from the price volatility often associated with specialized purine starting materials. Furthermore, the elimination of phosphorus oxychloride removes the need for expensive corrosion-resistant equipment and complex effluent treatment systems dedicated to phosphate removal. This simplification of the manufacturing infrastructure translates directly into lower capital expenditure (CAPEX) and reduced operational overheads. The robustness of the reaction conditions also implies a higher success rate in batch production, reducing the risk of costly batch failures and ensuring a more reliable delivery schedule for downstream API manufacturers.
- Cost Reduction in Manufacturing: The replacement of guanine with 2,4-diamino-5-nitroso-6-hydroxypyrimidine results in a significantly lower raw material cost base. Additionally, the use of triphosgene instead of phosphorus oxychloride eliminates the generation of high-phosphorus wastewater, thereby removing the substantial costs associated with specialized chemical waste disposal and neutralization. The high molar yield reported (>90%) means that less raw material is wasted per kilogram of finished product, further enhancing the overall economic efficiency of the manufacturing process.
- Enhanced Supply Chain Reliability: By utilizing widely available solvents like toluene and DMF, along with stable solid reagents like triphosgene and sodium metabisulfite, the process reduces dependency on hazardous or hard-to-source chemicals. The simplified workflow, which avoids unstable thio-intermediates found in older routes, ensures greater process stability and consistency. This reliability is crucial for maintaining continuous production schedules and meeting the rigorous Just-In-Time delivery requirements of large-scale pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction conditions that are easily managed in standard stainless steel reactors. The absence of heavy metals and phosphorus byproducts simplifies regulatory compliance regarding environmental discharge limits. Solvent recovery systems can be easily integrated to recycle toluene and DMF, aligning the manufacturing process with modern sustainability goals and reducing the carbon footprint of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on yield expectations, impurity management, and process safety. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for procurement teams assessing the long-term viability of this supply source.
Q: Why is this new synthesis route preferred over the traditional guanine-based method?
A: The traditional method uses expensive guanine and phosphorus oxychloride, generating difficult-to-treat high-phosphorus wastewater. This new route uses cheaper pyrimidine derivatives and triphosgene, significantly reducing raw material costs and environmental impact while achieving yields over 90%.
Q: How does the process handle the solubility issues of the starting material?
A: The process utilizes N,N-Dimethylformamide (DMF) not just as a solvent but as a protective agent that forms a soluble intermediate. This ensures the substrate dissolves effectively in organic solvents like toluene, allowing the chlorination reaction with triphosgene to proceed smoothly without precipitation issues.
Q: What are the purity specifications achievable with this method?
A: Experimental data from the patent indicates that the final 2-amino-6-chloroguanine product can achieve a purity of 99.0% after standard recrystallization and decolorization steps, meeting stringent quality requirements for antiviral drug synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-6-Chloroguanine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is critical for the future of the pharmaceutical industry. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN113292507B are fully realized in practical manufacturing environments. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-amino-6-chloroguanine meets the exacting standards required for antiviral drug synthesis. Our commitment to quality and consistency makes us a trusted partner for global pharmaceutical companies seeking to optimize their supply chains.
We invite you to collaborate with us to explore how this advanced synthesis method can benefit your specific production needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us to request specific COA data, route feasibility assessments, and samples to validate the superior quality of our intermediates. Let us help you secure a sustainable and cost-effective supply of high-purity 2-amino-6-chloroguanine for your next generation of antiviral therapies.
